Share This Page
Drug Price Trends for VITAMIN D2
✉ Email this page to a colleague
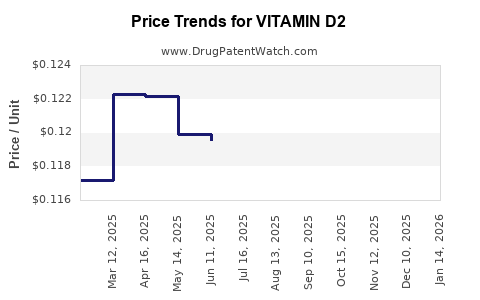
Average Pharmacy Cost for VITAMIN D2
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VITAMIN D2 1.25 MG(50,000 UNIT) | 42806-0547-05 | 0.11551 | EACH | 2026-03-18 |
| VITAMIN D2 1.25 MG(50,000 UNIT) | 42806-0547-01 | 0.11551 | EACH | 2026-03-18 |
| VITAMIN D2 1.25 MG(50,000 UNIT) | 69452-0151-20 | 0.11551 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview and Price Projections for Vitamin D2 (Ergocalciferol)
Vitamin D2 (ergocalciferol) remains a key form of vitamin D used primarily in prescription formulations and dietary supplements for addressing vitamin D deficiency. Its market dynamics are influenced by regulatory factors, healthcare trends, manufacturing capacities, and competition from vitamin D3 (cholecalciferol).
Market Size and Trends
Global Market Valuation (2022): Estimated at approximately USD 1.2 billion, with a compound annual growth rate (CAGR) of 4-5% projected through 2027.
Regional Breakdown:
- North America accounts for roughly 45% of the market, driven by high supplement consumption and a focus on deficiency management.
- Europe contributes around 30%, with increasing prescriptions and supplement use.
- Asia-Pacific shows rapid growth, driven by rising health awareness and supplement markets, expected to reach a CAGR of 6% over the next five years.
Key Factors:
- Rising prevalence of vitamin D deficiency globally, especially among elderly populations and individuals with limited sun exposure.
- Increasing use of vitamin D2 in pharmaceutical formulations for deficiency treatment.
- Growing adoption of supplements in developed regions, though prescription products still dominate due to regulatory standards.
Competitive Landscape
Major players include:
- Abbott Laboratories/AbbVie
- Fresenius Kabi
- Mylan (now part of Viatris)
- Zhejiang Tianyue (China)
- Teva Pharmaceuticals
Vitamin D2 is available as prescription medications, over-the-counter supplements, and functional foods. While D2 has historically held market share dominance in certain regions due to regulatory policies, D3 has gained prominence owing to its superior bioavailability.
Regulatory Environment
- In the US, the FDA approves vitamin D2 for prescription use but classifies it as a drug.
- EU regulations endorse vitamin D2 for pharmaceutical production, with some countries emphasizing D3’s advantages.
- In many countries, vitamin D2 is categorized as a dietary supplement ingredient, subject to manufacturing standards.
Price Dynamics and Projections
Current Price Range (2023):
- Prescription formulations: USD 0.15 - 0.30 per 1,000 IU dose.
- Over-the-counter supplements: USD 5 - USD 15 per bottle (around 100-200 capsules).
- Bulk manufacturing: Approximately USD 0.10 - USD 0.20 per gram of active ingredient.
Factors Affecting Price:
- Raw material costs linked to ergosterol sources (fungi) or chemical synthesis.
- Regulatory compliance and certification expenses.
- Market competition, especially from D3 products under patent or exclusivity protections.
Price Projections (2024-2027):
- Slight decline expected in product prices due to increased manufacturing efficiency and market competition, especially as more manufacturers enter the supplement space.
- Prescription product prices may remain stable or slightly increase, driven by healthcare cost inflation and stricter regulation.
- Average price decreases projected by 10-15% for over-the-counter formats.
Comparison with Vitamin D3
- Market share of D3 is increasing due to its higher bioavailability (up to 50% more effective).
- D3 dominates both prescription and supplement markets, comprising over 70% of total vitamin D consumption by volume as of 2022.
- D2 remains prevalent in specific markets, especially where regulatory frameworks favor its use or where D3 formulations face patent restrictions.
Market Entry and Investment Opportunities
- Manufacturing efficiencies and bioavailability enhancements for D2 could carve niche markets, especially in countries with regulatory favors.
- Innovation in delivery formats, such as sustained-release capsules or stability-enhanced formulations, could increase competitiveness.
- Developing combined formulations (e.g., vitamin D + calcium or other micronutrients) presents growth opportunities.
Key Takeaways
- The global vitamin D2 market was valued at approximately USD 1.2 billion in 2022, with moderate but steady growth projections.
- Price stability persists in prescription formulations, whereas over-the-counter supplement prices are trending downward due to increased competition and manufacturing efficiencies.
- D3 is expanding its market share, but D2 retains relevance in certain regions and therapeutic niches.
- Regulatory trends favoring D2 in some jurisdictions could influence future pricing and market share.
- Investment in manufacturing and formulation innovation can provide strategic advantages in niche segments.
FAQs
1. What are the primary drivers for growth in the vitamin D2 market?
Increasing prevalence of deficiency, expanding supplement use, and regulatory acceptance in certain regions.
2. How does the bioavailability of vitamin D2 compare to vitamin D3?
Vitamin D3 has approximately 1.5 to 2 times higher bioavailability than D2, influencing market preference.
3. What regulatory challenges affect vitamin D2 pricing?
Variations in approval pathways, quality standards, and classification as a drug or supplement impact manufacturing costs and pricing.
4. Are there any upcoming patent expirations or regulatory changes that could affect prices?
Most formulations are off patent, but regional regulatory shifts may influence cost dynamics and availability.
5. Which regions offer the most growth potential for vitamin D2?
Asia-Pacific and emerging markets with rising health awareness and supplement consumption.
References
- MarketWatch. "Vitamin D Market Size, Share & Trends Analysis." 2022.
- Grand View Research. "Vitamin D Market Analysis," 2023.
- US FDA. "Regulatory Details for Ergocalciferol," 2022.
- European Medicines Agency. "Vitamin D2 Use and Regulation," 2022.
- Statista. "Global Vitamin Supplement Market Forecast," 2023.
More… ↓
