Carbidopa - Generic Drug Details
✉ Email this page to a colleague
What are the generic drug sources for carbidopa and what is the scope of freedom to operate?
Carbidopa
is the generic ingredient in seventeen branded drugs marketed by Alvogen, Ani Pharms, Aurobindo Pharma, Beximco Pharms Usa, Edenbridge Pharms, Novel Labs Inc, Zydus Pharms, Aton, Mylan, Rising, Sun Pharm, Wockhardt Ltd, Orion Pharma, Impax Labs Inc, Abbvie, Accord Hlthcare, Alembic, Apotex, Impax Labs, Kv Pharm, Sciegen Pharms Inc, Sun Pharm Inds, Organon Llc, Ranbaxy, Ucb Inc, Actavis Elizabeth, Apotex Inc, Aurobindo Pharma Ltd, Dr Reddys Labs Sa, Rubicon, SCS, Watson Labs, Avion Pharms, and Organon, and is included in forty-eight NDAs. There are eleven patents protecting this compound. Additional information is available in the individual branded drug profile pages.There are eighteen drug master file entries for carbidopa. Ten suppliers are listed for this compound. There is one tentative approval for this compound.
Summary for carbidopa
| US Patents: | 11 |
| Tradenames: | 17 |
| Applicants: | 34 |
| NDAs: | 48 |
| Drug Master File Entries: | 18 |
| Finished Product Suppliers / Packagers: | 10 |
| Raw Ingredient (Bulk) Api Vendors: | 78 |
| Clinical Trials: | 207 |
| Patent Applications: | 5,830 |
| Formulation / Manufacturing: | see details |
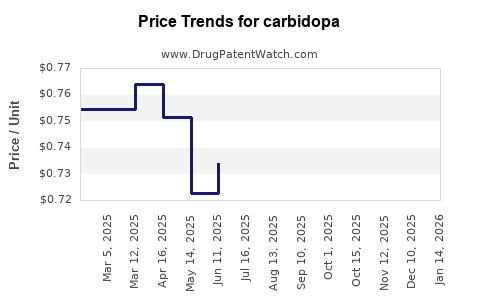
| Drug Prices: | Drug price trends for carbidopa |
| What excipients (inactive ingredients) are in carbidopa? | carbidopa excipients list |
| DailyMed Link: | carbidopa at DailyMed |
Recent Clinical Trials for carbidopa
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Bronx VA Medical Center | Phase 1 |
| University of Minnesota | Early Phase 1 |
| VistaGen Therapeutics, Inc. | Phase 2 |
Generic filers with tentative approvals for CARBIDOPA
| Applicant | Application No. | Strength | Dosage Form |
| ⤷ Try a Trial | ⤷ Try a Trial | 61.25MG;245MG | CAPSULE, EXTENDED RELEASE;ORAL |
| ⤷ Try a Trial | ⤷ Try a Trial | 48.75MG;195MG | CAPSULE, EXTENDED RELEASE;ORAL |
| ⤷ Try a Trial | ⤷ Try a Trial | 36.25MG;145MG | CAPSULE, EXTENDED RELEASE;ORAL |
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.
Medical Subject Heading (MeSH) Categories for carbidopa
US Patents and Regulatory Information for carbidopa
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sun Pharm | CARBIDOPA, LEVODOPA AND ENTACAPONE | carbidopa; entacapone; levodopa | TABLET;ORAL | 079085-002 | May 10, 2012 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Rising | CARBIDOPA, LEVODOPA AND ENTACAPONE | carbidopa; entacapone; levodopa | TABLET;ORAL | 213212-004 | Jan 25, 2022 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Organon | SINEMET | carbidopa; levodopa | TABLET;ORAL | 017555-002 | Approved Prior to Jan 1, 1982 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Mylan | CARBIDOPA, LEVODOPA AND ENTACAPONE | carbidopa; entacapone; levodopa | TABLET;ORAL | 203424-005 | Aug 13, 2020 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Mylan | CARBIDOPA AND LEVODOPA | carbidopa; levodopa | TABLET, EXTENDED RELEASE;ORAL | 075091-002 | Apr 21, 2000 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Impax Labs | CARBIDOPA AND LEVODOPA | carbidopa; levodopa | TABLET, ORALLY DISINTEGRATING;ORAL | 090631-001 | Jun 8, 2010 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for carbidopa
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Aton | LODOSYN | carbidopa | TABLET;ORAL | 017830-001 | Approved Prior to Jan 1, 1982 | ⤷ Try a Trial | ⤷ Try a Trial |
| Aton | LODOSYN | carbidopa | TABLET;ORAL | 017830-001 | Approved Prior to Jan 1, 1982 | ⤷ Try a Trial | ⤷ Try a Trial |
| Aton | LODOSYN | carbidopa | TABLET;ORAL | 017830-001 | Approved Prior to Jan 1, 1982 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |