Cefprozil - Generic Drug Details
✉ Email this page to a colleague
What are the generic sources for cefprozil and what is the scope of patent protection?
Cefprozil
is the generic ingredient in two branded drugs marketed by Apotex Inc, Aurobindo Pharma, Chartwell Rx, Lupin, Orchid Hlthcare, Ranbaxy Labs Ltd, Sandoz, Corden Pharma, Teva, and Wockhardt, and is included in seventeen NDAs. Additional information is available in the individual branded drug profile pages.There are thirteen drug master file entries for cefprozil. Six suppliers are listed for this compound.
Summary for cefprozil
| US Patents: | 0 |
| Tradenames: | 2 |
| Applicants: | 10 |
| NDAs: | 17 |
| Drug Master File Entries: | 13 |
| Finished Product Suppliers / Packagers: | 6 |
| Raw Ingredient (Bulk) Api Vendors: | 55 |
| Clinical Trials: | 14 |
| Patent Applications: | 6,539 |
| Formulation / Manufacturing: | see details |
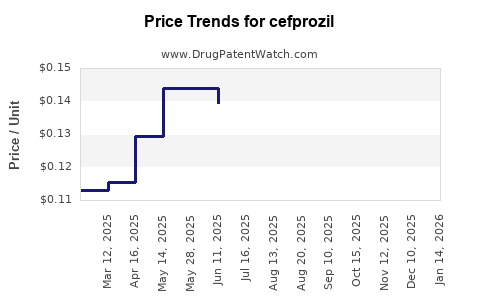
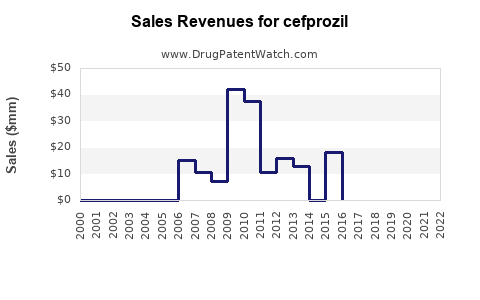
| Drug Prices: | Drug price trends for cefprozil |
| What excipients (inactive ingredients) are in cefprozil? | cefprozil excipients list |
| DailyMed Link: | cefprozil at DailyMed |
Recent Clinical Trials for cefprozil
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| The Affiliated Hospital of Qingdao University | Phase 1 |
| University of Louisville | Early Phase 1 |
| James Graham Brown Cancer Center | Early Phase 1 |
US Patents and Regulatory Information for cefprozil
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ranbaxy Labs Ltd | CEFPROZIL | cefprozil | FOR SUSPENSION;ORAL | 065202-001 | Jun 30, 2006 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Teva | CEFPROZIL | cefprozil | TABLET;ORAL | 065208-002 | Dec 6, 2005 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Lupin | CEFPROZIL | cefprozil | TABLET;ORAL | 065276-001 | Dec 8, 2005 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |