Share This Page
Drug Sales Trends for BETAMETHASONE DIPROPIONATE
✉ Email this page to a colleague
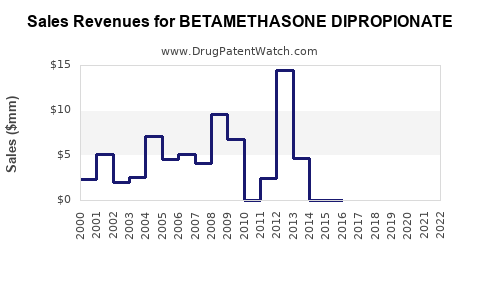
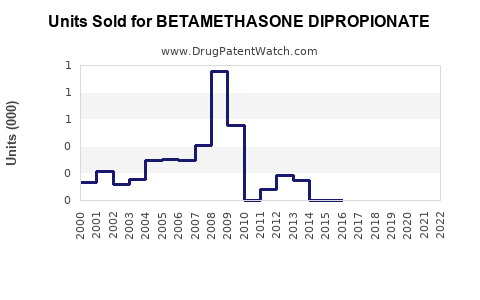
Annual Sales Revenues and Units Sold for BETAMETHASONE DIPROPIONATE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BETAMETHASONE DIPROPIONATE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BETAMETHASONE DIPROPIONATE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BETAMETHASONE DIPROPIONATE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| BETAMETHASONE DIPROPIONATE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| BETAMETHASONE DIPROPIONATE | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Betamethasone Dipropionate: Market Analysis and Sales Projections
Betamethasone dipropionate, a high-potency topical corticosteroid, holds a significant position in the dermatological market. Its efficacy in treating inflammatory skin conditions, including eczema, psoriasis, and dermatitis, drives sustained demand. The global market is characterized by a mature landscape with established generic competition alongside branded formulations. Future market growth will be influenced by patent expirations of key branded products, the emergence of novel delivery systems, and increased healthcare access in emerging economies.
What is the current market size and historical sales performance of betamethasone dipropionate?
The global market for topical corticosteroids, which includes betamethasone dipropionate, is substantial. While precise market segmentation for betamethasone dipropionate alone is often integrated within broader corticosteroid reports, industry estimates place the topical corticosteroid market in the billions of U.S. dollars annually. Historical sales have been driven by the broad applicability of the drug across various dermatological indications.
The market has experienced steady growth, albeit at a moderate pace due to the presence of generic alternatives. For instance, the U.S. market for topical corticosteroids, where betamethasone dipropionate is a frequently prescribed ingredient, demonstrates consistent sales volume. Data from IQVIA, a healthcare analytics firm, tracks the prescription volumes and sales of various corticosteroid products, indicating sustained prescription rates for betamethasone dipropionate formulations over the past decade. Brand-name products like Diprolene (Schering-Plough, now Merck) have historically been significant revenue drivers, but their market share has been progressively eroded by generic entries post-patent expiry.
Key Sales Drivers:
- Prevalence of Skin Conditions: Rising incidence of eczema, psoriasis, and allergic contact dermatitis globally.
- Physician Prescribing Habits: Established efficacy and physician familiarity with betamethasone dipropionate.
- Over-the-Counter (OTC) Availability: Certain lower-potency formulations and concentrations may be available OTC in some regions, expanding accessibility.
Challenges:
- Generic Competition: Widespread availability of lower-cost generic versions.
- Side Effects: Long-term use can lead to skin thinning, striae, and telangiectasias, prompting cautious prescribing.
- Development of Biologics: Newer, targeted biologic therapies are gaining traction for severe psoriasis and atopic dermatitis, potentially impacting the market share of traditional corticosteroids for these specific conditions.
What is the intellectual property landscape surrounding betamethasone dipropionate?
The intellectual property landscape for betamethasone dipropionate itself is largely expired, as the compound has been known and utilized for decades. The active pharmaceutical ingredient (API) is off-patent globally. However, patents can still exist around specific formulations, delivery systems, manufacturing processes, or combination therapies involving betamethasone dipropionate.
Key Patent Considerations:
- API Patents: Expired.
- Formulation Patents: These patents protect novel vehicles (e.g., foams, sprays, liposomes) or excipient combinations that enhance efficacy, reduce side effects, or improve patient compliance. For example, patents may cover specific cream or ointment bases designed for better penetration or reduced greasiness.
- Delivery System Patents: Innovations in topical delivery, such as specialized applicators or sustained-release technologies, can be patented.
- Combination Therapy Patents: Patents protecting the use of betamethasone dipropionate in combination with other active ingredients (e.g., antifungals, antibiotics) for synergistic therapeutic effects.
- Manufacturing Process Patents: Novel, more efficient, or environmentally friendly methods of synthesizing betamethasone dipropionate or its intermediates could be patented, although these are less common for well-established drugs.
Examples of Patent Activity:
- Novel Formulations: Companies may seek patents for improved formulations that offer enhanced efficacy or a better side-effect profile. For instance, a patent could cover a specific emollient base that improves the absorption of betamethasone dipropionate in patients with dry, scaly skin.
- Combination Products: Patents for combination products, such as betamethasone dipropionate with clotrimazole (an antifungal), are crucial for market differentiation and extended exclusivity. These are often marketed under brand names and can extend revenue streams beyond the API patent expiry.
The expiry of primary patents for betamethasone dipropionate has led to a highly competitive generic market. Companies active in this space include those specializing in API manufacturing and generic drug formulation. The focus of innovation and intellectual property is now on differentiated products and novel delivery mechanisms.
What are the key therapeutic indications and competitive landscape for betamethasone dipropionate?
Betamethasone dipropionate is primarily used for the relief of inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses. Its high potency makes it suitable for more severe or resistant conditions.
Primary Indications:
- Psoriasis: Particularly plaque psoriasis.
- Eczema (Atopic Dermatitis): For moderate to severe forms.
- Contact Dermatitis: Allergic and irritant types.
- Seborrheic Dermatitis: Scalp and facial involvement.
- Lichen Planus: Cutaneous lesions.
- Discoid Lupus Erythematosus: Localized lesions.
Competitive Landscape:
The competitive landscape for betamethasone dipropionate is segmented into branded products, generic formulations, and competing drug classes.
-
Other Potent Topical Corticosteroids:
- Clobetasol Propionate: Similar potency and indications, often considered the benchmark for super-potent corticosteroids.
- Halobetasol Propionate: Another super-potent corticosteroid.
- Fluocinonide: A potent corticosteroid with a broader range of formulations.
- Mometasone Furoate: While often classified as a mid-to-high potency corticosteroid depending on formulation, it competes in some indications.
-
Less Potent Topical Corticosteroids: Hydrocortisone, triamcinolone acetonide, and others are used for milder conditions and form a vast segment of the topical corticosteroid market, indirectly competing by being the first-line choice for less severe cases.
-
Non-Steroidal Dermatological Agents:
- Calcineurin Inhibitors: Tacrolimus and pimecrolimus are used for atopic dermatitis, particularly in sensitive areas where corticosteroids might be contraindicated due to side effects.
- PDE4 Inhibitors: Crisaborole offers a non-steroidal option for atopic dermatitis.
- Biologics: For moderate to severe psoriasis and atopic dermatitis, agents like ustekinumab, secukinumab, and dupilumab represent a significant and growing competitive threat, offering targeted mechanisms of action.
-
Generic vs. Branded Products:
- Branded Products: Historically, products like Diprolene (Merck) have held significant market share. These often benefit from established brand recognition and physician trust.
- Generic Products: A multitude of generic manufacturers offer betamethasone dipropionate in various strengths and formulations (creams, ointments, lotions). The price differential is substantial, driving significant volume for generics. Key generic players include Teva Pharmaceuticals, Mylan (Viatris), Sun Pharmaceutical Industries, and Accord Healthcare.
The market is characterized by a large number of generic entries following patent expiries of major branded products. This intense competition drives down prices for the API and generic formulations.
What are the projected market growth and sales forecasts for betamethasone dipropionate?
Projecting the market growth for betamethasone dipropionate requires considering several factors, including the overall growth of the topical corticosteroid market, the impact of generic competition, the prevalence of dermatological conditions, and the competitive pressure from newer therapeutic classes.
The market for betamethasone dipropionate is expected to experience modest growth, primarily driven by the increasing prevalence of inflammatory skin conditions globally. However, this growth will be tempered by several factors.
Key Growth Drivers:
- Rising Prevalence of Skin Diseases: Increasing rates of eczema, psoriasis, and allergic dermatitis, particularly in developing regions, will sustain demand. The World Health Organization (WHO) and various dermatological societies report increasing patient numbers for these conditions.
- Aging Population: The elderly population often experiences a higher incidence of skin conditions, contributing to demand for dermatological treatments.
- Emerging Market Penetration: Increased healthcare infrastructure and access to dermatological treatments in emerging economies (Asia Pacific, Latin America) will create new demand centers.
- Novel Formulations and Delivery Systems: While the API is generic, innovation in formulations (e.g., improved topical vehicles for enhanced delivery, combination products) can create niche market opportunities and command premium pricing.
Key Restraining Factors:
- Intense Generic Competition: The generic nature of the API will continue to exert downward pressure on prices and profit margins. The market is highly fragmented with numerous generic suppliers.
- Competition from Biologics and Targeted Therapies: For severe cases of psoriasis and atopic dermatitis, the growing adoption of biologics and other novel non-steroidal treatments will divert market share away from potent topical corticosteroids like betamethasone dipropionate.
- Side Effect Concerns: Physician and patient awareness of potential long-term side effects limits the use of potent topical steroids, especially for chronic management.
Sales Projections:
Given these dynamics, the global market for betamethasone dipropionate is projected to grow at a Compound Annual Growth Rate (CAGR) of 2.5% to 4.0% over the next five to seven years.
- Estimated Current Market Value (for betamethasone dipropionate formulations): Approximately $700 million to $900 million USD globally, considering all branded and generic sales.
- Projected Market Value in 5 Years: This would translate to an estimated market value of $815 million to $1.07 billion USD.
Regional Performance:
- North America and Europe: These regions will continue to be significant markets due to high healthcare spending and prevalence of skin conditions. Growth will be slower, driven by advanced formulations and a stable base of chronic dermatological patients.
- Asia Pacific: This region is expected to exhibit the highest growth rate (CAGR of 4.5% to 6.0%) due to increasing healthcare expenditure, a growing middle class, and rising awareness and diagnosis of dermatological disorders.
- Latin America, Middle East & Africa: These regions will also contribute to growth, albeit from a smaller base, as healthcare access improves.
The sales performance will be a mix of high-volume, low-margin generic sales and lower-volume, potentially higher-margin branded or specialized formulation sales. Companies that can innovate in drug delivery or offer combination products with demonstrated clinical benefits will be best positioned to capture value.
What are the manufacturing and supply chain considerations for betamethasone dipropionate?
The manufacturing of betamethasone dipropionate involves complex multi-step chemical synthesis. As a well-established drug, manufacturing processes are optimized for efficiency and cost. The supply chain is global, with API production often concentrated in countries with strong chemical manufacturing capabilities and lower production costs.
Manufacturing Process:
- API Synthesis: Betamethasone dipropionate is synthesized from precursor steroids. The process typically involves multiple chemical reactions, purification steps, and quality control measures to ensure purity and potency. Key raw materials are sourced from various chemical suppliers.
- Formulation: The API is then formulated into various dosage forms, primarily creams, ointments, lotions, and solutions. This involves mixing the API with excipients (e.g., emollients, emulsifiers, preservatives, thickeners) under controlled conditions. The choice of excipients is critical for stability, efficacy, and patient acceptance.
- Quality Control: Stringent quality control measures are implemented at every stage, from raw material sourcing to finished product release, to comply with regulatory standards (e.g., FDA, EMA). This includes testing for identity, purity, potency, and microbial contamination.
Supply Chain:
- API Manufacturers: A significant number of API manufacturers globally produce betamethasone dipropionate. Key manufacturing hubs include India and China, known for their cost-effective production of generic APIs. However, manufacturers in Europe and North America also exist, often focusing on higher-purity or specialized grades.
- Formulators and Finished Dosage Manufacturers: These are pharmaceutical companies that purchase the API and manufacture the final drug products. This segment includes both large multinational corporations with brand portfolios and numerous generic drug companies.
- Distribution: Finished products are distributed through pharmaceutical wholesalers and directly to pharmacies and healthcare providers. The supply chain is subject to regulations regarding storage, transportation, and handling of pharmaceutical products.
- Regulatory Compliance: All manufacturing sites and processes must comply with Good Manufacturing Practices (GMP) standards set by regulatory authorities. Audits and inspections are common.
Key Supply Chain Considerations:
- Raw Material Sourcing: The availability and cost of precursor chemicals are critical. Geopolitical factors or disruptions in the supply of these raw materials can impact production.
- Cost Management: Given the high level of generic competition, efficient manufacturing and supply chain management are essential for profitability.
- Regulatory Hurdles: Obtaining and maintaining regulatory approvals for manufacturing sites and products in different markets is a significant undertaking. Changes in regulations can necessitate process modifications or revalidation.
- Intellectual Property in Processes: While the API is off-patent, patents on novel or significantly improved manufacturing processes can provide a competitive advantage.
- Supply Chain Resilience: Recent global events have highlighted the importance of resilient supply chains. Companies are increasingly looking to diversify suppliers and reduce reliance on single geographic sources.
- Environmental, Social, and Governance (ESG) Factors: Increasing scrutiny is being placed on the environmental impact of chemical manufacturing, including waste management and energy consumption.
The supply chain for betamethasone dipropionate is mature and highly competitive. Success hinges on cost-efficiency, robust quality assurance, and reliable global distribution networks.
What are the key challenges and opportunities for betamethasone dipropionate manufacturers?
Manufacturers of betamethasone dipropionate face a dual challenge of a highly competitive generic market and evolving therapeutic landscapes, alongside opportunities for differentiation and market expansion.
Challenges:
- Price Erosion from Generic Competition: The most significant challenge is the intense price competition from numerous generic manufacturers. This limits profit margins and necessitates aggressive cost management.
- Stagnant Market Growth for Standard Formulations: The market for basic creams and ointments is largely saturated, with growth driven primarily by volume rather than price increases.
- Competition from Biologics and Novel Therapies: For severe dermatological conditions like moderate-to-severe psoriasis and atopic dermatitis, newer targeted therapies (biologics, JAK inhibitors) are gaining market share, potentially reducing the reliance on potent topical corticosteroids.
- Side Effect Profile Limitations: The known side effects of long-term or widespread topical corticosteroid use (e.g., skin atrophy, adrenal suppression) can limit their use, particularly in sensitive areas or for chronic management, pushing patients and physicians towards alternatives.
- Regulatory Scrutiny: Increased regulatory focus on impurities, manufacturing quality, and environmental impact can lead to higher compliance costs.
Opportunities:
- Novel Drug Delivery Systems: Developing and patenting improved topical formulations that enhance efficacy, reduce side effects, or improve patient compliance can create market differentiation. Examples include:
- Foams and Sprays: For easier application and reduced greasiness.
- Liposomal Formulations: To improve skin penetration and drug delivery.
- Combination Products: Formulations combining betamethasone dipropionate with other actives (e.g., antifungals, antibiotics, keratolytics) to address complex dermatological conditions and provide a more comprehensive treatment.
- Emerging Markets Expansion: Significant growth potential exists in emerging economies in Asia, Latin America, and Africa, where healthcare access is improving and the prevalence of skin diseases is high. Manufacturers can establish a strong presence by offering cost-effective generic options.
- Specialty Formulations for Specific Indications: Tailoring formulations for particular patient populations or specific skin conditions (e.g., scalp formulations, products for sensitive skin) can create niche markets.
- Contract Manufacturing: Companies with robust manufacturing capabilities and a strong quality track record can leverage their expertise to provide contract manufacturing services for other pharmaceutical companies.
- Cost Optimization through Process Innovation: Continuous improvement in manufacturing processes to reduce costs and increase efficiency can provide a competitive edge in the generic market.
- Strategic Partnerships and Acquisitions: Collaborating with or acquiring companies that possess innovative delivery technologies or strong market access in key regions can accelerate growth.
Manufacturers that can successfully navigate the price pressures of the generic market by innovating in product formulation, expanding into high-growth geographies, or optimizing their operational efficiency are best positioned for sustained success.
Key Takeaways
- Betamethasone dipropionate is a well-established, high-potency topical corticosteroid with a significant market share in the treatment of inflammatory skin conditions.
- The intellectual property landscape for the active pharmaceutical ingredient (API) is expired, leading to a highly competitive generic market. Innovation is focused on patented formulations and delivery systems.
- Key indications include psoriasis, eczema, and contact dermatitis. Competition arises from other potent topical corticosteroids, non-steroidal agents, and emerging biologics.
- The market is projected to grow at a modest CAGR of 2.5% to 4.0% globally, driven by the increasing prevalence of skin diseases and expansion in emerging markets, offset by generic price erosion and competition from newer therapies.
- Manufacturing is globalized, with API production concentrated in cost-competitive regions like India and China. Supply chain resilience and cost management are critical.
- Opportunities lie in novel delivery systems, emerging market penetration, specialty formulations, and cost optimization. Challenges include intense price competition and the rise of biologics.
Frequently Asked Questions
-
Are there any new clinical trials underway for betamethasone dipropionate? Clinical trials primarily focus on novel formulations, combination therapies, or comparative effectiveness studies against newer treatments rather than the API itself, which is a well-understood compound. Information on specific ongoing trials can be found in databases like ClinicalTrials.gov.
-
What is the typical price range for generic betamethasone dipropionate formulations? The price of generic betamethasone dipropionate formulations varies widely based on the specific product (cream, ointment, lotion), strength, quantity, and geographical market. However, in the U.S., a typical 15g or 30g tube of a generic betamethasone dipropionate cream or ointment can range from approximately $10 to $30 USD, significantly lower than branded counterparts.
-
What are the main side effects associated with the long-term use of betamethasone dipropionate? Long-term or excessive use of betamethasone dipropionate can lead to localized side effects such as skin thinning (atrophy), stretch marks (striae), dilated blood vessels (telangiectasias), acneiform eruptions, and depigmentation. Systemic absorption can rarely lead to effects similar to oral corticosteroids, particularly with widespread application on large surface areas or under occlusion.
-
How does betamethasone dipropionate compare in potency to other common topical corticosteroids? Betamethasone dipropionate is classified as a super-potent or class I corticosteroid. It is considered more potent than triamcinolone acetonide (class II or III depending on formulation) and hydrocortisone (class VI or VII). Its potency is comparable to clobetasol propionate and halobetasol propionate.
-
What regulatory bodies oversee the approval and manufacturing of betamethasone dipropionate products? The approval and manufacturing of betamethasone dipropionate products are overseen by national and regional regulatory agencies. Key bodies include the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), Health Canada, Japan's Pharmaceuticals and Medical Devices Agency (PMDA), and the Therapeutic Goods Administration (TGA) in Australia.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Approval Process. Retrieved from https://www.fda.gov/about-fda/center-drug-evaluation-and-research-cder/drug-approval-process [2] European Medicines Agency. (n.d.). How we authorise medicines. Retrieved from https://www.ema.europa.eu/en/how-we-authorise-medicines [3] IQVIA. (n.d.). Market Intelligence and Analytics. Retrieved from https://www.iqvia.com/ (Note: Specific reports detailing topical corticosteroid market data are proprietary and require subscription.) [4] World Health Organization. (n.d.). Skin diseases. Retrieved from https://www.who.int/health-topics/skin-diseases [5] United States Patent and Trademark Office. (n.d.). Patents. Retrieved from https://www.uspto.gov/patents [6] ClinicalTrials.gov. (n.d.). Betamethasone dipropionate. Retrieved from https://clinicaltrials.gov/ (Searches for specific trials related to the compound.) [7] Viatris Inc. (n.d.). Product Information. Retrieved from https://www.viatris.com/ (Example of a generic manufacturer's product information.) [8] Merck & Co., Inc. (n.d.). Products. Retrieved from https://www.merck.com/ (Example of a branded pharmaceutical company's product information.)
More… ↓
