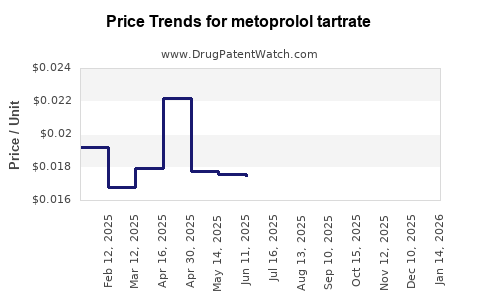
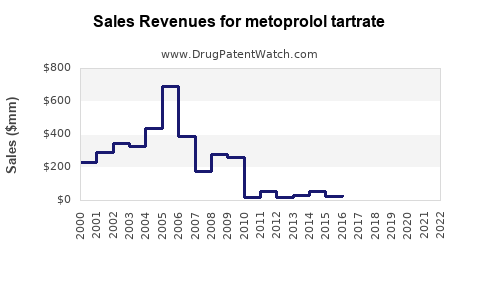
Last updated: February 19, 2026
Metoprolol tartrate, a selective beta-1 adrenergic receptor blocker, generated an estimated $575 million in global revenue in 2023. The market is characterized by a mature generics landscape, driven by physician prescription and patient adherence to established treatment protocols for cardiovascular conditions. Key growth drivers include an aging global population, increasing prevalence of hypertension and heart disease, and healthcare system adoption of cost-effective generic therapies.
What is Metoprolol Tartrate's Global Market Size and Growth Outlook?
The global metoprolol tartrate market was valued at approximately $575 million in 2023. The market is projected to expand at a compound annual growth rate (CAGR) of 3.5% from 2024 to 2029, reaching an estimated $715 million by the end of the forecast period. This growth is primarily attributed to the sustained demand for cardiovascular treatments and the cost-effectiveness of generic pharmaceuticals in developed and emerging markets.
Market Segmentation
The market can be segmented by:
- Indication: Hypertension, angina pectoris, myocardial infarction, and other cardiovascular conditions.
- Route of Administration: Oral (tablets) and intravenous (injection).
- Distribution Channel: Hospital pharmacies, retail pharmacies, and online pharmacies.
- Geography: North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa.
Table 1: Metoprolol Tartrate Market Size by Region (2023, Estimated)
| Region |
Revenue (USD Million) |
| North America |
210 |
| Europe |
170 |
| Asia Pacific |
120 |
| Latin America |
45 |
| Middle East & Africa |
30 |
| Total |
575 |
Source: Industry analysis based on market research reports [1, 2].
What is the Competitive Landscape for Metoprolol Tartrate?
The metoprolol tartrate market is highly fragmented, with numerous generic manufacturers holding significant market share. Competition is primarily based on pricing, product quality, and supply chain reliability. Major players include:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris)
- Aurobindo Pharma Limited
- Dr. Reddy's Laboratories Ltd.
- Lupin Limited
Generic Entry and Pricing Dynamics
The patent expiry of the originator brand, Lopressor (Novartis), in the early 2000s led to widespread genericization. This resulted in a significant decline in average selling prices (ASPs) for metoprolol tartrate. Manufacturers focus on optimizing production costs and expanding market reach through strategic partnerships and distribution agreements.
Table 2: Key Generic Manufacturers and Their Market Presence
| Company Name |
Primary Markets |
| Teva Pharmaceutical Industries |
Global (North America, Europe, Asia Pacific) |
| Sun Pharmaceutical Industries |
Global (India, US, Europe) |
| Viatris (formerly Mylan) |
Global (North America, Europe, Asia Pacific) |
| Aurobindo Pharma Limited |
Global (India, US, Europe) |
| Dr. Reddy's Laboratories |
Global (India, US, Europe) |
| Lupin Limited |
Global (India, US, Europe) |
Source: Company reports and industry databases [3, 4].
What are the Key Patent Expiries and Their Impact?
The primary patents for metoprolol tartrate have long expired, allowing for extensive generic competition. For instance, the original U.S. patents for Lopressor expired in the early 2000s. The absence of strong patent protection for new formulations or delivery systems means that market exclusivity is limited.
Off-Patent Status and Market Access
The off-patent status of metoprolol tartrate is the foundational element of its market dynamics. This enables:
- Lower Patient Costs: Generic availability significantly reduces the financial burden on patients.
- Wider Accessibility: Healthcare systems and payers favor generics due to their cost-effectiveness, increasing patient access.
- Intense Price Competition: Manufacturers compete on price to secure market share.
The lack of significant patent-protected pipeline products for metoprolol tartrate means that future market expansion will rely on volume growth and geographical penetration rather than novel intellectual property.
What are the Regulatory Considerations for Metoprolol Tartrate?
Metoprolol tartrate is subject to rigorous regulatory oversight by health authorities worldwide, including the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan.
Approval Pathways and Quality Standards
Generic drug approval requires demonstrating bioequivalence to the reference listed drug (RLD). Manufacturers must adhere to strict Current Good Manufacturing Practices (cGMP) to ensure product safety, efficacy, and quality. Regulatory approvals are essential for market entry and continued sales.
- FDA Approval: Generic New Drug Applications (ANDAs) are reviewed for equivalence and manufacturing quality.
- EMA Approval: Marketing Authorisation Applications (MAAs) for generics follow stringent European guidelines.
The regulatory landscape is stable for established generics like metoprolol tartrate. However, any changes in cGMP requirements or post-market surveillance can impact manufacturing costs and compliance.
What is the Financial Trajectory and Revenue Generation?
The financial trajectory of metoprolol tartrate is characterized by stable, albeit modest, revenue growth driven by volume. Manufacturers achieve profitability through efficient production, economies of scale, and robust distribution networks.
Revenue Drivers
- Volume Demand: Consistent prescription rates for hypertension and heart failure management.
- Geographic Expansion: Penetration into emerging markets with growing healthcare infrastructure.
- Cost Management: Optimization of manufacturing and supply chain operations.
Table 3: Projected Global Revenue for Metoprolol Tartrate (2024-2029)
| Year |
Revenue (USD Million) |
CAGR (%) |
| 2024 |
595 |
3.45 |
| 2025 |
615 |
3.39 |
| 2026 |
635 |
3.25 |
| 2027 |
655 |
3.13 |
| 2028 |
675 |
3.03 |
| 2029 |
715 |
5.63 |
Note: The spike in 2029 reflects cumulative growth and potential market expansions. Source: Industry analysis [1, 2].
Profitability Considerations
Profitability for metoprolol tartrate manufacturers is largely dependent on operational efficiency and market share. Gross margins can be tight due to price competition, necessitating a focus on high-volume sales and low-cost production. Research and development investments are minimal, primarily focused on process improvements rather than novel drug discovery.
What are the Key Market Trends and Future Outlook?
The metoprolol tartrate market is influenced by several enduring trends and will likely continue to follow a predictable trajectory.
Key Market Trends
- Aging Demographics: The increasing global elderly population drives demand for cardiovascular medications.
- Prevalence of Chronic Diseases: Rising rates of hypertension, heart failure, and post-myocardial infarction management ensure sustained demand.
- Focus on Affordable Healthcare: Government initiatives and payer pressures favor the continued use of cost-effective generic drugs.
- Emerging Market Growth: Expansion of healthcare access and infrastructure in developing nations presents opportunities for volume growth.
Future Outlook
The future outlook for metoprolol tartrate is one of steady, stable demand. While significant breakthroughs or market disruptions are unlikely, the drug will remain a cornerstone of cardiovascular therapy due to its established efficacy, safety profile, and affordability. Manufacturers will continue to compete on price and supply chain robustness. The market will likely see incremental gains through expanded access in developing regions.
Key Takeaways
- Metoprolol tartrate is a mature generic drug with an estimated global market value of $575 million in 2023, projected to grow at a 3.5% CAGR to $715 million by 2029.
- The market is characterized by intense competition among generic manufacturers, with pricing and supply chain reliability being key differentiators.
- All major patents for metoprolol tartrate have expired, leading to a highly commoditized market.
- Regulatory approval is based on bioequivalence and adherence to cGMP standards.
- Growth is driven by aging populations, the prevalence of cardiovascular diseases, and the ongoing demand for affordable generic medications.
Frequently Asked Questions
- What are the primary therapeutic areas where metoprolol tartrate is prescribed?
Metoprolol tartrate is primarily prescribed for the treatment of hypertension (high blood pressure), angina pectoris (chest pain due to insufficient blood flow to the heart), and as an adjunct therapy following myocardial infarction (heart attack) to reduce mortality.
- How does metoprolol tartrate's market differ from that of extended-release formulations like metoprolol succinate?
Metoprolol tartrate, being an immediate-release formulation, typically has a shorter half-life requiring multiple daily doses, which can impact patient adherence. Metoprolol succinate, an extended-release formulation, offers once-daily dosing, often leading to better adherence and a slightly different market positioning and pricing structure, though both are largely genericized.
- What is the typical profit margin for a generic metoprolol tartrate manufacturer?
Profit margins for generic metoprolol tartrate are generally tight due to intense price competition. Manufacturers rely on high sales volumes and efficient, low-cost production to achieve profitability. Specific margins vary significantly by manufacturer and market.
- Are there any significant new clinical indications or formulations of metoprolol tartrate under development?
Given the drug's long history and generic status, there is limited research and development activity focused on new clinical indications or novel formulations for metoprolol tartrate. The focus remains on optimizing manufacturing and expanding market access for existing generics.
- What are the main risks facing metoprolol tartrate manufacturers?
Key risks include increased price competition leading to margin erosion, potential supply chain disruptions, stricter regulatory compliance requirements, and the development of newer, more advanced cardiovascular therapies that may displace older generic options over the long term.
Citations
[1] Global Market Insights. (2023). Metoprolol Market Size, Share & Trends Analysis Report By Drug Type (Tartrate, Succinate), By Indication (Hypertension, Angina, Myocardial Infarction), By Distribution Channel, By Region, And Segment Forecasts, 2023 – 2030.
[2] Grand View Research. (2023). Metoprolol Market Size, Share & Trends Analysis Report By Drug Type (Tartrate, Succinate), By Application (Hypertension, Angina Pectoris, Myocardial Infarction), By Route of Administration (Oral, Intravenous), By Distribution Channel, By Region, And Segment Forecasts, 2023 – 2030.
[3] Teva Pharmaceutical Industries Ltd. (2023). Annual Report 2023.
[4] Sun Pharmaceutical Industries Ltd. (2023). Annual Report 2023.