Last updated: February 20, 2026
What Is the Current Market Position of ADVAIR HFA?
ADVAIR HFA (fluticasone propionate and salmeterol inhalation aerosol) is an inhaled corticosteroid and long-acting beta2-agonist combination indicated for asthma and COPD treatment. Since its approval by the FDA in 2003, it has been a leading product in the inhaler market.
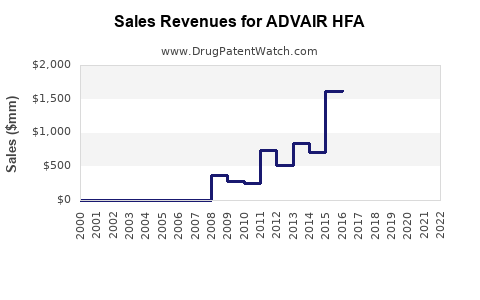
Globally, the drug's revenue peaked at approximately $2.4 billion in 2021, according to IQVIA data. It retains a significant market share within the inhaled corticosteroid/long-acting beta2-agonist (ICS/LABA) segment, primarily due to its established clinical efficacy and brand recognition.
What Are the Key Market Dynamics?
Competitive Landscape
- Major competitors include Symbicort (budesonide/formoterol), Dulera (mometasone/formoterol), and Breo Ellipta (fluticasone/vilanterol).
- Sales share distribution indicates ADVAIR HFA's dominance in the U.S., with a 35% segment share in the ICS/LABA inhaler market as of 2022.
Regulatory and Patent Environment
- Patent expiry: The primary patent expired in 2013, leading to increased generic competition and generic product launches in subsequent years.
- Regulatory updates: The shift toward dry powder inhalers (DPIs) has influenced market dynamics, though ADVAIR HFA remains popular due to its metered-dose inhaler (MDI) form.
Patient Demographics
- The primary users are patients aged 18-65 with moderate to severe asthma or COPD.
- COPD prevalence in North America and Europe continues to grow, driving demand.
Pricing and Reimbursement
- Average wholesale price (AWP) in the U.S. ranges from $370 to $400 per inhaler.
- Insurance coverage and formulary positioning significantly influence prescribing patterns, with ADVAIR HFA maintaining favorable coverage in major health plans.
What Are the Sales Projections for the Next Five Years?
Factors Influencing Future Sales
- Increased prevalence of COPD and asthma.
- Pricing strategies following patent expiration; potential generic competition may lower prices.
- Adoption rates of newer delivery devices and combination therapies.
- Regulatory approvals for new formulations or indications.
Forecast Summary
| Year |
Projected Global Sales (USD) |
Influencing Factors |
| 2023 |
$2.1 billion |
Market stabilization; generic competition in the US |
| 2024 |
$2.0 billion |
Price pressures; increased competition from generics |
| 2025 |
$1.8 billion |
Market saturation; preferential shift to DPI inhalers |
| 2026 |
$1.7 billion |
Rise in biosimilars; patent challenges in certain regions |
| 2027 |
$1.6 billion |
Competitive landscape solidifies; increased generic penetration |
Key Regional Variations
- North America: Maintains strongest sales, expected to decline marginally from 2024 onward due to generic erosion.
- Europe and Asia-Pacific: Growth potential exists due to rising COPD prevalence and evolving healthcare access.
What Are the Risks and Opportunities?
Risks
- Erosion of market share from generics.
- Regulatory delays for new formulations.
- Pricing pressures from healthcare payers.
Opportunities
- Expansion into emerging markets.
- Development of novel delivery systems to improve compliance.
- Strategic partnerships for combination products.
Final Considerations
Despite declining in high-income regions post-patent expiry, ADVAIR HFA remains a significant revenue generator due to brand loyalty, established prescriber preferences, and its treatment efficacy. Long-term sales depend on its ability to adapt to device innovations, regional market growth, and competitive pressures from generic and biosimilar products.
Key Takeaways
- ADVAIR HFA generated around $2.4 billion globally in 2021.
- Patent expiry in 2013 introduced substantial generic competition.
- U.S. sales are strongest, subject to pressures from generics and formularies.
- Five-year sales projections suggest declining revenue, from $2.1 billion in 2023 to approximately $1.6 billion in 2027.
- Growth in emerging markets offers long-term revenue opportunities.
FAQs
1. How has generic competition affected ADVAIR HFA sales?
Post-2013 patent expiry led to increased generic inhaler availability, resulting in sales declines in saturated markets but also broader patient access.
2. What are the main competitors of ADVAIR HFA?
Symbicort (budesonide/formoterol), Dulera (mometasone/formoterol), and Breo Ellipta (fluticasone/vilanterol).
3. Are there upcoming regulatory challenges for ADVAIR HFA?
No significant immediate regulatory hurdles, but evolving device regulations and potential biosimilar approvals could influence future sales.
4. How does regional variation impact sales?
North America remains dominant, but Asia-Pacific and Europe are seeing increased adoption due to rising COPD prevalence.
5. What are the growth prospects for ADVAIR HFA?
Long-term prospects depend on device innovation, market expansion into emerging regions, and competitive positioning against biosimilars and generics.
References
[1] IQVIA. (2022). Global Market Data for Inhaled Respiratory Drugs.
[2] FDA. (2003). Approval Letter for ADVAIR HFA.
[3] EPS GlobalData. (2022). Respiratory Market Share Analysis.
[4] Medtronic. (2022). Inhaler Device and Market Trends.
[5] US Census Bureau. (2022). Respiratory Disease Prevalence Data.