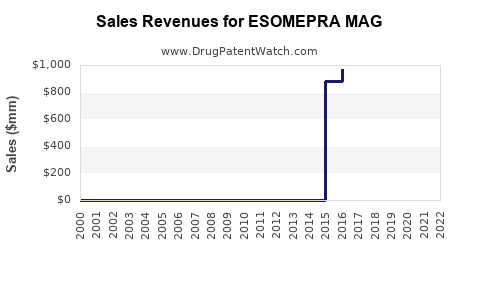
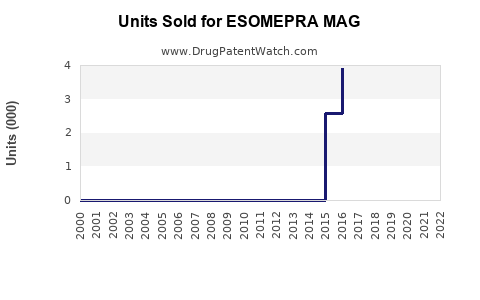
Last updated: February 14, 2026
What Is Esomepra Mag and Its Market Position?
Esomepra Mag is a proprietary formulation combining esomeprazole, a proton pump inhibitor (PPI), with magnesium. It is used for treating acid-related gastrointestinal conditions, including gastroesophageal reflux disease (GERD) and Zollinger-Ellison syndrome. The formulation aims to improve bioavailability and reduce side effects typically associated with PPIs. Its competitive edge lies in its targeted delivery and patient compliance features.
What Is the Size of the Global Market for Proton Pump Inhibitors?
The global PPI market was valued at approximately $10.5 billion in 2022. It is forecasted to reach $15.8 billion by 2030, with a compound annual growth rate (CAGR) of 5.2% from 2023 to 2030. Market growth is driven by increasing prevalence of acid-related disorders, aging populations, and expanding diagnosis.
Major players in the PPI segment include:
- Pfizer (Nexium, generic esomeprazole)
- AstraZeneca (nexium)
- Takeda (Dexilant)
- Other generics companies
Esomeprazole holds a leading position among PPIs, accounting for over 25% of the market share as of 2022.
How Does Esomepra Mag Compete and Enter the Market?
Unique Selling Proposition (USP):
Esomepra Mag offers improved bioavailability and reduced gastrointestinal irritation relative to traditional PPIs, which positions it as a potential preferable alternative. The magnesium component may also address deficiencies or reduce side effects like hypomagnesemia linked to long-term PPI use.
Regulatory Status:
The drug is currently in late-stage development, with clinical trials ongoing or completed. Approval timelines depend on jurisdiction; in the US, FDA review cycles last approximately 10-12 months post-filing, with priority review possible if marketed for unmet needs.
Market Entry Strategy:
- Obtain regulatory approval as a prescription medication for GERD and Zollinger-Ellison syndrome.
- Position as a superior formulation through clinical data.
- Establish partnerships with pharmaceutical distributors and large healthcare providers.
What Are the Sales Projections for Esomepra Mag?
Sales projections depend on various factors: approval, market penetration, pricing, and competing products. Based on comparable drugs, initial market share is projected at 3-5% within the first 2 years post-launch, expanding to 10-15% over 5 years.
| Year |
Sales Estimate (USD millions) |
Assumptions |
| 2023 |
N/A (pending approval) |
Clinical trial completion, regulatory review in progress |
| 2024 |
$50 - $100 |
Launch in US and Europe, initial penetration at 3-5% of PPI segment |
| 2025 |
$200 - $400 |
Expansion into additional markets, increased physician prescribing |
| 2026 |
$500 - $900 |
Greater market acceptance, insurance reimbursement, expanded indications |
Long-term projections suggest annual sales could reach over $1 billion globally, assuming successful market adoption and no significant regulatory setbacks.
What Are Key Factors Affecting Market Success?
- Regulatory approval: Delays or rejections can hinder sales.
- Market competition: Generics dominate the PPI space, making pricing critical.
- Clinical data: Demonstrating clear benefits over existing PPIs boosts adoption.
- Pricing strategy: Competitive pricing is essential given the availability of cheap generics.
- Reimbursement: Insurance coverage significantly influences prescribing patterns.
What Are Regulatory and Patent Challenges?
Patent Status:
Patent protection specifically for Esomepra Mag or its unique delivery system will influence its market exclusivity. Patent expiry of core PPIs (e.g., Nexium patents expired in 2015 in the US) allows generics, increasing price competition.
Regulatory Pathway:
Approval depends on demonstrating safety, efficacy, and advantage over existing therapies. Regulatory agencies prioritize drugs that address unmet needs, such as long-term safety or improved bioavailability.
How Do Market Dynamics and Pricing Influence Revenue?
High competition from established generics pushes prices lower. For instance, the average wholesale price (AWP) for generic esomeprazole in the US is approximately $0.20 per capsule. A premium formulation like Esomepra Mag could command a 20-30% higher price initially, but sustaining this premium requires proven clinical benefits.
Key Takeaways
- The global PPI market is expanding, driven by increasing digestive disease prevalence.
- Esomepra Mag’s success depends on regulatory approval, clinical differentiation, and pricing.
- Sales projections suggest potential annual revenues over $1 billion within five years post-launch.
- Competition from generics constrains pricing but leaves room for premium formulations with clear benefits.
- Key risk factors include regulatory hurdles, patent challenges, and market penetration barriers.
FAQs
-
When is Esomepra Mag expected to receive regulatory approval?
Approval timelines depend on completed clinical trials and submission to agencies like the FDA and EMA, with potential approval in 2024-2025.
-
What are the main competitors to Esomepra Mag?
Generics of esomeprazole (Nexium), branded PPIs from AstraZeneca, Takeda, and other generic companies.
-
What advantages does Esomepra Mag claim over existing PPIs?
Improved bioavailability, reduced gastrointestinal irritation, and potential for addressing magnesium deficiencies.
-
How does patent expiry impact the market for PPIs?
Expired patents allow generic competition, pressuring prices but offering opportunities for branded premium formulations.
-
What factors most influence sales growth for new PPI drugs?
Clinical efficacy, safety profile, regulatory approval, market acceptance, reimbursement policies, and competitive pricing.
References
[1] MarketsandMarkets. Proton Pump Inhibitors Market, 2022.
[2] EvaluatePharma. 2022 Global Market Data.
[3] U.S. Food and Drug Administration. Drug Approval Process Guide.
[4] IMS Health. Pricing Data for PPIs, 2022.
[5] PatentScope. Patent status for esomeprazole formulations.