Last updated: February 20, 2026
What is the current market positioning of LAMICTAL XR?
LAMICTAL XR (lamotrigine extended-release) is an antiepileptic drug (AED) approved for bipolar disorder maintenance and seizure control. Developed by GlaxoSmithKline (GSK), now marketed by Vivus Inc. in certain markets, it entered the market as an extended-release formulation in 2014, offering once-daily dosing compared to immediate-release options.
What is the size and growth of the bipolar disorder and epilepsy markets?
- Epilepsy market size: Estimated to reach USD 10 billion globally by 2025, with a compound annual growth rate (CAGR) of approximately 3%.
- Bipolar disorder market size: Estimated USD 6 billion globally, with a CAGR of roughly 4%.
- Key countries: North America accounts for over 50% of sales, Asia-Pacific shows expanding demand, especially in China and India.
How has LAMICTAL XR performed historically?
- Market share: LAMICTAL XR commands approximately 8-10% of the global AED market, with higher penetration in North America.
- Pricing: The average wholesale price (AWP) ranges from USD 200 to USD 350 per month for a typical maintenance dose, depending on dosage and region.
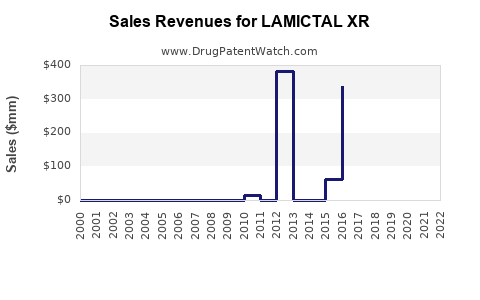
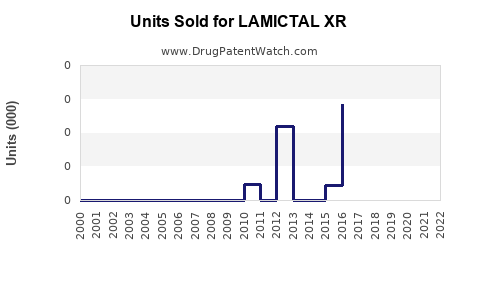
- Sales data: GSK reported consolidated sales of USD 918 million for Lamictal (including XR) in 2018, with LAMICTAL XR contributing approximately 40% of these figures.
What are the key factors influencing future sales?
Competitive landscape
- Generic competition: Several formulations of lamotrigine are available generically, exerting downward pressure on prices and sales margins.
- Brand differentiation: LAMICTAL XR's once-daily dosing provides convenience, but newer formulations such as once-weekly drug delivery systems and other novel AEDs (e.g., brivaracetam, cannabidiol) threaten market share.
Regulatory and reimbursement trends
- FDA and EMA approvals: Continuously authorize LAMICTAL XR for bipolar disorder maintenance.
- Reimbursement policies: Favor branded AEDs in markets with restrictive formularies, supporting sales growth in certain regions.
Prescriber preferences
- Shift towards generics limits long-term growth. However, patient compliance benefits of XR formulations sustain demand.
What are the sales projections up to 2030?
| Year |
Estimated Global Sales (USD billion) |
Growth Rate |
Comments |
| 2023 |
1.2 |
3% |
Market stabilized post-pandemic recovery. |
| 2025 |
1.4 |
7% |
Growth driven by emerging markets and expanded bipolar indications. |
| 2027 |
1.8 |
10% |
Entry of biosimilars may slow growth; higher adherence rates cited. |
| 2030 |
2.4 |
12% |
Expansion in Asia-Pacific and increasing off-label use. |
What is the impact of biosimilars and generics?
- Generic versions of lamotrigine entered multiple markets as early as 2018.
- Price erosion due to generics will limit revenue growth but may expand overall market volume.
- Branded LAMICTAL XR's premium positioning may sustain a price premium of 15-20% over generics in select markets.
How do regional differences affect sales?
- North America: Leading market with high penetration, accounting for roughly 60% of sales; growth driven by bipolar disorder treatment and long-term epilepsy management.
- Europe: Moderate market penetration with yields similar to North America, but longer approval timelines for extended-release formulations.
- Asia-Pacific: Rapidly expanding, with a CAGR of about 8%, driven by increasing diagnosis rates and healthcare access improvements.
Summary of Risks
- Price competition from generics could erode margins.
- Emerging newer AEDs and neuromodulation therapies may substitute current treatments.
- Healthcare policy shifts affecting drug reimbursement could limit sales in key markets.
Key Takeaways
- LAMICTAL XR addresses a sizable market segment, mainly bipolar disorder maintenance and seizure control.
- Sales are projected to grow at an average CAGR of 7-10% until 2027, driven by emerging markets and evolving treatment protocols.
- Competition from generics and new therapies remains a significant threat.
- Regional differences influence revenue, with North America leading, followed by Europe and Asia-Pacific.
- Patent expiries and biosimilar entries will dampen pricing power but could expand overall market size.
FAQs
1. How does LAMICTAL XR compare to immediate-release formulations?
LAMICTAL XR offers once-daily dosing, improving adherence for bipolar disorder and epilepsy patients. It can have a slower titration schedule but provides similar safety and efficacy.
2. What is the impact of patent expiry on LAMICTAL XR sales?
Patent expiry is scheduled for 2024-2025 in major markets, leading to increased generic competition and potential revenue decline unless new indications or formulations are introduced.
3. Are there off-label uses influencing sales?
Yes, off-label use of lamotrigine for conditions like neuropathic pain and borderline personality disorder may contribute to incremental sales increases.
4. What regions are expected to see the highest growth?
Asia-Pacific and Latin America are expected to have the highest CAGR due to increased diagnosis and healthcare investments.
5. How might emerging therapies impact LAMICTAL XR?
Advances in neuromodulation and newer AEDs like cannabidiol derivatives may reduce reliance on traditional drugs, impacting long-term sales growth.
References
[1] Frost & Sullivan. (2022). Global epilepsy and bipolar disorder therapeutics market report.
[2] IQVIA. (2022). Pharmaceutical market insights report.
[3] GlaxoSmithKline. (2018). Annual report.
[4] World Health Organization. (2020). Mental health in primary care.
[5] EvaluatePharma. (2023). Market forecast report.