Last updated: February 19, 2026
Benzontate is a non-narcotic antitussive medication used to relieve cough. Its market trajectory is shaped by patent expirations, generic competition, and evolving treatment guidelines for cough and cold symptoms.
What is Benzontate's Current Market Position?
Benzontate is an established, over-the-counter (OTC) and prescription cough suppressant. Its primary mechanism of action involves anesthetizing stretch receptors in the respiratory tract, thereby reducing the urge to cough. It is available in various formulations, including capsules, liquid gels, and solutions.
Key Market Characteristics:
- Established Efficacy: Benzontate has a long history of clinical use and recognized efficacy in symptomatic relief of cough.
- Non-Opioid Mechanism: Its non-narcotic status is a significant differentiator, particularly in a market where opioid-based cough suppressants are subject to strict regulation and concerns about abuse.
- Broad Accessibility: Available as both a prescription medication and in some OTC formulations, it reaches a wide patient population.
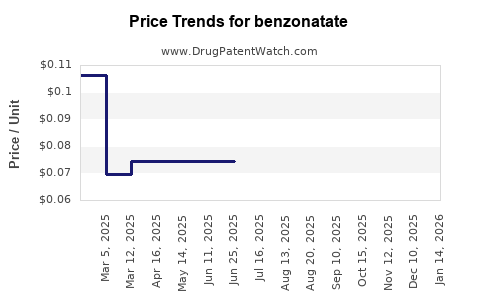
- Generic Penetration: The market has experienced significant generic penetration following patent expirations, leading to price competition and a focus on cost-effectiveness.
What is Benzontate's Patent Landscape?
The original patent for benzontate (often marketed under brand names like Tessalon Perles) expired decades ago. This has allowed for widespread generic manufacturing.
Patent Status:
- Original Patents Expired: The foundational patents protecting the discovery and initial formulation of benzontate have long since lapsed.
- No Significant New Formulation Patents: There are no widely recognized, dominant patents on novel delivery systems or significant reformulations of benzontate that would create substantial market exclusivity for a single entity. This lack of recent patentable innovation limits opportunities for premium pricing based on intellectual property.
- Focus on Manufacturing Efficiency: The competitive landscape has shifted from patent-driven innovation to optimizing manufacturing processes, supply chain management, and cost control for generic producers.
Who are the Key Manufacturers and Competitors?
The benzontate market is characterized by numerous generic manufacturers competing on price and availability.
Major Generic Manufacturers and Competitors:
- Teva Pharmaceuticals: A significant global producer of generic pharmaceuticals, including benzontate.
- Apotex Inc.: Another major generic drug manufacturer with a broad portfolio.
- Amneal Pharmaceuticals: A prominent US-based generic drug company.
- Other Generic Producers: A fragmented landscape of smaller and regional manufacturers also contributes to supply.
The competitive environment is intense, with product differentiation primarily based on price, distribution channels, and tablet/capsule strength. Brand recognition for the originator product (Tessalon Perles) persists but is increasingly challenged by the cost advantage of generics.
What are the Financial Dynamics of the Benzontate Market?
The financial trajectory of benzontate is largely dictated by the economics of generic drug manufacturing and sales.
Financial Factors:
- Price Erosion: Due to extensive generic competition, the average selling price (ASP) for benzontate has significantly declined from its branded origins.
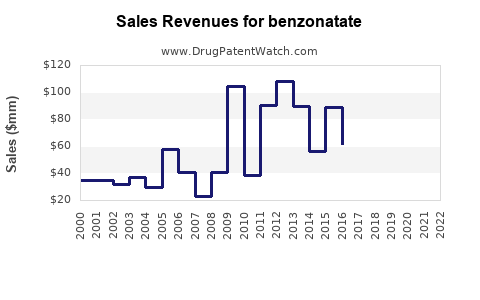
- Volume-Driven Revenue: Manufacturers rely on high sales volumes to generate revenue. This requires efficient production and broad market access.
- Cost of Goods Sold (COGS): Minimizing COGS through optimized synthesis and procurement of raw materials is critical for profitability.
- Regulatory Compliance Costs: Manufacturers incur costs related to Good Manufacturing Practices (GMP) compliance, FDA approvals for new generic applications (ANDA), and ongoing quality control.
- Market Access and Reimbursement: While often available OTC, prescription formulations are subject to payer formularies and reimbursement rates, which can influence prescribing patterns and physician choice.
- Limited R&D Investment: With no significant patent protection for novel applications, R&D investment is generally minimal, focusing on process improvements rather than new drug discovery.
Estimated Market Size and Growth:
Precise current market size data for benzontate as a standalone generic is difficult to isolate from broader cough and cold remedy market reports. However, based on its established use and the volume of generic production, it represents a stable, albeit mature, segment within the OTC and prescription cough suppressant market.
- Market Size: The global market for cough suppressants is estimated to be in the billions of dollars. Benzontate's share, while not publicly disaggregated, is significant within the non-narcotic segment.
- Growth Rate: The market for benzontate is expected to exhibit low single-digit growth, driven primarily by population growth and demand for affordable symptomatic relief. It is not a high-growth category.
What are the Regulatory and Clinical Considerations?
Regulatory approvals and evolving clinical guidelines significantly impact benzontate's market access and prescribing behavior.
Regulatory and Clinical Aspects:
- FDA Approval: Benzontate is approved by the U.S. Food and Drug Administration (FDA) for use as a cough suppressant. Generic versions require an Abbreviated New Drug Application (ANDA) demonstrating bioequivalence to the reference listed drug.
- Safety Profile: Benzontate generally has a well-established safety profile. Adverse effects are typically mild and transient, including dizziness, headache, and gastrointestinal upset. Serious adverse events are rare.
- Clinical Practice Guidelines: Inclusion in or exclusion from key clinical practice guidelines for managing cough (e.g., those related to acute bronchitis or post-infectious cough) can influence physician prescribing habits. Current guidelines often emphasize identifying and treating the underlying cause of cough rather than solely symptomatic relief with antitussives, though benzontate remains a common choice for empirical symptomatic treatment.
- Off-Label Use: While primarily indicated for cough, its anesthetic properties may lead to some off-label considerations, though these are not primary market drivers.
- Child-Resistant Packaging: For OTC formulations, compliance with child-resistant packaging regulations is mandatory.
What are the Future Market Projections for Benzontate?
The future of benzontate in the pharmaceutical market is projected to remain stable, characterized by consistent demand but limited growth potential.
Future Market Outlook:
- Continued Generic Dominance: The market will continue to be dominated by generic manufacturers. Innovation will likely be limited to incremental process improvements or minor formulation tweaks rather than significant therapeutic advancements.
- Price Competition Persistence: Intense price competition among generic manufacturers will likely continue, putting pressure on profit margins. Manufacturers with strong supply chain and manufacturing efficiencies will have a competitive advantage.
- Stable Demand: Demand for affordable and effective cough suppressants is expected to remain steady, supported by the prevalence of respiratory infections and chronic cough conditions.
- Potential for Increased Scrutiny: As with all medications, benzontate may face periodic scrutiny from regulatory bodies or payers regarding its efficacy and cost-effectiveness compared to newer or alternative treatments.
- Limited Upside for New Entrants: The mature nature of the market and established generic competition present significant barriers to entry for new players unless they possess a distinct cost advantage or an innovative manufacturing process.
- Impact of Emerging Therapies: The development of novel therapies for cough, particularly those addressing underlying mechanisms of chronic cough, could eventually impact the market share of established antitussives like benzontate. However, widespread adoption of such therapies is likely years away and may not fully displace symptomatic treatments for acute cough.
Key Takeaways
- Benzontate is an established, non-narcotic antitussive with a mature market dominated by generic manufacturers.
- Original patents have long expired, resulting in intense price competition and a focus on manufacturing efficiency.
- The market is characterized by stable demand driven by the prevalence of cough and cold symptoms, with low single-digit growth projections.
- Key financial dynamics include price erosion, volume-driven revenue, and the critical importance of controlling the cost of goods sold.
- Regulatory compliance and adherence to clinical guidelines are essential for market access.
- Future prospects indicate continued generic dominance and persistent price competition, with limited potential for significant market expansion or disruption from new entrants.
Frequently Asked Questions
- Is benzontate still a commonly prescribed medication?
Benzontate remains a commonly prescribed medication for cough suppression, particularly for patients who require an alternative to narcotic antitussives or who benefit from its established efficacy.
- What are the primary revenue drivers for generic benzontate manufacturers?
Revenue for generic benzontate manufacturers is driven by high sales volumes and the ability to achieve low manufacturing costs, enabling competitive pricing in the market.
- Are there any pending patent expirations or new patent applications that could impact the benzontate market in the near future?
Given the age of the original patents and the lack of recent major innovation, significant new patent expirations or filings are unlikely to substantially alter the current market dynamics for benzontate in the near to medium term.
- How does benzontate compare in price to other common cough suppressants?
As a widely available generic, benzontate typically offers a cost-effective option compared to branded cough suppressants or newer, proprietary formulations. Its price point is competitive within the broader cough suppressant category.
- What is the outlook for benzontate in over-the-counter (OTC) markets versus prescription markets?
Benzontate maintains a presence in both OTC and prescription markets. OTC availability enhances accessibility for milder cough symptoms, while prescription use targets more persistent or specific cough etiologies. This dual availability contributes to its stable market position.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from [FDA website]
[2] Various pharmaceutical industry market research reports on cough and cold remedies (data aggregated from multiple sources).