Last updated: April 24, 2026
Where does albuterol sell, and what drives demand?
Albuterol (albuterol sulfate and related salts) is a core, off-patent short-acting beta2-agonist (SABA) used for rapid relief of bronchospasm in asthma and COPD. Demand is driven by (1) high underlying prevalence of obstructive lung disease, (2) clinical need for rescue therapy, and (3) the long-tail of chronic management where patients keep SABA as an on-hand rescue option.
Primary end-use channels
- Retail pharmacy: prescription SABA therapy remains a large volume driver in US-style markets; dispensing volumes typically favor multiple dosage forms where reimbursed.
- Hospital and urgent care: acute exacerbation care uses nebulized and inhaled albuterol, with procurement tied to formulary cycles and supply continuity.
- Institutional/contract purchasing: long-term care and emergency systems shift between SKUs based on price, device fit, and supply stability.
Demand drivers by segment
- Asthma rescue use: episodic use patterns with ongoing purchase repeat.
- COPD exacerbations: SABA use continues in many regimens, especially where access to long-acting bronchodilators or inhaled steroid combinations is limited.
- Device preference and adherence: pressurized metered-dose inhalers (MDIs), breath-actuated inhalers, and nebulizer solutions each have different utilization patterns tied to patient age and technique.
How is competition structured, and what does that do to pricing?
Albuterol markets are highly competitive and largely generic. Brand-led value is limited by patent and exclusivity expiry across multiple formulations and delivery systems, which compresses pricing and concentrates revenue in whichever companies hold competitive manufacturing scale, device economics, and distribution leverage.
Competitive structure
- Generic albuterol inhalers: multiple manufacturers compete on price and supply reliability.
- Nebulized solutions: competitive tendering and pharmacy sourcing lead to thinner margins versus patent-protected speciality categories.
- Formulation and device differentiation: while the molecule is generic, device compatibility, aerosol performance, packaging, and patient usability drive substitution.
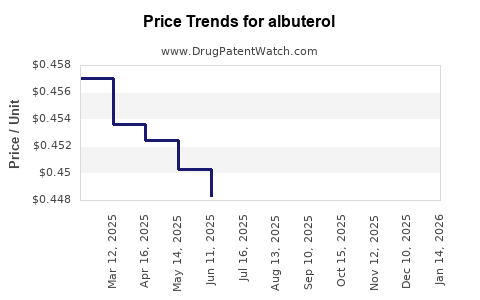
Pricing and margin implications
- Price compression is structurally high because the active ingredient is mature and widely manufactured.
- Revenue growth, where present, is usually volume-led rather than price-led.
- Gross margin variability comes from manufacturing scale, supply chain efficiency, and contract terms with large pharmacy benefit managers and institutions.
What is the financial trajectory profile for albuterol?
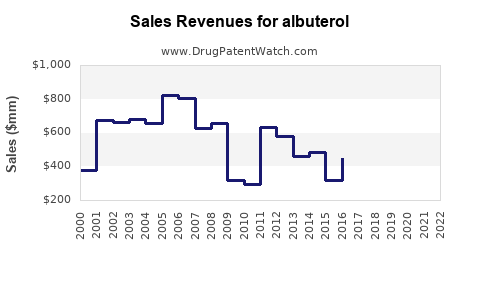
For an off-patent mature respiratory molecule like albuterol, financial trajectories typically follow a pattern: steady baseline revenues, limited upside from market expansion, and periodic disruptions from formulation switches, supply shocks, reimbursement changes, and competitive entry.
Baseline trajectory characteristics
- Volume stability: long-term clinical need keeps consumption from collapsing even as pricing falls.
- Value dilution over time: as additional generics enter and payer pressure intensifies, unit revenue declines.
- Spike risk: demand spikes occur around respiratory seasonality and during periods of supply disruption, then normalize after constraints ease.
- Asset concentration risk: companies with scale and procurement leverage tend to win market share when competitors face manufacturing or quality issues.
Which albuterol products and events shape the numbers most?
The main sources of identifiable financial inflection points are not the molecule’s clinical value but the commercial mechanics: who controls supply, which devices get preferred formulary status, and which presentations become the default payer-covered options.
Key market-shaping categories
- MDI albuterol: high substitution between generics; device and formulation equivalency govern switching.
- Nebulizer solutions: often governed by institutional ordering and ongoing inventory management.
- Pediatric and special populations: dosing formats and ease of administration affect volume distribution.
Typical inflection triggers
- Generic entry waves: new approvals shift competitive intensity and reduce pricing.
- Regulatory and quality supply events: manufacturing interruptions can temporarily increase demand for available SKUs, then reverse.
- Payer reimbursement updates: formulary exclusions and preferred coverage alter share.
How does seasonality affect revenue and cash flow?
Albuterol usage tracks respiratory seasonality. The commercial consequence is uneven quarterly revenue and inventory planning.
Seasonality pattern (typical)
- Higher demand in fall and winter due to respiratory viral burden and exacerbations.
- Lower demand in spring and summer, with baseline chronic rescue use persisting.
Financial knock-on effects
- Working capital swings: inventory built ahead of peak season and reduced after.
- Forecasting and procurement: institutions and wholesalers adjust ordering pace to avoid stockouts while minimizing holding costs.
What do investors and operators watch in albuterol?
For albuterol, the actionable scoreboard is not pipeline value; it is execution across supply, cost, and contracts.
Metrics that predict financial outcomes
- Market share by presentation (MDis vs nebulizer solutions).
- Net price trends (after rebates and chargebacks where applicable).
- Gross margin stability (manufacturing yields, input costs, and contract mix).
- Supply continuity (capacity utilization and regulatory track record).
- Payer coverage changes (preferred products by plan and institution).
What is the likely medium-term trajectory for revenue and profitability?
Given the maturity and generic status, the medium-term outlook is typically:
- Revenue growth limited to modest volume gains, population changes, and substitution dynamics between presentations.
- Profitability capped by price pressure and competition.
- Winners concentrate among manufacturers with scale, strong supply reliability, and cost leadership.
How do broader respiratory dynamics interact with albuterol?
Respiratory care has been shifting toward combination therapy (e.g., inhaled corticosteroid/long-acting bronchodilator regimens) for control rather than rescue. That shift can gradually reduce SABA reliance in controlled asthma, but it does not eliminate SABA use because exacerbations still require rapid bronchodilation.
Interaction effects
- Asthma control trend: lower rescue frequency per patient for some cohorts, offset by overall prevalence and switching behavior.
- COPD management evolution: long-acting bronchodilators reduce symptoms but do not remove SABA as rescue in many regimens.
- Exacerbation patterns: weather, infection prevalence, and healthcare access impact rescue consumption more than regimen theory.
What scenarios most plausibly change the financial path?
Three scenario types dominate albuterol’s financial variance in mature markets.
1) Supply-led market share shifts
- Manufacturing disruptions raise short-term revenue for available SKUs and wholesalers.
- Normalization returns shares but may lock in payer familiarity with the available supplier.
2) Payer and formulary tightening
- Preferred coverage can move market share quickly between equivalent generics.
- Rebates and contract terms can compress net price even when list price holds.
3) Device substitution cycles
- Changes in patient preference and clinician practice influence MDI versus nebulized solution demand.
Key Takeaways
- Albuterol is a mature, largely generic SABA with demand anchored by persistent clinical need for rescue bronchodilation in asthma and COPD.
- Market dynamics are dominated by generic competition, device and presentation substitution, seasonality, and payer/institution procurement rules.
- The financial trajectory is typically stable-to-slow revenue growth with structurally pressured pricing and profitability, with inflection points most often triggered by supply continuity, formulary shifts, and seasonal inventory cycles.
FAQs
1) Is albuterol’s market growth driven by price or volume?
Primarily volume and share shifts across presentations, with sustained price compression.
2) What product category has the biggest impact on albuterol’s commercial outcomes?
MDIs and nebulized solutions compete for share based on payer preference and patient/device fit.
3) How does respiratory seasonality affect financials?
It creates demand peaks in fall and winter, driving inventory build and working-capital swings.
4) What most often creates short-term revenue spikes for albuterol?
Supply interruptions, availability constraints, and temporary switching to available SKUs.
5) Does long-acting therapy eliminate albuterol usage?
No. Control regimens reduce average rescue frequency for some patients, but exacerbations and rescue needs keep SABA demand in place.
References
[1] U.S. Food and Drug Administration (FDA). Albuterol sulfate product information and regulatory resources. (FDA website).
[2] GINA (Global Initiative for Asthma). Global Strategy for Asthma Management and Prevention. (GINA report).
[3] GOLD (Global Initiative for Chronic Obstructive Lung Disease). 2024 Pocket Guide to COPD Diagnosis, Management, and Prevention. (GOLD report).
[4] National Institutes of Health and other clinical guideline bodies. Asthma and COPD rescue therapy guidance (SABA role).