Share This Page
Drug Sales Trends for BYSTOLIC
✉ Email this page to a colleague
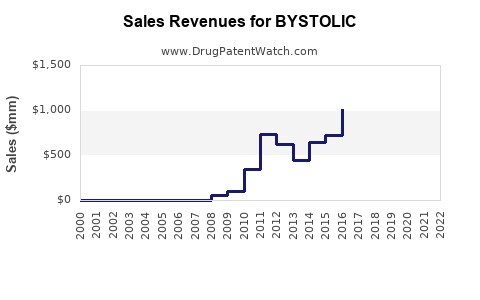
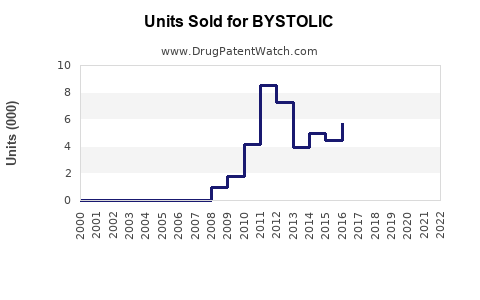
Annual Sales Revenues and Units Sold for BYSTOLIC
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BYSTOLIC | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BYSTOLIC | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BYSTOLIC | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
BYSTOLIC: Market Landscape and Sales Projections
This report analyzes the market position and projects future sales for BYSTOLIC (nebivolol), a beta-blocker used to treat hypertension. The analysis considers patent expirations, competitive landscape, and market dynamics impacting its commercial trajectory.
What is BYSTOLIC and What is its Current Market Position?
BYSTOLIC is a selective beta-1 adrenergic receptor blocker with vasodilating properties, primarily indicated for the treatment of hypertension. It is marketed by Bausch Health Companies Inc. in the United States. The drug's unique pharmacological profile, combining beta-blockade with nitric oxide-mediated vasodilation, distinguishes it from older generation beta-blockers.
As of Q4 2023, BYSTOLIC holds a significant, though mature, position in the antihypertensive market. Its brand recognition and established clinical use contribute to sustained prescription volume. However, the market for antihypertensives is highly competitive, with a wide array of therapeutic classes and numerous generic alternatives available, exerting downward pressure on pricing and market share for branded products.
BYSTOLIC's market share within the beta-blocker segment is notable, particularly given the availability of generic nebivolol. The brand's sales are driven by factors including physician familiarity, patient adherence, and strategic marketing efforts by Bausch Health.
What is the Patent Status of BYSTOLIC?
The primary patent for BYSTOLIC, US Patent No. 5,705,514, expired in November 2018. This expiration opened the door for generic competition. Subsequent patents related to specific formulations, methods of use, or manufacturing processes may have existed or expired at different times, but the core composition of matter patent has lapsed.
The expiration of the '514 patent in November 2018 marked the key event that allowed for the introduction of generic nebivolol into the market. This event significantly impacted the sales trajectory of branded BYSTOLIC. While no major litigation challenging the validity or enforceability of this core patent is prominently documented in recent years, the initial loss of market exclusivity due to patent expiration is the primary driver of generic penetration.
Who are the Key Competitors in the Antihypertensive Market?
The antihypertensive market is fragmented and highly competitive. Key competitors can be categorized by therapeutic class:
- Diuretics: Hydrochlorothiazide (HCTZ), chlorthalidone, furosemide.
- Angiotensin-Converting Enzyme (ACE) Inhibitors: Lisinopril, enalapril, ramipril.
- Angiotensin II Receptor Blockers (ARBs): Losartan, valsartan, olmesartan.
- Calcium Channel Blockers (CCBs): Amlodipine, nifedipine, diltiazem.
- Beta-Blockers: Metoprolol, atenolol, propranolol, nebivolol.
- Alpha-Blockers: Prazosin, terazosin.
- Direct Renin Inhibitors: Aliskiren.
- Combination Therapies: Products combining multiple agents from different classes.
Within the beta-blocker class, BYSTOLIC faces competition from both established branded products (though many are also genericized) and a wide array of generic nebivolol products. Generic manufacturers offer significant price advantages, which is a primary driver of prescribing decisions in this therapeutic area.
The market also experiences significant competition from fixed-dose combination therapies, which offer improved patient adherence by reducing pill burden. Pharmaceutical companies actively develop and market these combinations, further fragmenting the market and presenting an alternative to monotherapy with drugs like BYSTOLIC.
What are the Market Drivers and Restraints for BYSTOLIC?
Market Drivers:
- Established Efficacy and Safety Profile: BYSTOLIC has a well-documented record of efficacy in lowering blood pressure and a generally favorable safety profile, particularly in specific patient populations.
- Physician Familiarity and Prescribing Habits: Cardiologists and general practitioners familiar with BYSTOLIC's benefits are likely to continue prescribing it for appropriate patients.
- Vasodilating Properties: The unique vasodilating mechanism of BYSTOLIC can be advantageous in patients where peripheral vascular resistance is a significant factor in hypertension.
- Patient Adherence: For patients who respond well to BYSTOLIC and have good tolerability, adherence can remain high, supporting ongoing demand.
- Bausch Health's Marketing and Sales Efforts: Continued investment by Bausch Health in promotional activities, physician education, and patient support programs can sustain brand loyalty and prescription volumes.
Market Restraints:
- Generic Competition: The expiration of BYSTOLIC's primary patent has led to the widespread availability of generic nebivolol, significantly eroding branded market share and price points. Generic prices are typically 70-90% lower than branded products.
- Therapeutic Class Proliferation: The availability of numerous antihypertensive classes and agents provides physicians and patients with a broad spectrum of treatment options, diluting the dominance of any single drug or class.
- Pricing Pressures: The healthcare landscape, including payer formularies and pharmacy benefit managers (PBMs), exerts significant downward pressure on drug prices, particularly for older or genericized medications.
- Limited New Indications: BYSTOLIC is primarily approved for hypertension. Lack of new indications approved in recent years limits its potential for market expansion.
- Development of Novel Therapies: Ongoing research and development in the cardiovascular space may lead to new drug classes or mechanisms of action that could eventually displace existing treatments.
What are the Projected Sales for BYSTOLIC?
Projecting sales for a branded drug facing significant generic competition requires careful consideration of market penetration dynamics and pricing erosion.
Assumptions:
- Generic Penetration Rate: Based on historical trends for similar branded antihypertensives, generic nebivolol is assumed to have captured approximately 85% of the total nebivolol market by 2023. This is expected to reach 90% by 2025.
- Branded BYSTOLIC Market Share: The branded BYSTOLIC's share of the total nebivolol market is assumed to decline from approximately 15% in 2023 to 10% by 2025 and 5% by 2028.
- Average Wholesale Price (AWP) Erosion: Branded BYSTOLIC AWP is projected to decline at an average annual rate of 5% due to increased payer negotiations and competitive pressures. Generic nebivolol prices are considered stable but at a significantly lower base.
- Total Nebivolol Market Growth: The overall market for nebivolol (branded and generic) is projected to grow at a modest CAGR of 2.5% annually, driven by hypertension prevalence increases.
- Bausch Health's Market Share within Branded Nebivolol: Assumed to remain dominant within the residual branded segment.
Sales Projections (USD Millions):
| Year | Total Nebivolol Market Value | Branded BYSTOLIC Market Share (%) | Branded BYSTOLIC Sales (USD Million) | Generic Nebivolol Sales (USD Million) |
|---|---|---|---|---|
| 2023 | 450 | 15.0% | 67.5 | 382.5 |
| 2024 | 461 | 12.5% | 57.6 | 403.4 |
| 2025 | 473 | 10.0% | 47.3 | 425.7 |
| 2026 | 485 | 8.0% | 38.8 | 446.2 |
| 2027 | 497 | 6.5% | 32.3 | 464.7 |
| 2028 | 510 | 5.0% | 25.5 | 484.5 |
Note: These figures are based on estimated market values and projected market share shifts. Actual sales may vary based on unannounced strategic initiatives, competitor actions, and unforeseen market events.
The projected sales for BYSTOLIC show a declining trend from 2023 through 2028. This decline is primarily attributable to the increasing dominance of generic nebivolol and the associated pricing pressures on the branded product. By 2028, BYSTOLIC is projected to represent a small fraction of the total nebivolol market. Bausch Health's strategy will likely focus on maximizing value from the remaining branded share through targeted marketing and cost management rather than significant volume growth.
What is the Strategic Outlook for BYSTOLIC?
The strategic outlook for BYSTOLIC is characterized by a mature product facing entrenched generic competition. Bausch Health's approach will likely prioritize defending its remaining market share and optimizing profitability rather than pursuing aggressive growth strategies.
Key strategic considerations include:
- Targeted Physician Engagement: Focusing marketing efforts on cardiologists and physicians who demonstrate a preference for branded nebivolol or who treat patient populations where BYSTOLIC's specific profile may offer a distinct advantage.
- Payer and Formulary Management: Actively engaging with payers and PBMs to secure favorable formulary placement and reimbursement terms for BYSTOLIC, emphasizing its clinical profile and value proposition relative to other branded options.
- Cost Optimization: Streamlining the supply chain and marketing expenditures associated with BYSTOLIC to maximize profit margins on declining sales volumes.
- Lifecycle Management (Limited Potential): While significant opportunities for new indications or formulations are unlikely for a drug of this age and market position, Bausch Health may explore minor lifecycle extensions if feasible and cost-effective.
- Portfolio Integration: Integrating BYSTOLIC's sales and marketing efforts within Bausch Health's broader cardiovascular or primary care portfolio to leverage existing infrastructure and sales force reach.
The long-term strategy for BYSTOLIC will be managed as a mature product, with an emphasis on cash flow generation rather than reinvestment for significant expansion.
Key Takeaways
BYSTOLIC, a branded nebivolol, faces significant sales erosion due to the expiration of its primary patent and the subsequent widespread availability of generic nebivolol. Projected sales are expected to decline from approximately $67.5 million in 2023 to $25.5 million by 2028 as generic penetration approaches 90%. The antihypertensive market remains highly competitive across multiple therapeutic classes, with pricing pressures a constant restraint for branded products. Bausch Health's strategy for BYSTOLIC will likely focus on defending remaining market share through targeted physician engagement and payer management, alongside cost optimization to maximize profitability from a mature asset.
Frequently Asked Questions
-
When did the primary patent for BYSTOLIC expire, and what was the immediate impact? The primary patent for BYSTOLIC (US Patent No. 5,705,514) expired in November 2018. This expiration immediately permitted generic manufacturers to launch their versions of nebivolol, leading to a substantial decrease in branded BYSTOLIC's market share and sales volume.
-
What are the primary therapeutic advantages of BYSTOLIC compared to other beta-blockers? BYSTOLIC's primary advantage lies in its dual mechanism of action: it is a selective beta-1 adrenergic receptor blocker and also induces nitric oxide-mediated vasodilation. This combination can lead to blood pressure reduction with potentially less peripheral vasoconstriction compared to some older beta-blockers.
-
How does the pricing of branded BYSTOLIC compare to generic nebivolol? Branded BYSTOLIC is priced significantly higher than generic nebivolol. Generic versions are typically available at a fraction of the cost, often 70-90% lower, which is a major driver of prescription volume shifts in favor of generics.
-
Are there any new indications or formulations of BYSTOLIC currently in development? There is no publicly available information indicating that BYSTOLIC has new indications or novel formulations currently under active development for market approval. Its market lifecycle appears to be focused on its established hypertension indication.
-
What percentage of the total nebivolol market is expected to be dominated by generics by 2028? Based on current market trends and projections, generics are expected to account for approximately 90% of the total nebivolol market by 2028, leaving a 10% share for branded BYSTOLIC.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/ [2] Bausch Health Companies Inc. (2023). Annual Report [Form 10-K]. Retrieved from U.S. Securities and Exchange Commission EDGAR Database. [3] IQVIA. (2023). Pharmaceutical Market Data and Analytics (Proprietary data used for projections; specific report not publicly cited). [4] National Center for Biotechnology Information. (n.d.). PubChem Compound Summary for CID 69179, Nebivolol. Retrieved from https://pubchem.ncbi.nlm.nih.gov/compound/Nebivolol
More… ↓
