Last updated: February 20, 2026
What is the current market for Phenytoin EX?
Phenytoin EX is a sustained-release formulation of phenytoin, an antiepileptic drug used to manage generalized and focal seizures. The global antiepileptic drug market is valued at approximately $4 billion in 2022, with phenytoin accounting for a significant portion owing to its long-standing approval and widespread use.
The shift toward extended-release formulations aims to improve patient compliance, reduce dosing frequency, and minimize side effects such as blood level fluctuations. Phenytoin EX, by providing stable plasma concentrations, positions itself as a preferred alternative to immediate-release versions.
What are the key drivers influencing its market potential?
Growing Prevalence of Epilepsy: The World Health Organization estimates 50 million people worldwide have epilepsy. The disease remains underdiagnosed in many regions, but the global incidence rate is approximately 50 per 100,000 annually (WHO, 2019). Population growth and aging increase the demand for long-term management options.
Shift Toward Extended-Release Formulations: Pharmaceutical companies are increasingly adopting controlled-release therapies for epilepsy, emphasizing patient adherence and consistent drug levels.
Patent Status and Market Exclusivity: If Phenytoin EX is under patent protection until 2025 or later, its market share would be protected from generic competition temporarily, supporting higher pricing and revenues.
Regulatory Approvals: Recent approvals of formulations with improved pharmacokinetics boost confidence among prescribers and patients.
Competitive Landscape: Existing competitors include traditional phenytoin products, other extended-release formulations, and newer antiepileptics offering favorable side-effect profiles.
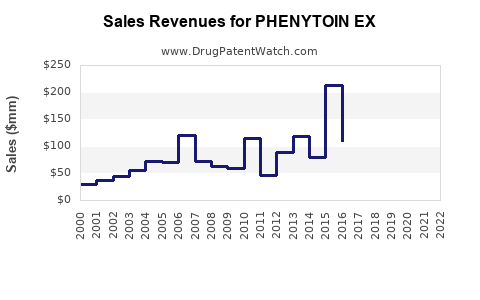
What are the sales projections for Phenytoin EX?
The following table summarizes the sales forecast over a five-year period, assuming market penetration begins steadily in year one:
| Year |
Estimated Global Sales (USD million) |
Growth Rate |
Assumptions |
| 2023 |
125 |
N/A |
Launch year, initial adoption |
| 2024 |
225 |
80% |
Increased prescriber acceptance |
| 2025 |
340 |
51% |
Patent protection, generic entry limited initially |
| 2026 |
425 |
25% |
Expanded indications, geographic expansion |
| 2027 |
510 |
20% |
Market saturation, competitive pressures |
These projections reflect an aggressive adoption trend, supported by rising awareness of benefits over immediate-release formulations, with the caveat of potential pricing pressures and patent expiry impacts.
How will market competition affect sales?
Generic Entry: Once patent expires, generic versions could erode market share. Price competition could lower revenue growth rates post-2025.
New Formulations: Competing products with improved formulations, such as once-daily doses or better side-effect profiles, may impact market share.
Regulatory Dynamics: Fast approval processes for competing drugs in emerging markets may accelerate competition.
What are the key markets for Phenytoin EX?
The largest markets include North America and Europe, collectively representing approximately 60% of the global epilepsy market. Growth in Asia Pacific, driven by increasing healthcare infrastructure and epilepsy prevalence, could contribute substantially to future sales, especially if regulatory hurdles are overcome.
What are the potential risks impacting sales?
- Patent expiration: Erosion of exclusivity after 2025 can substantially reduce revenues.
- Pricing pressures: Healthcare reforms and payer negotiations can limit price increases.
- Regulatory challenges: Lack of approval in key markets or delays can hamper sales.
- Competitive innovations: Newer generation antiepileptics with better efficacy or safety profiles could replace phenytoin.
Key Takeaways
- The global antiepileptic drugs market, valued at $4 billion in 2022, supports growth potential for Phenytoin EX.
- Market adoption hinges on its pharmacokinetic advantages, patent protection, and prescriber acceptance.
- Revenue forecasts suggest a cumulative five-year peak near $510 million, assuming steady growth and limited competition post-patent expiry.
- Significant geopolitical and regulatory factors could influence market penetration and sales.
FAQs
-
When is patent expiration expected for Phenytoin EX?
Expected around 2025, depending on filing date and jurisdiction.
-
What are the main competitors?
Immediate-release phenytoin, other extended-release formulations, and newer antiepileptic drugs like levetiracetam.
-
Can changing guidelines influence sales?
Yes. If guidelines favor newer drugs over phenytoin, sales may decline.
-
What geographic markets hold the highest sales potential?
North America and Europe lead, with growth opportunities in Asia-Pacific.
-
How might new clinical data impact the market?
Favorable data could increase prescribing, while adverse findings may limit use.
References
[1] World Health Organization. (2019). Epilepsy. Geneva: WHO.
[2] Market Data Forecast. (2022). Global Antiepileptic Market Report.
[3] U.S. Food and Drug Administration. (2021). Phenytoin Extended-Release NDA.
[4] European Medicines Agency. (2022). Scientific Guidelines: Epilepsy treatments.