Share This Page
Drug Price Trends for ULTRA LUBRICANT EYE DROPS
✉ Email this page to a colleague
Average Pharmacy Cost for ULTRA LUBRICANT EYE DROPS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
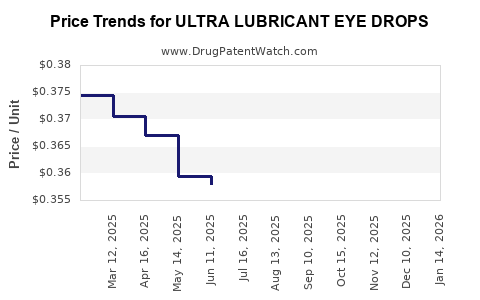
| ULTRA LUBRICANT EYE DROPS | 70000-0457-01 | 0.36205 | ML | 2026-03-18 |
| ULTRA LUBRICANT EYE DROPS | 70000-0457-01 | 0.36123 | ML | 2026-02-18 |
| ULTRA LUBRICANT EYE DROPS | 70000-0457-01 | 0.35997 | ML | 2026-01-21 |
| ULTRA LUBRICANT EYE DROPS | 70000-0457-01 | 0.35843 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ULTRA LUBICANT EYE DROPS
Market Overview
Ultra Lubricant Eye Drops are classified as artificial tears used to lubricate and soothe dry eyes. The global ophthalmic lubricants and dry eye treatments market has experienced steady growth driven by aging populations, increased screen time, and rising awareness of dry eye syndrome.
The global market size was valued at approximately USD 1.2 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2030, reaching an estimated USD 1.8 billion by 2030 [1].
Market Segments
-
Geography
- North America remains the largest market due to high awareness, well-developed healthcare infrastructure, and aging demographics.
- Europe follows, with similar drivers and an increasing prevalence of dry eye disease.
- Asia-Pacific exhibits the fastest growth, propelled by rising disposable incomes, urbanization, and expanding healthcare access.
-
Product Types
- Preservative-free eye drops are gaining market share over preserved varieties due to safety profiles.
- Multi-dose bottles and single-use vials are common delivery forms, with multi-dose products dominating due to convenience and cost advantages.
-
Distribution Channels
- Over-the-counter (OTC) sales account for roughly 65% of the market.
- Prescription sales comprise 35%, primarily for patients with severe dry eye conditions.
Competitive Landscape
Major players include Alcon, Bausch + Lomb, Johnson & Johnson, and Rohto. Private labels and generics are also increasing penetration, especially in emerging markets. Innovators are focusing on preservative-free formulations, better artificial tear compositions, and improved delivery mechanisms.
Pricing Trends and Projections
Current retail prices for a standard 10 mL bottle of Ultra Lubricant Eye Drops range between USD 10 and USD 15 in North America and Europe. Prices are influenced by:
- Formulation (preservative-free vs preserved)
- Packaging (multi-dose vs single-use)
- Brand reputation and marketing efforts
Price Evolution (2023-2030)
| Year | Average Price per 10 mL Bottle (USD) | Price Trend |
|---|---|---|
| 2023 | 12.50 | Base price, influenced by market competition |
| 2025 | 13.00 | Slight increase due to raw material cost inflation |
| 2027 | 13.50 | Entry of premium preservative-free formulations |
| 2030 | 14.00 | Stabilization, market saturation, and increased competition |
Projection assumptions include stable raw material costs, moderate inflation, and continued demand growth.
Pricing Drivers
- Regulatory approval and reimbursement mechanisms influence the positioning and pricing power.
- Manufacturing costs rise with stricter quality controls and preservative-free manufacturing requirements.
- Market penetration strategies by brands may involve competitive pricing, impacting average prices.
Regulatory and Market Entry Barriers
Stringent regulatory pathways in the US (FDA) and Europe (EMA) compare to accelerated registration pathways in emerging markets. Higher regulatory costs and compliance timelines can impact pricing strategies.
Future Market Dynamics
- There will be an increase in generic and private-label options, exerting downward pressure on prices.
- Innovation in formulations and packaging could command premium pricing.
- Consumer preferences for preservative-free products may increase average prices for the segment.
Key Takeaways
- The global ophthalmic lubricants market, including Ultra Lubricant Eye Drops, is projected to grow at a CAGR of 4.5% through 2030.
- Prices for standard 10 mL bottles hover around USD 12.50, with a gradual upward trend projected.
- Competition from generics and private labels could lead to price erosion, but innovations may sustain premium pricing.
- North America dominates the market, with Asia-Pacific's rapid growth offering price and volume expansion opportunities.
- Regulatory pathways and manufacturing complexities influence cost and pricing structures.
FAQs
-
What is the primary driver of demand for Ultra Lubricant Eye Drops?
Aging populations and increased screen time associated with digital device usage drive higher demand for dry eye treatments. -
How are regulatory changes affecting pricing?
Stricter approvals and compliance in established markets increase manufacturing costs, influencing prices upward; regulatory variability in emerging markets may impact pricing strategies differently. -
What factors could lead to price declines in the segment?
Entry of generic and private-label products, along with manufacturer cost reductions through process innovations, can lead to lower prices. -
What is the outlook for premium preservative-free formulations?
These formulations command higher prices due to safety perceptions, and their market share is expected to grow. -
How might market saturation impact prices by 2030?
Increased competition and market saturation could stabilize or reduce prices despite ongoing innovation.
References
- Market Research Future. "Ophthalmic Lubricants Market Size, Share & Trends Analysis Report." 2022.
More… ↓
