Last updated: February 13, 2026
What Is the Current Market for Losartan Potassium?
Losartan potassium is an angiotensin II receptor blocker (ARB) approved for treating hypertension and reducing stroke risk. It has also been prescribed for diabetic nephropathy and heart failure. Globally, the drug generates approximately $2.3 billion annually as of 2022, with the U.S. accounting for roughly 55% of sales [1]. The drug’s market size is influenced by its widespread use in chronic disease management, ongoing patent protection, and generic availability.
How Has the Market for Losartan Evolved?
Market growth was steady from 2010 to 2018, driven by increased hypertension prevalence and prescribing rates. Generic entries in 2011 significantly lowered prices, leading to market expansion but compressed profit margins for branded producers. The introduction of combination therapies incorporating losartan now also influences prescribing patterns, with fixed-dose combinations becoming more common.
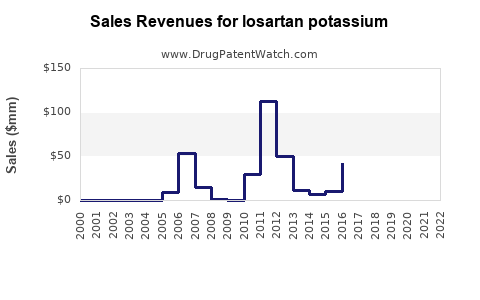
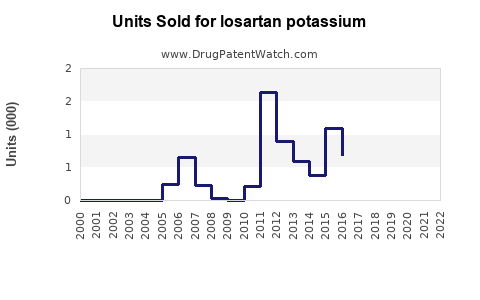
The patent expiry in many jurisdictions, including the U.S. in 2011, opened the market to generics, causing a sharp decline in brand-name sales but increasing overall volume. Despite this, branded losartan sales persisted in some regions due to patent protections and market segmentation strategies.
What Are the Key Drivers of Growth and Decline?
Drivers of Growth
- Aging population globally increases the prevalence of hypertension.
- Increased awareness and screening for cardiovascular risks.
- Expansion into new therapeutic areas, including diabetic nephropathy.
Drivers of Decline
- Patent expirations and subsequent rise of generics.
- Heightened competition from other ARBs, such as valsartan and telmisartan.
- Pricing pressures and increasing focus on cost-effective treatments.
What Are Projected Sales Figures and Market Trends?
2023–2028 Revenue Projections
- Expected compound annual growth rate (CAGR): approximately 3.2% [2].
- Estimated global sales reach $2.8 billion by 2028.
- North American markets will maintain about 50-55% share due to established prescriber bases.
Market Segmentation
- Hypertension management: 70%
- Diabetic nephropathy: 15%
- Heart failure: 10%
- Other indications (e.g., stroke prevention): 5%
Competitive Landscape
- Top manufacturers include Merck & Co., Teva Pharmaceutical Industries, and Sandoz.
- Private label generics dominate in price-sensitive markets, constraining branded sales growth.
- Innovator companies have limited pipeline updates, but combination formulations are emerging.
What Potential Market Risks Could Affect Future Sales?
- Price competition due to widespread generic availability.
- Regulatory pressures demanding new clinical trials or labeling updates.
- Competition from alternative drug classes like ACE inhibitors and newer agents such as sacubitril/valsartan.
- Patent litigation and patent extensions, which could temporarily extend exclusivity in certain markets.
How Does the Competition Impact Losartan’s Future?
Since 2011, multiple ARBs have entered the market, offering similar efficacy with varying side effect profiles. Valsartan and irbesartan are primary competitors in the hypertension segment. The development of combination drugs combining losartan with other antihypertensives enhances adherence but can also fragment the market share.
Efforts to develop improved formulations, including sustained-release and fixed-dose combinations, aim to sustain revenue streams. Patent filings for such formulations suggest strategic moves to extend product lifecycle.
What Are the Implications for Stakeholders?
- Pharmaceutical companies must innovate to maintain market share.
- Healthcare providers will continue to balance cost and clinical efficacy.
- Payers seek more cost-effective alternatives due to pricing pressures.
Key Takeaways
- Losartan potassium remains a significant player in hypertension treatment, with steady global sales projected at approximately $2.8 billion by 2028.
- Patent expiry and generic competition have compressed profitability but expanded volume.
- Increased adoption of combination therapies and expanded indications can sustain growth.
- Competition from other ARBs and new drug classes presents ongoing challenges.
- Market risks include pricing pressures, regulatory changes, and patent disputes.
FAQs
1. Is the patent for losartan still active?
Most patents expired in key markets by 2011, making generics widely available.
2. What are the main competitors to losartan?
Other ARBs like valsartan, irbesartan, and candesartan dominate the market in hypertensive care.
3. Can losartan be used for indications beyond hypertension?
Yes, it is approved for diabetic nephropathy and heart failure, with emerging off-label uses.
4. How do generics influence losartan’s market share?
Generics significantly lower prices, increasing utilization but reducing profit margins for original manufacturers.
5. Are there ongoing innovations around losartan?
Yes, fixed-dose combinations and sustained-release formulations are in development to extend commercial viability.
References
[1] IQVIA. Global Prescription Market Data, 2022.
[2] Transparency Market Research. Losartan Market Forecast, 2022–2028.