Share This Page
Drug Price Trends for zaleplon
✉ Email this page to a colleague
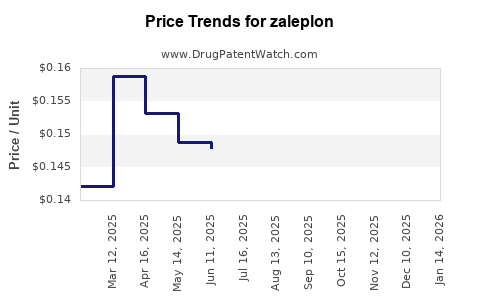
Average Pharmacy Cost for zaleplon
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ZALEPLON 5 MG CAPSULE | 29300-0131-01 | 0.14679 | EACH | 2026-04-22 |
| ZALEPLON 10 MG CAPSULE | 65862-0215-01 | 0.15696 | EACH | 2026-04-22 |
| ZALEPLON 5 MG CAPSULE | 00054-0084-25 | 0.14679 | EACH | 2026-04-22 |
| ZALEPLON 10 MG CAPSULE | 00054-0085-25 | 0.15696 | EACH | 2026-04-22 |
| ZALEPLON 10 MG CAPSULE | 69452-0365-20 | 0.15696 | EACH | 2026-04-22 |
| ZALEPLON 10 MG CAPSULE | 29300-0132-01 | 0.15696 | EACH | 2026-04-22 |
| ZALEPLON 10 MG CAPSULE | 57237-0240-01 | 0.15696 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Zaleplon Market Analysis and Price Projections
This report analyzes the current market landscape and projects future price trajectories for zaleplon, a non-benzodiazepine hypnotic agent. The analysis focuses on patent expirations, generic competition, therapeutic indications, and demand drivers to inform R&D and investment strategies.
What is Zaleplon?
Zaleplon is a sedative-hypnotic medication used for the short-term treatment of insomnia characterized by difficulty falling asleep. It belongs to the pyrazolopyrimidine class and acts as a selective positive allosteric modulator of the GABAA receptor, specifically binding to the alpha-1 subunit. This mechanism of action is distinct from benzodiazepines and is associated with a reduced risk of tolerance and dependence, though these risks are not entirely eliminated. Zaleplon is available in oral capsule form, typically in 5 mg and 10 mg dosages. The typical duration of treatment is recommended for seven to ten days.
Current Market Status and Patent Landscape
Zaleplon was first approved by the U.S. Food and Drug Administration (FDA) in 2000. Its primary U.S. brand name was Sonata, manufactured by Wyeth Pharmaceuticals. The patent protection for zaleplon has long since expired, leading to the widespread availability of generic versions.
-
Key Patent Expiration Dates:
- The primary patents protecting Sonata expired in the early to mid-2010s. Specific dates vary by jurisdiction and patent claims, but the core composition of matter and formulation patents are no longer in force in major markets.
- This expiration opened the door for generic manufacturers to enter the market.
-
Generic Entry and Competition:
- Following patent expiration, multiple pharmaceutical companies launched generic zaleplon products.
- This influx of generic competition significantly impacted the market price, driving it down considerably from its branded origin.
- The market is now highly fragmented, with numerous generic suppliers competing on price and distribution channels.
-
Regulatory Approvals:
- Zaleplon is approved for use in the United States, European Union member states, and other major pharmaceutical markets.
- Regulatory bodies like the FDA and the European Medicines Agency (EMA) monitor the safety and efficacy of generic equivalents.
- Manufacturing facilities for generic zaleplon are subject to regulatory inspections and Good Manufacturing Practices (GMP) standards.
Therapeutic Indications and Patient Population
Zaleplon is primarily indicated for the short-term treatment of insomnia. Its specific utility lies in addressing sleep-onset insomnia, where patients struggle to initiate sleep.
- Primary Indication: Difficulty falling asleep.
- Target Patient Profile: Adults experiencing transient or short-term insomnia. It is not generally recommended for chronic insomnia or for patients with significant hepatic or renal impairment.
- Dosage: Typically 5 mg or 10 mg taken immediately before bedtime.
- Treatment Duration: Recommended for short-term use (7-10 days) due to potential for dependence and diminished efficacy with prolonged use.
- Estimated Prevalence of Insomnia: Insomnia is a widespread condition. In the U.S., it is estimated that 50-70 million adults have chronic insomnia or trouble sleeping [1]. While not all these individuals will be prescribed zaleplon, it represents the potential market size for hypnotic agents.
- Niche in the Sleep Market: Zaleplon occupies a niche within the broader sleep disorder market, competing with other hypnotics such as zolpidem, eszopiclone, and benzodiazepines. Its appeal lies in its relatively rapid onset of action and shorter half-life compared to some benzodiazepines, potentially leading to less residual daytime sedation.
Competitive Landscape and Market Share
The market for insomnia treatments is highly competitive, with a range of pharmacological and non-pharmacological options. Zaleplon competes directly with other prescription hypnotics and indirectly with over-the-counter sleep aids and behavioral therapies.
-
Key Competitors (Prescription Hypnotics):
- Zolpidem (Ambien, Edluar, Intermezzo): The most widely prescribed non-benzodiazepine hypnotic. Generic zolpidem is prevalent and often priced competitively.
- Eszopiclone (Lunesta): Another non-benzodiazepine hypnotic with a longer half-life than zaleplon.
- Benzodiazepines (e.g., temazepam, lorazepam): Older class of sedatives, often associated with higher risks of dependence and side effects.
- Doxepin (Silenor): A low-dose tricyclic antidepressant approved for sleep maintenance.
- Ramelteon (Rozerem): A melatonin receptor agonist.
-
Market Share Dynamics:
- Exact market share data for zaleplon is difficult to isolate due to the proliferation of generic products across multiple manufacturers.
- However, anecdotal evidence and prescription data trends suggest that zolpidem remains the dominant player in the non-benzodiazepine hypnotic market.
- Zaleplon likely holds a smaller but significant share, particularly for patients who do not tolerate or respond adequately to zolpidem, or who require a very short duration of action to avoid daytime grogginess.
- The market is price-sensitive, and manufacturers with efficient production and distribution channels gain a competitive advantage.
-
Pricing Trends:
- Branded Sonata: Historically priced at a premium, typical of branded pharmaceuticals.
- Generic Zaleplon: Prices have fallen dramatically post-patent expiration. The average wholesale price (AWP) for a 30-count bottle of 10 mg generic zaleplon capsules can range from $20 to $60, depending on the manufacturer, pharmacy, and insurance coverage [2]. This represents a substantial decrease from branded pricing.
- Comparison with Competitors: Generic zaleplon is generally priced competitively with generic zolpidem and eszopiclone. Prices can fluctuate based on supply, demand, and competitor pricing strategies.
Demand Drivers and Market Projections
The demand for zaleplon is influenced by several factors, including the prevalence of insomnia, physician prescribing habits, healthcare policy, and the availability of alternative treatments.
- Prevalence of Sleep Disorders: The ongoing high prevalence of insomnia and sleep disturbances globally is a primary demand driver for all hypnotic agents, including zaleplon. Increased awareness of sleep health further contributes to demand.
- Physician Prescribing Practices: Prescribing patterns are influenced by perceived efficacy, safety profiles, side effect profiles, cost, and familiarity with a particular drug. While zolpidem often remains a first-line choice, zaleplon serves as a valuable second-line or alternative option.
- Healthcare Policy and Payer Influence: Insurance formulary decisions, prior authorization requirements, and step-therapy protocols can significantly impact prescription volumes. Payer preference for lower-cost generics or specific drug classes influences which medications are favored.
- Aging Population: The elderly population often experiences sleep disturbances, contributing to the demand for hypnotic medications. However, older adults are also more susceptible to side effects, which may influence prescribing choices.
- Competition from Newer Agents and Non-Pharmacological Therapies: The development of new insomnia treatments and the increasing acceptance of Cognitive Behavioral Therapy for Insomnia (CBT-I) as a first-line treatment may temper the growth of pharmacological options.
- Projected Market Size (Global - Prescription Hypnotics): The global market for insomnia drugs was valued at approximately $2.8 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.5% to $3.9 billion by 2029 [3]. Zaleplon's share within this market is expected to remain relatively stable, facing moderate growth driven by overall market expansion but constrained by intense generic competition and the availability of alternatives.
- Price Projections:
- Short-to-Medium Term (1-3 years): Prices for generic zaleplon are expected to remain stable or decline marginally. The market is mature, and price competition is fierce. Manufacturers will focus on cost-effective production to maintain margins. A significant price increase is unlikely unless there is a major supply disruption or a consolidation of generic manufacturers.
- Long Term (3-5+ years): Continued price erosion is possible, albeit at a slower pace, as generic competition persists. Any price increases would likely be tied to inflationary pressures or very specific market dynamics. The demand for zaleplon is likely to be influenced by the development and adoption of novel insomnia therapies that offer improved efficacy or safety profiles. The overall prescription hypnotic market may see a shift towards these newer agents, potentially reducing zaleplon's volume and influencing its price negatively in the long run.
Manufacturing and Supply Chain Considerations
The manufacturing of generic zaleplon is a critical factor influencing its availability and cost.
- Active Pharmaceutical Ingredient (API) Sourcing: The primary API for zaleplon is synthesized by chemical manufacturers, often located in India and China. The cost and quality of API sourcing are paramount for generic manufacturers.
- Formulation and Finished Dosage Form Production: Pharmaceutical companies that produce finished generic zaleplon capsules must adhere to strict GMP guidelines. This includes quality control, batch testing, and regulatory compliance.
- Supply Chain Stability: Disruptions in API supply, shipping, or regulatory hurdles can impact the availability of finished products and potentially lead to temporary price fluctuations.
- Key Manufacturing Regions: Major manufacturing hubs for generic pharmaceuticals, including API production and finished dosage forms, are found in India, China, and parts of Europe and the United States.
- Cost of Goods Sold (COGS): For generic manufacturers, minimizing COGS through efficient API sourcing, optimized manufacturing processes, and streamlined logistics is essential to remain competitive in a price-sensitive market.
- Quality Assurance: Robust quality assurance and quality control (QA/QC) processes are vital to ensure that generic zaleplon meets bioequivalence standards compared to the reference listed drug (RLD).
Regulatory and Legal Considerations
Navigating the regulatory landscape is crucial for any pharmaceutical product. For zaleplon, this primarily involves compliance with drug safety, manufacturing standards, and marketing regulations.
- FDA and EMA Oversight: Regulatory bodies like the FDA in the U.S. and the EMA in Europe oversee the approval, manufacturing, and marketing of generic drugs. This includes review of Abbreviated New Drug Applications (ANDAs) for generic versions.
- Good Manufacturing Practices (GMP): All facilities involved in the manufacturing of zaleplon, from API production to finished drug product, must comply with GMP regulations. Regular inspections by regulatory authorities ensure adherence.
- Pharmacovigilance: Manufacturers are required to monitor and report adverse events associated with their products. This ongoing safety surveillance is critical for maintaining regulatory approval.
- Intellectual Property: While primary composition of matter patents have expired, potential for litigation related to manufacturing processes, polymorphs, or new formulations could arise, though it is less common for mature generics.
- Controlled Substance Scheduling: Zaleplon is not scheduled as a controlled substance by the U.S. Drug Enforcement Administration (DEA), unlike some benzodiazepines. However, it is still subject to prescription requirements and careful monitoring by healthcare professionals due to its hypnotic effects and potential for misuse.
- Labeling and Advertising: Generic drug labeling must be consistent with the RLD's approved labeling. Advertising and promotional activities are subject to regulations concerning truthfulness, substantiation, and fair balance of risks and benefits.
Key Takeaways
- Generic zaleplon has a mature market characterized by intense price competition, with significant price reductions post-patent expiration.
- The demand for zaleplon is sustained by the high prevalence of insomnia, but growth is tempered by a crowded therapeutic landscape and the rise of non-pharmacological treatments.
- Prices are expected to remain stable or decline marginally in the short to medium term due to established generic competition.
- Manufacturing efficiency, API sourcing reliability, and stringent quality control are critical for generic producers to maintain market share and profitability.
- Regulatory compliance with GMP and pharmacovigilance remains paramount for all manufacturers.
Frequently Asked Questions
-
What is the average price range for a 30-day supply of generic zaleplon? The average wholesale price (AWP) for a 30-count bottle of generic zaleplon 10 mg capsules typically ranges from $20 to $60, varying by manufacturer and pharmacy.
-
Are there any significant patent expiries anticipated for zaleplon that could impact its market? No, the primary composition of matter and formulation patents for zaleplon have long since expired, leading to the current generic market.
-
What is zaleplon's primary competitive advantage over other non-benzodiazepine hypnotics like zolpidem? Zaleplon's advantage lies in its shorter half-life, which may lead to less residual daytime sedation for some patients, and its specific indication for sleep-onset insomnia.
-
What are the key risks associated with zaleplon use that might limit its market growth? Risks include potential for dependence, tolerance, misuse, and cognitive and behavioral side effects, although generally considered less severe than with benzodiazepines.
-
What is the projected growth rate for the overall prescription hypnotic market, and how does zaleplon fit into this forecast? The global prescription hypnotic market is projected to grow at a CAGR of approximately 4.5%. Zaleplon's share is expected to remain relatively stable within this growing market, facing competition from both established generics and potentially newer therapeutic options.
Citations
[1] National Institutes of Health. (2021). Insomnia. National Heart, Lung, and Blood Institute. Retrieved from https://www.nhlbi.nih.gov/health/insomnia
[2] GoodRx. (n.d.). Zaleplon prices, coupons, and patient assistance programs. Retrieved from https://www.goodrx.com/zaleplon (Note: Prices are dynamic and subject to change).
[3] Grand View Research. (2023). Insomnia drugs market size, share & trends analysis report by drug class (hypnotics & sedatives, Melatonin receptor agonists, others), by indication (sleep-onset insomnia, sleep-maintenance insomnia), by region, and segment forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/insomnia-drugs-market
More… ↓
