Last updated: February 19, 2026
Letrozole, a nonsteroidal aromatase inhibitor, is a critical pharmaceutical agent primarily utilized in the treatment of hormone receptor-positive breast cancer in postmenopausal women. Its efficacy in reducing estrogen levels, a key driver of tumor growth in such cancers, has established its position in oncological therapy. The market trajectory of letrozole is significantly influenced by its patent landscape, regulatory approvals, and the competitive environment shaped by generic entry.
What is the Current Market Size and Projected Growth for Letrozole?
The global market for letrozole is substantial, driven by the high incidence of breast cancer and its established role in adjuvant and metastatic treatment regimens. Market valuation is influenced by prescription volumes, pricing strategies, and the penetration of both originator and generic products.
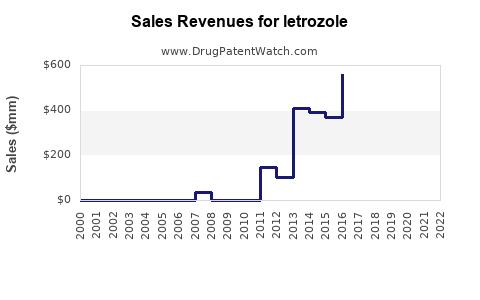
The worldwide market for letrozole was valued at approximately $650 million in 2022 [1]. Projections indicate a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030, with an estimated market size reaching $920 million by 2030 [1]. This growth is attributed to increasing breast cancer diagnoses, expanding access to oncology treatments in emerging markets, and ongoing research exploring its potential in other therapeutic areas.
Who are the Key Players and What is the Competitive Landscape?
The letrozole market is characterized by the presence of originator companies and a significant number of generic manufacturers. The expiration of primary patents has opened the market to extensive generic competition, leading to price erosion and a focus on market share.
The originator product for letrozole is Femara®, originally developed by Novartis AG [2]. Following patent expirations, numerous generic manufacturers have entered the market. Key generic players include:
- Teva Pharmaceutical Industries Ltd. [3]
- Dr. Reddy's Laboratories Ltd. [3]
- Sun Pharmaceutical Industries Ltd. [3]
- Aurobindo Pharma Ltd. [3]
- Mylan N.V. (now Viatris Inc.) [3]
- Zydus Lifesciences Ltd. [3]
The competitive landscape is characterized by a mature market with established products and ongoing price negotiations with healthcare providers and pharmacy benefit managers. Market share is often dictated by manufacturing scale, distribution networks, and the ability to offer competitive pricing.
What is the Intellectual Property Landscape for Letrozole?
The patent protection for letrozole has largely expired in major markets, paving the way for widespread generic availability. The initial patent for letrozole was filed by the Research Institute of Scripps Clinic and covered the compound itself.
Key patent information and expiration timelines in major jurisdictions are critical for understanding market exclusivity:
- United States: The primary compound patents for letrozole have expired. While method-of-use patents may exist, they generally offer limited protection against generic competition once the core compound is off-patent [4]. The FDA approved the first generic versions of letrozole in the early 2000s.
- Europe: Similar to the U.S., the main patent protection for letrozole has expired across European Union member states. European Patent EP0297572, related to the compound, expired years ago [4].
- Other Jurisdictions: Patent expiries in other significant markets, including Canada, Australia, and Japan, have also facilitated generic entry.
The expiration of these core patents has fundamentally shifted the market dynamics, moving from a period of patent-protected revenue for the originator to a highly competitive generic environment.
What are the Regulatory Considerations and Approvals for Letrozole?
Letrozole is approved by major regulatory bodies worldwide for its indicated uses. Regulatory approval is a prerequisite for market entry, and post-approval, manufacturers must adhere to manufacturing standards and pharmacovigilance requirements.
Key regulatory approvals and considerations include:
- U.S. Food and Drug Administration (FDA): Letrozole is approved for treating postmenopausal women with hormone receptor-positive, HER2-negative early breast cancer. It is also approved for extended adjuvant treatment and for first-line treatment of postmenopausal women with advanced or metastatic breast cancer, either alone or in combination with other therapies [5].
- European Medicines Agency (EMA): Similar indications are approved by the EMA, allowing its use in breast cancer treatment across EU member states [6].
- Other Regulatory Agencies: Approvals are held by Health Canada, the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan, and other national regulatory authorities globally.
Generic manufacturers must obtain Abbreviated New Drug Applications (ANDAs) in the U.S. or equivalent marketing authorizations in other regions, demonstrating bioequivalence to the reference listed drug (Femara®).
What are the Therapeutic Uses and Market Drivers for Letrozole?
Letrozole's primary therapeutic role is in oncology, specifically for breast cancer. Its mechanism of action and established clinical benefits drive its demand.
The main therapeutic uses and market drivers include:
- Adjuvant Therapy: Used after surgery to reduce the risk of cancer recurrence in postmenopausal women with early-stage, hormone receptor-positive breast cancer [5].
- Extended Adjuvant Therapy: Used following standard adjuvant endocrine therapy to further reduce the risk of recurrence [5].
- Metastatic Breast Cancer: Used as a first-line or second-line treatment for advanced or metastatic hormone receptor-positive breast cancer [5].
- Fertility Treatment: Off-label use in women of reproductive age to induce ovulation as part of assisted reproductive technologies. This represents a smaller but growing segment of demand [7].
Market drivers are primarily linked to:
- Rising Incidence of Breast Cancer: Global increases in breast cancer diagnoses directly correlate with demand for effective endocrine therapies.
- Aging Global Population: The demographic shift towards older populations, where breast cancer is more prevalent, increases the patient pool.
- Clinical Guideline Recommendations: Inclusion in established treatment guidelines by organizations like the National Comprehensive Cancer Network (NCCN) and the European Society for Medical Oncology (ESMO) ensures continued prescribing [8].
- Cost-Effectiveness of Generics: The availability of affordable generic letrozole makes it a preferred choice for many healthcare systems and patients.
What is the Impact of Generic Competition on Letrozole Pricing and Revenue?
The introduction of generic letrozole has led to significant price reductions and a shift in revenue generation dynamics from originator to generic manufacturers.
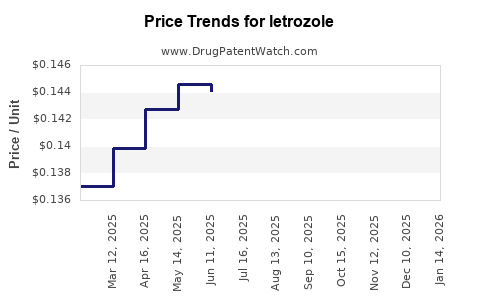
- Price Erosion: Upon the entry of multiple generic competitors, the average selling price (ASP) of letrozole has decreased substantially. For instance, in the U.S., the price of generic letrozole tablets can range from $0.50 to $3.00 per tablet, depending on the manufacturer and volume, a stark contrast to the originator's pricing pre-genericization [9].
- Revenue Shift: Originator revenue from Femara® has declined significantly post-patent expiry. Revenue generation now primarily accrues to generic manufacturers who compete on volume and price.
- Market Volume: While prices have decreased, the overall volume of letrozole prescriptions has likely increased due to greater affordability and broader access.
The market is now characterized by high-volume, low-margin sales for generic producers. Profitability for these companies is dependent on efficient manufacturing, supply chain management, and securing favorable contracts.
What are the Future Outlook and Potential Opportunities for Letrozole?
The future of the letrozole market is largely defined by its established role in breast cancer treatment and emerging applications, alongside the ongoing dynamics of the generic pharmaceutical sector.
- Continued Demand in Oncology: Letrozole is expected to maintain its position as a standard-of-care treatment for hormone receptor-positive breast cancer. Its efficacy and safety profile are well-established, ensuring continued prescription volumes.
- Exploration in New Indications: Research into letrozole's potential in other cancers, such as endometrial cancer, or in combination therapies for more complex breast cancer subtypes, could create new market opportunities. Studies have explored its use in gynecological conditions beyond fertility, though widespread regulatory approval for these indications is limited [10].
- Geographic Expansion: Increasing healthcare infrastructure and access to advanced cancer treatments in emerging economies will drive demand for established generics like letrozole.
- Competition and Innovation: While the core compound is off-patent, innovation might emerge in formulation, drug delivery systems, or combination products that offer enhanced patient compliance or efficacy. However, the primary market will remain dominated by generic oral tablets.
The market's maturity suggests that significant revenue growth will be driven by volume increases and geographic penetration rather than price increases or new, high-value indications.
Key Takeaways
- Letrozole's global market was valued at approximately $650 million in 2022 and is projected to grow at a CAGR of 4.5% to reach $920 million by 2030.
- The market is highly competitive, dominated by generic manufacturers following the expiration of key patents for the originator product, Femara®.
- Major regulatory bodies, including the FDA and EMA, have approved letrozole for various stages of hormone receptor-positive breast cancer treatment.
- Primary market drivers include the rising incidence of breast cancer, an aging global population, and established clinical guideline recommendations.
- Generic entry has led to substantial price erosion, shifting revenue generation to high-volume, lower-margin sales for generic producers.
- Future growth will likely stem from continued demand in oncology, potential exploration of new indications, and geographic market expansion.
FAQs
1. What is the primary therapeutic use of letrozole?
Letrozole is primarily used in the treatment of hormone receptor-positive breast cancer in postmenopausal women.
2. When did the patents for letrozole's originator product, Femara®, expire in major markets?
The key compound patents for Femara® expired in major markets, including the U.S. and Europe, in the early to mid-2000s, allowing for the entry of generic versions.
3. Besides breast cancer, are there any other significant therapeutic applications for letrozole?
Letrozole is also used off-label to induce ovulation in fertility treatments. Research continues into potential uses in other gynecological conditions and cancers, though these are not yet widespread approved indications.
4. How has the entry of generic letrozole impacted its price?
The widespread availability of generic letrozole has led to significant price reductions, with generic tablet prices often falling below $3.00 per tablet, compared to significantly higher prices for the originator product.
5. What is the expected trajectory of the letrozole market in the next five to ten years?
The letrozole market is expected to experience steady growth driven by increasing breast cancer diagnoses and its established role in treatment protocols. Growth will primarily be volume-driven, with generic competition continuing to define the pricing landscape.
Citations
[1] Global Market Insights. (2023). Letrozole Market Size, Share & Trends Analysis Report.
[2] U.S. Food and Drug Administration. (n.d.). Femara® (letrozole) tablets. Retrieved from FDA drug database.
[3] Various Pharmaceutical Company Reports and Investor Filings. (2022-2023).
[4] European Patent Office. (n.d.). Patent Register. Retrieved from EPO database.
[5] National Cancer Institute. (2021). Letrozole. Retrieved from NCI website.
[6] European Medicines Agency. (n.d.). European Public Assessment Reports (EPARs). Retrieved from EMA website.
[7] American College of Obstetricians and Gynecologists. (2018). Committee Opinion No. 711: Use of Aromatase Inhibitors for Ovarian Stimulation.
[8] National Comprehensive Cancer Network. (2023). NCCN Clinical Practice Guidelines in Oncology: Breast Cancer.
[9] GoodRx. (2023). Letrozole Prices, Coupons, and Patient Assistance Programs. Retrieved from GoodRx website.
[10] Webb, A. L., et al. (2019). Aromatase inhibitors in gynecologic oncology: current and future applications. International Journal of Gynecological Cancer, 29(7), 1173-1180.