Fingolimod - Generic Drug Details
✉ Email this page to a colleague
Summary for fingolimod
| International Patents: | 137 |
| US Patents: | 3 |
| Tradenames: | 1 |
| Applicants: | 1 |
| NDAs: | 1 |
| Drug Master File Entries: | 20 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 44 |
| Clinical Trials: | 52 |
| Patent Applications: | 2,547 |
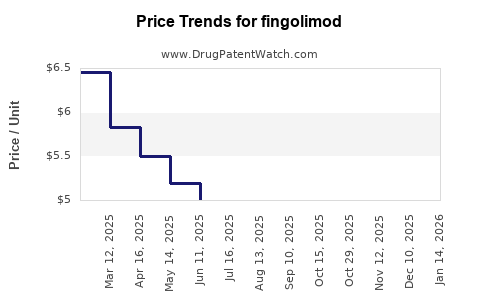
| Drug Prices: | Drug price trends for fingolimod |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for fingolimod |
| What excipients (inactive ingredients) are in fingolimod? | fingolimod excipients list |
| DailyMed Link: | fingolimod at DailyMed |
Recent Clinical Trials for fingolimod
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| TG Therapeutics, Inc. | PHASE2 |
| Novartis | PHASE2 |
| Arizona State University | PHASE2 |
Generic filers with tentative approvals for FINGOLIMOD
| Applicant | Application No. | Strength | Dosage Form |
| ⤷ Start Trial | ⤷ Start Trial | EQ 0.5MG BASE | CAPSULE;ORAL |
| ⤷ Start Trial | ⤷ Start Trial | EQ 0.5MG BASE | CAPSULE;ORAL |
| ⤷ Start Trial | ⤷ Start Trial | 0.5MG | CAPSULE;ORAL |
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.
US Patents and Regulatory Information for fingolimod
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Teva Pharms Usa | FINGOLIMOD HYDROCHLORIDE | fingolimod hydrochloride | CAPSULE;ORAL | 212152-001 | Nov 12, 2021 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Cycle | TASCENSO ODT | fingolimod lauryl sulfate | TABLET, ORALLY DISINTEGRATING;ORAL | 214962-001 | Dec 23, 2021 | RX | Yes | No | 10,925,829 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Hetero Labs Ltd V | FINGOLIMOD HYDROCHLORIDE | fingolimod hydrochloride | CAPSULE;ORAL | 207933-001 | May 18, 2020 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Accord Hlthcare | FINGOLIMOD HYDROCHLORIDE | fingolimod hydrochloride | CAPSULE;ORAL | 207991-001 | Oct 28, 2020 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Glenmark Pharms Ltd | FINGOLIMOD HYDROCHLORIDE | fingolimod hydrochloride | CAPSULE;ORAL | 207985-001 | Jun 18, 2020 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Cycle | TASCENSO ODT | fingolimod lauryl sulfate | TABLET, ORALLY DISINTEGRATING;ORAL | 214962-002 | Dec 9, 2022 | RX | Yes | Yes | 10,925,829 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |