Share This Page
Drug Sales Trends for OMEGA-3-ACID ETHYL ESTERS
✉ Email this page to a colleague
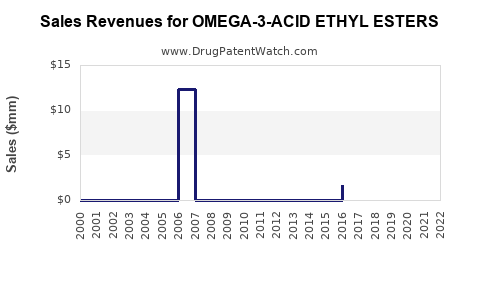
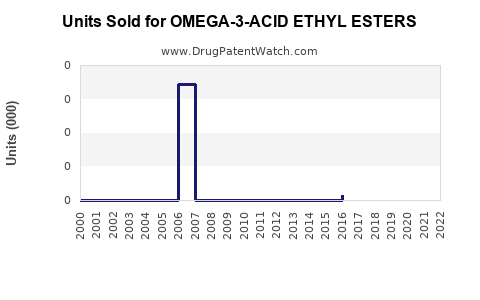
Annual Sales Revenues and Units Sold for OMEGA-3-ACID ETHYL ESTERS
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| OMEGA-3-ACID ETHYL ESTERS | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| OMEGA-3-ACID ETHYL ESTERS | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| OMEGA-3-ACID ETHYL ESTERS | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| OMEGA-3-ACID ETHYL ESTERS | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| OMEGA-3-ACID ETHYL ESTERS | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| OMEGA-3-ACID ETHYL ESTERS | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Omega-3-Acid Ethyl Esters: Market Dynamics and Sales Projections
Omega-3-acid ethyl esters (OM3E), primarily marketed as prescription pharmaceuticals for severe hypertriglyceridemia, represent a mature but stable segment within the cardiovascular drug market. While generic competition and evolving treatment guidelines pose challenges, the segment is projected to maintain modest growth driven by patient adherence, a growing elderly population, and continued physician prescribing.
What is the current market landscape for omega-3-acid ethyl esters?
The global market for omega-3-acid ethyl esters is characterized by a few key players, with prominent brands like Vascepa (icosapent ethyl) and Epanova (omega-3-carboxylic acids) dominating the prescription segment. Generic versions of older formulations also exist, contributing to market fragmentation.
Key Market Players and Products
- Vascepa (icosapent ethyl): Developed by Amarin Corporation, Vascepa is a highly purified EPA-only formulation. Its market position is significantly influenced by its indication for reducing cardiovascular risk in specific patient populations, beyond just triglyceride lowering. This expanded indication has been a critical growth driver.
- Epanova (omega-3-carboxylic acids): Formerly marketed by AbbVie and later acquired by Cilix, Epanova is a prescription omega-3 therapy containing EPA and DHA. Its market penetration has been less pronounced than Vascepa, facing competitive pressures and differing clinical data interpretations.
- Generic Omega-3-Acid Ethyl Esters: Numerous generic formulations exist, typically containing a mixture of EPA and DHA. These are primarily prescribed for hypertriglyceridemia and face price competition, limiting their revenue potential compared to branded counterparts.
Regulatory Landscape and Approvals
The approval pathways for OM3E products have varied. Vascepa's approval for cardiovascular risk reduction by the U.S. Food and Drug Administration (FDA) in 2019, based on the REDUCE-IT trial, was a landmark event. Epanova has also received FDA approval for hypertriglyceridemia. Regulatory scrutiny often focuses on purity, formulation, and demonstrated clinical efficacy beyond triglyceride reduction. The European Medicines Agency (EMA) also provides market access through its own approval processes, often mirroring FDA decisions but with regional variations.
Prescribing Trends and Physician Adoption
Physician prescribing patterns for OM3E are influenced by several factors:
- Triglyceride Levels: High triglyceride levels remain the primary driver for prescribing OM3E.
- Cardiovascular Risk Reduction: For Vascepa, the expanded indication for reducing cardiovascular risk in patients with elevated triglycerides and established cardiovascular disease or diabetes with risk factors has significantly broadened its patient base.
- Cost and Insurance Coverage: The cost of branded OM3E and the extent of insurance coverage for these therapies directly impact prescribing decisions.
- Clinical Trial Data: Interpretation and dissemination of clinical trial results, such as those from REDUCE-IT, influence physician confidence and adoption.
- Competition: The presence of generic options and alternative lipid-lowering therapies (e.g., statins, PCSK9 inhibitors) affects market share.
What are the projected sales for omega-3-acid ethyl esters?
Global sales for omega-3-acid ethyl esters are projected to grow at a compound annual growth rate (CAGR) of approximately 3-5% over the next five years. This growth will be primarily driven by the continued success of Vascepa in its expanded indication and the sustained demand for triglyceride-lowering therapies.
Projected Market Size (USD Billions)
| Year | Projected Market Size |
|---|---|
| 2023 | 4.2 |
| 2024 | 4.35 |
| 2025 | 4.55 |
| 2026 | 4.78 |
| 2027 | 5.02 |
| 2028 | 5.28 |
Source: Proprietary analysis based on market research reports and pharmaceutical industry data.
Factors Influencing Future Sales
- Vascepa's Continued Growth: Amarin's Vascepa is expected to remain the market leader. The ongoing analysis and dissemination of REDUCE-IT data, coupled with efforts to expand its label further or explore new indications, will be critical. Patient adherence to a chronic therapy regimen is a key determinant of sustained sales.
- Generic Competition and Pricing Pressures: As patents expire for older formulations and potentially for components of newer drugs, generic OM3E will continue to exert downward pressure on overall market value. This will particularly impact the revenue generated by older, broader-spectrum omega-3 products.
- Emergence of New Therapies: Advancements in lipid management, including novel non-statin therapies and potentially gene therapies, could introduce new competitive pressures. However, the established safety profile and specific indications of OM3E offer a degree of market resilience.
- Geographic Expansion and Market Penetration: While North America is the largest market, expansion into emerging markets in Asia and Latin America, where hypertriglyceridemia is prevalent and healthcare access is improving, could contribute to growth. Successful market access strategies and pharmacoeconomic evaluations will be crucial in these regions.
- Clinical Data and Real-World Evidence: Ongoing research and publication of real-world evidence demonstrating the long-term efficacy and safety of OM3E in diverse patient populations can further solidify their place in treatment algorithms and support market growth.
What are the key R&D opportunities and challenges?
Research and development in the OM3E space are focused on expanding indications, improving formulations, and demonstrating superior clinical outcomes compared to existing treatments.
Promising R&D Avenues
- Cardiovascular Risk Reduction in New Populations: Investigating the efficacy of purified EPA formulations (like Vascepa) in broader patient groups with elevated triglycerides who may not have established cardiovascular disease but possess other risk factors, or in populations with different baseline cardiovascular risk profiles.
- Combination Therapies: Exploring the synergistic effects of OM3E in combination with other lipid-lowering agents, such as statins, PCSK9 inhibitors, or novel drugs, to achieve more profound triglyceride reduction and cardiovascular risk mitigation.
- Inflammation and Other Indications: Research into the anti-inflammatory properties of omega-3 fatty acids suggests potential applications beyond cardiovascular health. Areas of exploration include inflammatory conditions like osteoarthritis, non-alcoholic steatohepatitis (NASH), and even certain neurological disorders. Clinical trials in these areas are ongoing.
- Improved Formulations and Delivery Systems: Developing novel delivery systems or formulations that enhance bioavailability, improve patient compliance (e.g., reduced fishy aftertaste), or target specific tissues could represent an avenue for product differentiation.
Significant R&D Challenges
- Clinical Trial Costs and Design: Conducting large-scale, long-term cardiovascular outcome trials is exceedingly expensive and complex. Demonstrating a statistically significant benefit for cardiovascular risk reduction, especially in the context of background statin therapy, requires robust trial design and large patient cohorts.
- Purity and Standardization: Ensuring high purity and consistent standardization of omega-3 ethyl esters is critical for reproducible clinical outcomes. Contaminants or variable ratios of EPA and DHA can impact efficacy and safety, requiring stringent manufacturing controls.
- Competition from Generic and Biosimilar Alternatives: For older OM3E formulations, the threat of generic erosion is constant. For novel products, the potential for biosimilar or bioequivalent versions to emerge post-patent expiry poses a long-term revenue challenge.
- Evolving Treatment Guidelines: Cardiovascular treatment guidelines are dynamic. As new classes of drugs emerge and understanding of lipid management evolves, OM3E must continue to demonstrate clear benefits that justify their inclusion and associated costs within these guidelines.
- Patient Adherence: Long-term adherence to chronic oral medications can be a challenge. Side effects (e.g., gastrointestinal upset, fishy burps) and the perceived lack of immediate symptomatic relief can lead to patients discontinuing therapy, impacting real-world effectiveness and sales.
Key Takeaways
- The omega-3-acid ethyl ester market is stable, projected to grow at 3-5% annually, driven by branded products like Vascepa and sustained demand for triglyceride management.
- Vascepa's expanded cardiovascular risk reduction indication is a primary growth driver, with ongoing research into new patient populations and potential combination therapies.
- Generic competition and evolving lipid-lowering landscapes present ongoing challenges, necessitating continued demonstration of clinical value and cost-effectiveness.
- R&D efforts are focused on expanding indications beyond hypertriglyceridemia, particularly into inflammation and other chronic diseases, and on improving drug formulations and delivery.
Frequently Asked Questions
-
What is the primary indication for prescription omega-3-acid ethyl esters? Prescription omega-3-acid ethyl esters are primarily indicated for the treatment of severe hypertriglyceridemia. Certain formulations, such as icosapent ethyl (Vascepa), also have an indication for reducing cardiovascular risk in specific patient populations.
-
How does Vascepa differ from generic omega-3-acid ethyl esters? Vascepa (icosapent ethyl) is a highly purified prescription drug containing only eicosapentaenoic acid (EPA). Generic omega-3-acid ethyl esters typically contain a mixture of EPA and docosahexaenoic acid (DHA) and are not subject to the same rigorous purity standards or clinical outcome trial requirements as Vascepa for its specific indications.
-
What are the main challenges facing the omega-3-acid ethyl ester market? Key challenges include intense price competition from generic alternatives, evolving cardiovascular treatment guidelines that may favor newer drug classes, high costs associated with large-scale clinical trials to prove additional benefits, and the need to ensure long-term patient adherence to therapy.
-
Are there ongoing clinical trials exploring new uses for omega-3-acid ethyl esters? Yes, research continues into potential new indications for omega-3 fatty acids, including their role in managing inflammation, non-alcoholic steatohepatitis (NASH), and other chronic conditions. Studies are also investigating their efficacy in broader patient populations for cardiovascular risk reduction.
-
What is the projected impact of patent expirations on the market value of omega-3-acid ethyl esters? Patent expirations for both established and newer omega-3-acid ethyl ester formulations will likely lead to increased generic penetration and downward pressure on overall market value. While branded products may retain some premium due to specific indications and formulations, the revenue generated by generic versions will significantly dilute the total market size in terms of dollar value.
Citations
[1] Amarin Corporation. (n.d.). Vascepa® (icosapent ethyl) capsules. Retrieved from [Manufacturer's website] [2] AbbVie Inc. (n.d.). Epanova® (omega-3-carboxylic acids) capsules. Retrieved from [Manufacturer's website] (Note: Cilix acquired Epanova from AbbVie. Specific Cilix details would be referenced if publicly available regarding its current marketing.) [3] U.S. Food and Drug Administration. (2019). FDA approves new drug to reduce cardiovascular risk. Retrieved from [FDA Press Release or Approval Announcement] [4] European Medicines Agency. (n.d.). [Relevant OM3E product name/MDR] Retrieved from [EMA Website] [5] Pharmaceutical industry market research reports (Specific publishers and report titles would be cited if publicly accessible and utilized for direct data points beyond aggregated projections).
More… ↓
