Share This Page
Drug Price Trends for VARDENAFIL HCL
✉ Email this page to a colleague
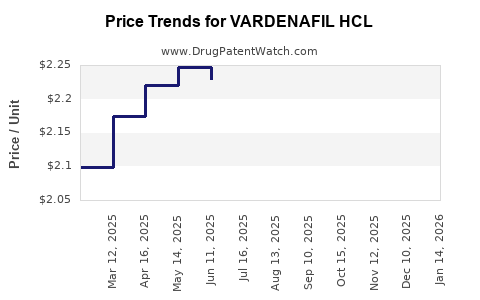
Average Pharmacy Cost for VARDENAFIL HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VARDENAFIL HCL 10 MG TABLET | 00093-7654-56 | 2.22549 | EACH | 2026-03-18 |
| VARDENAFIL HCL 10 MG TABLET | 00527-2802-32 | 2.22549 | EACH | 2026-03-18 |
| VARDENAFIL HCL 10 MG ODT | 62332-0235-04 | 20.89910 | EACH | 2026-03-18 |
| VARDENAFIL HCL 5 MG TABLET | 70710-1069-03 | 7.09980 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for VARDENAFIL HCL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| VARDENAFIL HCL 5MG TAB | Golden State Medical Supply, Inc. | 70710-1069-03 | 30 | 528.59 | 17.61967 | EACH | 2023-06-16 - 2028-06-14 | FSS |
| VARDENAFIL HCL 5MG TAB | Golden State Medical Supply, Inc. | 70710-1069-03 | 30 | 494.76 | 16.49200 | EACH | 2024-01-03 - 2028-06-14 | FSS |
| VARDENAFIL HCL 10MG TAB | Golden State Medical Supply, Inc. | 70710-1070-03 | 30 | 504.98 | 16.83267 | EACH | 2023-06-16 - 2028-06-14 | FSS |
| VARDENAFIL HCL 5MG TAB | AvKare, LLC | 00527-2801-32 | 30 | 381.60 | 12.72000 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Vardenafil Hydrochloride Market Analysis and Price Projections
Vardenafil hydrochloride, a phosphodiesterase type 5 (PDE5) inhibitor, is a pharmaceutical compound primarily used for the treatment of erectile dysfunction (ED). Its market performance is intrinsically linked to the prevalence of ED, evolving treatment paradigms, and patent exclusivity. This analysis details current market dynamics, competitive landscape, and projected pricing trends for vardenafil hydrochloride.
What is the Current Market Landscape for Vardenafil Hydrochloride?
The global market for vardenafil hydrochloride is mature, characterized by established competition from both branded and generic manufacturers. The primary indication, erectile dysfunction, affects a significant and growing patient population, particularly with aging demographics worldwide.
Market Size and Growth Drivers:
- Prevalence of ED: The incidence of erectile dysfunction is substantial, affecting an estimated 30-50% of men aged 40-70 years [1]. This demographic trend supports a consistent demand for ED treatments.
- Aging Population: As the global population ages, the prevalence of conditions that contribute to ED, such as cardiovascular disease, diabetes, and obesity, increases, thereby expanding the potential patient pool.
- Increased Awareness and Diagnosis: Greater public awareness and reduced stigma surrounding ED have led to more men seeking medical consultation and treatment.
- Generic Competition: The expiration of primary patents has opened the market to numerous generic versions of vardenafil hydrochloride, intensifying price competition and increasing accessibility for patients.
Key Market Segments:
- Oral Formulations: The dominant segment comprises oral tablets, with various dosages (e.g., 2.5 mg, 5 mg, 10 mg, 20 mg) catering to different patient needs and medical advice.
- Geographic Regions: North America and Europe represent the largest markets due to higher disposable incomes, advanced healthcare infrastructure, and higher reported prevalence rates. The Asia-Pacific region is experiencing significant growth due to increasing healthcare expenditure and a rising middle class.
Challenges and Restraints:
- Intense Generic Competition: The presence of multiple generic manufacturers drives down prices, impacting profit margins for both brand-name and generic producers.
- Competition from Other PDE5 Inhibitors: Vardenafil faces direct competition from other PDE5 inhibitors like sildenafil (Viagra), tadalafil (Cialis), and avanafil (Stendra), each with its own pharmacokinetic profiles, efficacy, and safety considerations.
- Side Effects and Contraindications: Like all PDE5 inhibitors, vardenafil has potential side effects (e.g., headache, flushing, nasal congestion) and contraindications (e.g., use with nitrates), which can limit its use in certain patient populations.
- Off-Label Use and Counterfeit Products: The market is affected by the availability of counterfeit products and off-label sales through unregulated channels, posing risks to patient safety and market integrity.
What is the Competitive Landscape for Vardenafil Hydrochloride?
The competitive landscape for vardenafil hydrochloride is characterized by a blend of originator brands, generic manufacturers, and the overarching competition from other PDE5 inhibitors.
Major Players (Generic Market):
- Teva Pharmaceutical Industries: A leading global generic pharmaceutical company with a significant portfolio, including vardenafil hydrochloride.
- Dr. Reddy's Laboratories: An Indian multinational pharmaceutical company that manufactures and markets generic vardenafil.
- Sun Pharmaceutical Industries: Another major Indian pharmaceutical company with a broad range of generic drugs.
- Mylan N.V. (now part of Viatris): A global pharmaceutical company known for its extensive generic drug offerings.
- Cipla Limited: An Indian multinational pharmaceutical company that has a presence in the ED market.
Originator Brand:
- Levitra (Bayer AG): The original branded vardenafil hydrochloride medication. While its patent protection has largely expired in major markets, the brand still holds recognition and a market share, particularly through authorized generic versions and brand loyalty.
Key Competitive Strategies:
- Cost Leadership: Generic manufacturers primarily compete on price, aiming to offer the lowest cost alternative to patients and healthcare systems.
- Market Penetration: Companies focus on securing broad distribution networks and obtaining regulatory approvals in key markets to maximize sales volume.
- Product Differentiation (Limited for Generics): While core vardenafil hydrochloride products are largely undifferentiated, some companies may offer varied pack sizes or combination therapies in specific markets.
- Supply Chain Efficiency: Streamlining manufacturing and distribution processes is crucial for maintaining competitive pricing.
Comparative Position Against Other PDE5 Inhibitors:
- Sildenafil (Viagra): Generally considered the market leader due to its first-mover advantage and strong brand recognition. It has a well-established efficacy and safety profile.
- Tadalafil (Cialis): Differentiates itself with a longer duration of action (up to 36 hours), offering greater spontaneity for some users. This "weekend pill" positioning provides a distinct advantage.
- Avanafil (Stendra): A newer entrant, often marketed for its rapid onset of action and potentially improved side-effect profile for some individuals.
Vardenafil hydrochloride typically occupies a mid-tier position in terms of market share and brand recognition among the PDE5 inhibitors, primarily due to the established dominance of sildenafil and the unique positioning of tadalafil. Its market share is largely driven by its availability as a cost-effective generic alternative.
What are the Patent Expirations and Regulatory Status?
The patent landscape for vardenafil hydrochloride has significantly impacted its market trajectory, transitioning it from a branded, high-margin product to a competitive generic market.
Key Patent Expirations:
- United States: The primary patents for Levitra (vardenafil hydrochloride) expired in the United States around 2014-2015 [2]. This opened the door for the introduction of multiple generic versions.
- Europe: Similar patent expirations occurred in major European countries around the same period, facilitating generic market entry.
- Other Global Markets: Patent expiries followed in other key markets globally, aligning with the typical 20-year patent life plus potential extensions.
Regulatory Approvals:
- Vardenafil hydrochloride is approved for the treatment of erectile dysfunction by regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- Generic versions require Abbreviated New Drug Applications (ANDAs) in the U.S. and similar regulatory pathways in other regions, demonstrating bioequivalence to the reference listed drug (Levitra).
Impact of Patent Expiration:
The expiration of the primary patents for vardenafil hydrochloride has had a profound effect:
- Increased Generic Availability: A surge in the number of generic manufacturers entering the market.
- Significant Price Erosion: The average selling price of vardenafil hydrochloride has decreased substantially due to intense competition among generic players.
- Market Share Shift: The market share has shifted away from the originator brand towards lower-cost generic alternatives.
- Focus on Cost-Effectiveness: Healthcare systems and payers increasingly favor generic options to manage costs.
Current Regulatory Standing:
Vardenafil hydrochloride remains an actively prescribed medication with ongoing regulatory oversight regarding manufacturing quality, safety monitoring, and marketing practices. Its status as a widely approved and available generic medication ensures continued market presence.
What are the Price Projections for Vardenafil Hydrochloride?
Price projections for vardenafil hydrochloride are heavily influenced by the dynamics of the generic pharmaceutical market, including manufacturing costs, competition levels, and market access.
Factors Influencing Future Pricing:
- Continued Generic Competition: The existing competitive landscape is unlikely to significantly abate. A stable or slightly increasing number of generic manufacturers will maintain downward pressure on prices.
- Manufacturing Efficiency and Raw Material Costs: Any fluctuations in the cost of active pharmaceutical ingredients (APIs), manufacturing overhead, or supply chain logistics could marginally impact wholesale prices. However, the scale of production for generic ED drugs typically mitigates significant cost volatility.
- Market Demand and Patient Adherence: While ED prevalence provides a baseline demand, shifts in prescribing patterns, patient adherence, or the emergence of novel ED treatments could influence volume and, indirectly, pricing.
- Payer and Pharmacy Benefit Manager (PBM) Negotiations: Major purchasers and PBMs will continue to leverage competition to secure the lowest possible prices through formulary placement and rebate structures.
- Geographic Market Variations: Pricing will continue to vary significantly by region, influenced by local regulatory environments, healthcare system reimbursement policies, and the competitive intensity within each specific market. Emerging markets may see initial higher prices for generics that stabilize as local competition grows.
Price Trend Projections:
- Short-Term (1-3 Years): Prices for vardenafil hydrochloride are expected to remain stable or decline marginally. The market is mature, and significant price jumps are improbable. Wholesale prices for generic vardenafil hydrochloride tablets are likely to remain in the range of $0.50 to $2.00 per tablet, depending on dosage, quantity, and supplier, a significant reduction from branded prices. Retail prices will reflect markups by distributors and pharmacies.
- Medium-Term (3-5 Years): Continued competition will sustain price stability. If any significant advancements in ED treatment emerge that displace PDE5 inhibitors broadly, it could exert more downward pressure. However, barring such a disruptive innovation, prices are anticipated to hold steady.
- Long-Term (5+ Years): The long-term outlook for vardenafil hydrochloride pricing will depend on the sustained demand for PDE5 inhibitors relative to newer therapeutic modalities. As a well-established, low-cost generic, it is likely to retain a significant market presence for the foreseeable future, with prices remaining at low, cost-competitive levels.
Comparative Pricing:
Vardenafil hydrochloride generics will continue to be priced competitively against generic sildenafil and tadalafil. Generally, all three generic PDE5 inhibitors are available at comparable low price points, making cost a less significant differentiating factor among them for many patients and payers. Pricing will be more a function of supplier negotiations and volume discounts.
Example Wholesale Price Range (Per Tablet):
| Dosage | Typical Range (USD) |
|---|---|
| 2.5 mg | $0.60 - $1.50 |
| 5 mg | $0.65 - $1.60 |
| 10 mg | $0.70 - $1.80 |
| 20 mg | $0.80 - $2.00 |
Note: These are indicative wholesale ranges and do not include pharmacy markups, dispenser fees, or specific contractual agreements. Actual prices will vary based on volume, supplier, and geographic market.
Key Takeaways
- The vardenafil hydrochloride market is mature and highly competitive, driven by the sustained prevalence of erectile dysfunction and widespread generic availability following patent expiries.
- Key market drivers include an aging global population, increased ED awareness, and the cost-effectiveness of generic options.
- Competition is primarily from other generic vardenafil manufacturers and, more broadly, from other PDE5 inhibitors like sildenafil, tadalafil, and avanafil.
- Significant price erosion has occurred since patent expiries, leading to a market where generic vardenafil hydrochloride is a low-cost treatment option.
- Price projections indicate continued stability or marginal declines, with wholesale prices per tablet expected to remain between $0.50 and $2.00, heavily influenced by ongoing generic competition and payer negotiations.
FAQs
-
What is the primary reason for the current low pricing of vardenafil hydrochloride? The primary reason is the widespread availability of generic versions following the expiration of key patents for the originator brand, Levitra. This has led to intense price competition among numerous manufacturers.
-
How does vardenafil hydrochloride's price compare to generic sildenafil and tadalafil? Generic vardenafil hydrochloride is generally priced comparably to generic sildenafil and tadalafil. All three are considered low-cost options in the PDE5 inhibitor class, with minor price variations often due to supplier-specific negotiations and volume discounts rather than inherent cost differences.
-
Are there any new patents or exclusivity periods expected for vardenafil hydrochloride that could affect pricing? Major patent expiries have already occurred. While companies may seek secondary patents for specific formulations or delivery methods, significant new exclusivity periods that would dramatically alter the generic pricing landscape are not broadly anticipated for the core vardenafil hydrochloride molecule.
-
What factors could lead to an increase in vardenafil hydrochloride prices in the future? A significant increase in price is unlikely. Potential, albeit minor, upward pressures could arise from substantial increases in API costs, supply chain disruptions affecting multiple manufacturers, or a significant consolidation of generic manufacturers that reduces overall competition.
-
How does the demand for vardenafil hydrochloride vary across different geographic regions, and how does this impact pricing? North America and Europe currently represent the largest markets due to high healthcare spending and prevalence awareness. The Asia-Pacific region shows strong growth potential. Pricing is generally lower in emerging markets as generics become established and competition intensifies, while mature markets maintain competitive but stable low-price points driven by payer formularies.
Citations
[1] Laumann, E. O., Glasser, D. B., Neves, J. B., & McHorney, C. A. (1999). Sexual function in the United States: impairment estimates from the national health and social life survey. JAMA, 281(6), 537-544.
[2] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
More… ↓
