BRIMONIDINE TARTRATE - Generic Drug Details
✉ Email this page to a colleague
What are the generic drug sources for brimonidine tartrate and what is the scope of patent protection?
Brimonidine tartrate
is the generic ingredient in nine branded drugs marketed by Padagis Israel, Galderma Labs Lp, Allergan, Abbvie, Alembic, Apotex, Bausch And Lomb, Dr Reddys Labs Sa, Indoco, Micro Labs, Rising, Sandoz, Somerset Theraps Llc, Teva Parenteral, Bausch And Lomb Inc, Alcon Labs Inc, Florida, Sentiss, and Upsher Smith Labs, and is included in thirty NDAs. There are twenty patents protecting this compound and three Paragraph IV challenges. Additional information is available in the individual branded drug profile pages.Brimonidine tartrate has one hundred and ninety-nine patent family members in thirty-two countries.
There are eleven drug master file entries for brimonidine tartrate. Fifteen suppliers are listed for this compound. There is one tentative approval for this compound.
Summary for BRIMONIDINE TARTRATE
| International Patents: | 199 |
| US Patents: | 20 |
| Tradenames: | 9 |
| Applicants: | 19 |
| NDAs: | 30 |
| Drug Master File Entries: | 11 |
| Finished Product Suppliers / Packagers: | 15 |
| Raw Ingredient (Bulk) Api Vendors: | 153 |
| Clinical Trials: | 54 |
| Patent Applications: | 2,418 |
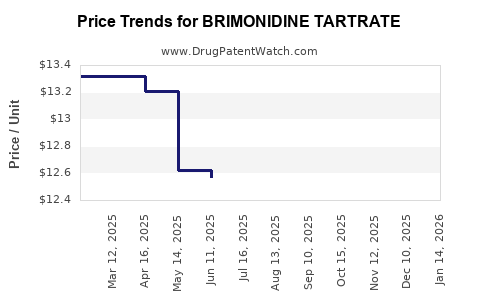
| Drug Prices: | Drug price trends for BRIMONIDINE TARTRATE |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for BRIMONIDINE TARTRATE |
| What excipients (inactive ingredients) are in BRIMONIDINE TARTRATE? | BRIMONIDINE TARTRATE excipients list |
| DailyMed Link: | BRIMONIDINE TARTRATE at DailyMed |
Recent Clinical Trials for BRIMONIDINE TARTRATE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Tulane University | Phase 4 |
| Visus Therapeutics | Phase 3 |
| Optall Vision | Phase 1 |
Generic filers with tentative approvals for BRIMONIDINE TARTRATE
| Applicant | Application No. | Strength | Dosage Form |
| ⤷ Try a Trial | ⤷ Try a Trial | 0.2%; 0.5% | SOLUTION; OPHTHALMIC |
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.
Pharmacology for BRIMONIDINE TARTRATE
| Drug Class | alpha-Adrenergic Agonist |
| Mechanism of Action | Adrenergic alpha-Agonists |
Medical Subject Heading (MeSH) Categories for BRIMONIDINE TARTRATE
Paragraph IV (Patent) Challenges for BRIMONIDINE TARTRATE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| LUMIFY | Ophthalmic Solution | brimonidine tartrate | 0.025% | 208144 | 1 | 2021-07-12 |
| MIRVASO | Topical Gel | brimonidine tartrate | 0.33% | 204708 | 1 | 2014-12-15 |
| ALPHAGAN P | Ophthalmic Solution | brimonidine tartrate | 0.1% | 021770 | 1 | 2006-12-20 |
| ALPHAGAN P | Ophthalmic Solution | brimonidine tartrate | 0.15% | 021262 | 1 | 2006-11-03 |
US Patents and Regulatory Information for BRIMONIDINE TARTRATE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Galderma Labs Lp | MIRVASO | brimonidine tartrate | GEL;TOPICAL | 204708-001 | Aug 23, 2013 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Alembic | BRIMONIDINE TARTRATE AND TIMOLOL MALEATE | brimonidine tartrate; timolol maleate | SOLUTION/DROPS;OPHTHALMIC | 215230-001 | Aug 25, 2023 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Florida | BRIMONIDINE TARTRATE AND TIMOLOL MALEATE | brimonidine tartrate; timolol maleate | SOLUTION/DROPS;OPHTHALMIC | 201949-001 | Oct 4, 2022 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Micro Labs | BRIMONIDINE TARTRATE | brimonidine tartrate | SOLUTION/DROPS;OPHTHALMIC | 217846-001 | Nov 22, 2023 | AT | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Bausch And Lomb Inc | LUMIFY | brimonidine tartrate | SOLUTION/DROPS;OPHTHALMIC | 208144-001 | Dec 22, 2017 | OTC | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for BRIMONIDINE TARTRATE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Allergan | ALPHAGAN | brimonidine tartrate | SOLUTION/DROPS;OPHTHALMIC | 020613-001 | Sep 6, 1996 | ⤷ Try a Trial | ⤷ Try a Trial |
| Abbvie | ALPHAGAN P | brimonidine tartrate | SOLUTION/DROPS;OPHTHALMIC | 021770-001 | Aug 19, 2005 | ⤷ Try a Trial | ⤷ Try a Trial |
| Abbvie | ALPHAGAN P | brimonidine tartrate | SOLUTION/DROPS;OPHTHALMIC | 021262-001 | Mar 16, 2001 | ⤷ Try a Trial | ⤷ Try a Trial |
| Abbvie | ALPHAGAN P | brimonidine tartrate | SOLUTION/DROPS;OPHTHALMIC | 021770-001 | Aug 19, 2005 | ⤷ Try a Trial | ⤷ Try a Trial |
| Abbvie | ALPHAGAN P | brimonidine tartrate | SOLUTION/DROPS;OPHTHALMIC | 021262-001 | Mar 16, 2001 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for BRIMONIDINE TARTRATE
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Canada | 2567401 | COMPOSES, PREPARATIONS ET METHODES PERMETTANT DE TRAITER OU DE PREVENIR DES TROUBLES INFLAMMATOIRES CUTANES (COMPOUNDS, FORMULATIONS, AND METHODS FOR TREATING OR PREVENTING INFLAMMATORY SKIN DISORDERS) | ⤷ Try a Trial |
| China | 103298451 | Brimonidine gel compositions and methods of use | ⤷ Try a Trial |
| Singapore | 189896 | BRIMONIDINE GEL COMPOSITIONS AND METHODS OF USE | ⤷ Try a Trial |
| Mexico | 2007002383 | COMPOSICIONES QUE CONTIENEN COMPONENTES AGONISTAS ALFA-2-ADRENERGICOS. (COMPOSITIONS CONTAINING ALPHA-2-ADRENERGIC AGONIST COMPONENTS.) | ⤷ Try a Trial |
| South Korea | 101257046 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for BRIMONIDINE TARTRATE
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1631293 | 300683 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: BRIMONIDINE EN FARMACEUTISCH AANVAARDBARE ZOUTEN DAARVAN VOOR TOEPASSING ALS GENEESMIDDEL VOOR DE BEHANDELING VAN DOOR ROSACEA GEINDUCEERDE ROODHEID; REGISTRATION NO/DATE: EU/1/13/904 20140225 |
| 1631293 | CR 2014 00031 | Denmark | ⤷ Try a Trial | PRODUCT NAME: BRIMONIDIN OG ET FARMACEUTISK ACCEPTABELT SALT DERAF, HERUNDER BRIMONIDIN TARTRAT; REG. NO/DATE: EU/1/13/904/001-003 20140221 |
| 1631293 | 2014/041 | Ireland | ⤷ Try a Trial | PRODUCT NAME: BRIMONIDINE AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF; REGISTRATION NO/DATE: EU/1/13/904 20140221 |
| 1631293 | 14C0056 | France | ⤷ Try a Trial | PRODUCT NAME: BRIMONIDINE OU L'UN DE SES SELS PHARMACEUTIQUEMENT ACCEPTABLES; REGISTRATION NO/DATE: EU/1/13/904 20140225 |
| 1631293 | C01631293/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: BRIMONIDIN; REGISTRATION NO/DATE: SWISSMEDIC 65180 21.11.2014 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.