Share This Page
Drug Price Trends for TRIFLURIDINE
✉ Email this page to a colleague
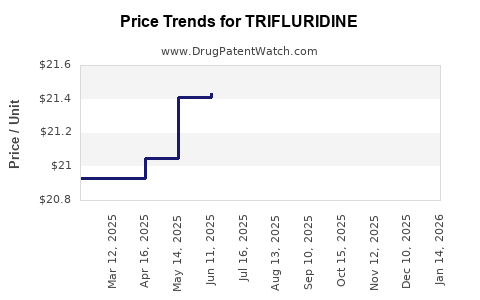
Average Pharmacy Cost for TRIFLURIDINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRIFLURIDINE 1% EYE DROPS | 61314-0044-75 | 23.49948 | ML | 2026-05-20 |
| TRIFLURIDINE 1% EYE DROPS | 61314-0044-75 | 22.83301 | ML | 2026-04-22 |
| TRIFLURIDINE 1% EYE DROPS | 61314-0044-75 | 22.19516 | ML | 2026-03-18 |
| TRIFLURIDINE 1% EYE DROPS | 61314-0044-75 | 21.96149 | ML | 2026-02-18 |
| TRIFLURIDINE 1% EYE DROPS | 61314-0044-75 | 21.59018 | ML | 2026-01-21 |
| TRIFLURIDINE 1% EYE DROPS | 61314-0044-75 | 21.71267 | ML | 2025-12-17 |
| TRIFLURIDINE 1% EYE DROPS | 61314-0044-75 | 21.73680 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TRIFLURIDINE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TRIFLURIDINE 1% SOLN,OPH | Sandoz, Inc. | 61314-0044-75 | 7.5ML | 80.93 | 10.79067 | ML | 2023-08-15 - 2028-08-14 | FSS |
| LONSURF 20MG/8.19MG TAB | Taiho Oncology, Inc. | 64842-1020-01 | 20 | 3187.98 | 159.39900 | EACH | 2022-04-01 - 2027-03-31 | FSS |
| LONSURF 15MG/6.14MG TAB | Taiho Oncology, Inc. | 64842-1025-02 | 40 | 5165.23 | 129.13075 | EACH | 2023-01-01 - 2027-03-31 | FSS |
| LONSURF 20MG/8.19MG TAB | Taiho Oncology, Inc. | 64842-1020-03 | 60 | 10411.92 | 173.53200 | EACH | 2023-01-01 - 2027-03-31 | FSS |
| LONSURF 20MG/8.19MG TAB | Taiho Oncology, Inc. | 64842-1020-01 | 20 | 3449.40 | 172.47000 | EACH | 2023-01-01 - 2027-03-31 | FSS |
| LONSURF 15MG/6.14MG TAB | Taiho Oncology, Inc. | 64842-1025-02 | 40 | 5496.83 | 137.42075 | EACH | 2024-01-01 - 2027-03-31 | FSS |
| LONSURF 20MG/8.19MG TAB | Taiho Oncology, Inc. | 64842-1020-03 | 60 | 10913.21 | 181.88683 | EACH | 2024-01-01 - 2027-03-31 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
TRIFLURIDINE MARKET ANALYSIS AND PRICE PROJECTIONS
This report analyzes the market for trifluridine, a nucleoside analog antiviral agent. The analysis focuses on its current applications, patent landscape, regulatory status, and projected market growth, with specific attention to price trends in key therapeutic areas.
CURRENT MARKET LANDSCAPE FOR TRIFLURIDINE
What are the primary approved indications for trifluridine?
Trifluridine is primarily approved for the topical treatment of herpetic keratitis, an infection of the cornea caused by the herpes simplex virus (HSV). In this indication, it functions as a thymidine kinase substrate, leading to the incorporation of trifluridine triphosphate into viral DNA, thereby inhibiting viral replication. It is also formulated in combination with tipiracil for the treatment of metastatic colorectal cancer (mCRC). This combination, known as trifluridine/tipiracil (marketed as Lonsurf), is a nucleoside analog that inhibits DNA synthesis and function.
What is the patent status of trifluridine and its formulations?
The patent landscape for trifluridine is segmented. The original compound trifluridine is off-patent, with its foundational patents having expired. However, new patents exist for specific formulations, manufacturing processes, and combination therapies.
Key Patent Considerations:
- Trifluridine/Tipiracil (Lonsurf): The primary patents covering this combination therapy have historically been held by Taiho Pharmaceutical. These patents relate to the composition of matter for the combination, methods of use, and specific dosing regimens. For example, U.S. Patent 9,585,002, titled "Pharmaceutical Composition and Method for Treatment," covers a pharmaceutical composition comprising trifluridine and tipiracil. This patent was filed in 2012 and has an expected expiration date of 2032, subject to potential extensions. Similar patents exist in other major markets, such as Europe and Japan.
- Generics: The expiration of basic compound patents for trifluridine has opened the door for generic manufacturers. However, the patents covering the trifluridine/tipiracil combination remain a significant barrier to entry for generic competition for this specific indication.
- New Formulations and Indications: Research and development efforts continue, potentially leading to new patents for novel delivery systems (e.g., extended-release formulations, ophthalmic solutions with improved stability) or for new therapeutic applications of trifluridine.
What is the regulatory status of trifluridine in major markets?
Trifluridine has received regulatory approval in key pharmaceutical markets.
Regulatory Approvals:
- United States (FDA): Trifluridine ophthalmic solution (e.g., Viroptic) is approved for the treatment of herpetic keratitis. Trifluridine/tipiracil (Lonsurf) is approved for the treatment of patients with metastatic colorectal cancer who have been previously treated with fluoropyrimidine-, oxaliplatin- and irinotecan-based chemotherapy.
- European Union (EMA): Trifluridine ophthalmic solution is approved for herpetic keratitis. Trifluridine/tipiracil (Lonsurf) is approved for metastatic colorectal cancer.
- Japan (PMDA): Trifluridine ophthalmic solution is approved. Trifluridine/tipiracil (Lonsurf) is approved for metastatic colorectal cancer.
Regulatory pathways for new indications or formulations would require extensive clinical trials and submissions to relevant health authorities.
MARKET SEGMENTATION AND GROWTH DRIVERS
What is the market size and projected growth for trifluridine in ophthalmology?
The ophthalmic market for trifluridine is relatively mature. Its use is primarily for a specific indication (herpetic keratitis) where established treatment protocols exist.
Ophthalmic Market Dynamics:
- Market Size: The global market for trifluridine ophthalmic solutions is estimated to be in the low tens of millions of U.S. dollars annually. This segment is characterized by stable demand but limited growth potential due to the availability of alternative antiviral treatments and the specific nature of the indication.
- Growth Drivers:
- Prevalence of HSV Infections: The ongoing prevalence of HSV infections contributes to a steady demand.
- Limited New Entrants: High regulatory hurdles and the availability of effective, albeit sometimes more expensive, alternatives can limit new entrants and innovation in this specific ophthalmic segment.
- Challenges:
- Competition: Acyclovir and ganciclovir are established alternatives, with some also available in ophthalmic formulations.
- Resistance: Though less common, the potential for viral resistance to nucleoside analogs exists.
What is the market size and projected growth for trifluridine/tipiracil in oncology?
The market for trifluridine/tipiracil is significantly larger and exhibits higher growth potential, driven by its application in advanced metastatic colorectal cancer.
Oncology Market Dynamics (Trifluridine/Tipiracil):
- Market Size: The global market for trifluridine/tipiracil (Lonsurf) for mCRC is estimated to be in the range of several hundred million U.S. dollars annually.
- Growth Drivers:
- Unmet Needs in mCRC: Despite advances, metastatic colorectal cancer remains a significant challenge, with a substantial patient population requiring effective salvage therapies.
- Clinical Efficacy: Trifluridine/tipiracil has demonstrated survival benefits in heavily pre-treated patients, positioning it as a valuable treatment option.
- Expansion into New Indications: Ongoing clinical trials are exploring the efficacy of trifluridine/tipiracil in other solid tumors, such as gastric cancer and pancreatic cancer. Approval in these new indications would substantially expand its market reach. For instance, a Phase 3 study in unresectable advanced or recurrent gastric cancer (TAGS trial) demonstrated a significant improvement in overall survival for trifluridine/tipiracil plus placebo compared to placebo alone.
- Geographic Expansion: Continued market penetration in emerging economies is expected to contribute to growth.
- Challenges:
- Toxicity Profile: Like many chemotherapy agents, trifluridine/tipiracil has a notable toxicity profile, including myelosuppression and gastrointestinal disturbances, requiring careful patient management.
- Competition: The mCRC landscape is competitive, with other chemotherapeutic agents, targeted therapies, and immunotherapies vying for market share.
- Patent Expirations: While major patents for the combination are in place, the eventual expiration will open the market to generics, potentially impacting pricing and market share for the branded product.
PRICE PROJECTIONS AND FACTORS INFLUENCING PRICING
How are trifluridine prices projected to evolve in its current therapeutic areas?
Price projections for trifluridine vary significantly between its ophthalmic and oncological applications due to differing market dynamics, patent protections, and value propositions.
Ophthalmic Trifluridine Price Projections:
- Current Pricing: Branded trifluridine ophthalmic solutions are priced in the range of $50-$150 per 7.5 mL bottle, depending on the region and specific product. Generic versions are available at lower price points.
- Projection: The price for ophthalmic trifluridine is expected to remain relatively stable. Modest increases may occur due to general inflation and manufacturing costs. The presence of generic alternatives will likely cap any significant price hikes for the branded product. Growth in this segment is not expected to be driven by price increases but rather by volume.
Oncological Trifluridine/Tipiracil (Lonsurf) Price Projections:
- Current Pricing: Trifluridine/tipiracil is a high-cost specialty drug, reflecting its complex development, clinical trial investment, and value in treating advanced cancer. A 30-day supply can range from $6,000 to $10,000 or more, depending on the dosage and region.
- Projection:
- Near-Term (1-3 years): Prices are expected to remain robust, driven by ongoing demand and potential expansion into new indications. Taiho Pharmaceutical will likely maintain premium pricing.
- Mid-Term (3-7 years): As key patents for the trifluridine/tipiracil combination approach expiration (e.g., U.S. Patent 9,585,002 expiring in 2032), the market may see some downward pressure from authorized generics or biosimil-like competition, although a direct biosimilar pathway for small molecules is not standard. However, direct generic competition could emerge following patent challenges or expiration. This could lead to a price reduction of 20-40% for the originator product as it competes with generics.
- Long-Term (7+ years): Post-patent expiry, significant price erosion is anticipated as multiple generic manufacturers enter the market. This could lead to price reductions of 50-70% or more compared to current levels, aligning more closely with other generic chemotherapy agents.
What factors will influence the price of trifluridine in the future?
Several factors will influence the future pricing of trifluridine products.
Key Pricing Influencers:
- Patent Expiration and Generic Competition: This is the most significant factor for trifluridine/tipiracil. As patents expire, generic competition will inevitably drive prices down. The ophthalmic formulation of trifluridine is already subject to generic pricing.
- Regulatory Exclusivity: Orphan drug designations or exclusivities for new indications can provide periods of market exclusivity, allowing for premium pricing. If trifluridine/tipiracil gains approval for new cancer types, these exclusivities could temporarily bolster pricing.
- Clinical Value and Outcomes: Demonstrating superior clinical outcomes, improved quality of life, or reduced overall healthcare system costs associated with trifluridine therapies (particularly Lonsurf) will support higher pricing. However, payer scrutiny is increasing, demanding robust cost-effectiveness data.
- Manufacturing Costs and Supply Chain Stability: The complexity of synthesizing trifluridine and its combination partner, tipiracil, as well as the global supply chain, will impact manufacturing costs. Any disruptions or significant increases in raw material prices could influence final product pricing.
- Reimbursement Policies and Payer Negotiations: Healthcare payers (insurance companies, government bodies) play a crucial role. Their reimbursement policies, formulary decisions, and negotiation leverage can significantly influence drug prices and market access. Stringent cost-containment measures by payers can lead to price concessions from manufacturers.
- Competitive Landscape: The availability and pricing of alternative treatments in both ophthalmology and oncology will create pricing boundaries. For instance, in mCRC, the price of Lonsurf is benchmarked against other chemotherapy regimens and targeted therapies.
- Research and Development Investment: The level of ongoing investment in new formulations, delivery methods, or expanded indications for trifluridine will influence the perceived value and the manufacturer's ability to command premium pricing for novel innovations.
MARKET CHALLENGES AND OPPORTUNITIES
What are the key challenges facing trifluridine products?
The trifluridine market faces specific challenges across its applications.
Market Challenges:
- Generic Erosion for Off-Patent Formulations: For the standalone ophthalmic trifluridine, the primary challenge is competition from lower-cost generic alternatives, limiting profitability for originators.
- Competition in Oncology: The mCRC treatment landscape is highly competitive, with continuous innovation in targeted therapies and immunotherapies. Trifluridine/tipiracil must consistently demonstrate its value proposition against emerging standards of care.
- Toxicity Management: The adverse event profile of trifluridine/tipiracil requires significant resources for patient monitoring and supportive care, which can impact overall treatment costs and patient adherence.
- Patent Expiry and Generic Entry: The impending expiry of key patents for trifluridine/tipiracil will usher in a new era of generic competition, necessitating strategic planning for manufacturers to maintain market position.
- Regulatory Hurdles for New Indications: Gaining regulatory approval for new indications requires substantial investment in clinical trials, with no guarantee of success.
What are the significant opportunities for trifluridine?
Despite challenges, trifluridine presents notable opportunities.
Market Opportunities:
- Expansion into New Oncology Indications: Successful clinical development and regulatory approval for trifluridine/tipiracil in other solid tumors (e.g., gastric, pancreatic) represent the most significant growth opportunity, vastly expanding its patient population and market revenue.
- Combination Therapies in Oncology: Further research into combining trifluridine/tipiracil with other agents (e.g., targeted therapies, immunotherapies) could enhance its efficacy and create new treatment paradigms, potentially leading to new patentable formulations or uses.
- Geographic Market Penetration: Increasing access and uptake in emerging markets for both ophthalmic and oncological applications can drive volume growth.
- Life Cycle Management: Development of novel formulations (e.g., improved oral delivery, reduced toxicity profiles, extended-release ophthalmic solutions) could extend the product lifecycle and create new market segments.
- Strategic Partnerships and Licensing: Opportunities exist for collaborations between pharmaceutical companies for co-development, co-promotion, or licensing of trifluridine-based assets, particularly for new indications or global market access.
KEY TAKEAWAYS
Trifluridine's market is bifurcated. The ophthalmic segment, primarily for herpetic keratitis, is mature, stable, and subject to generic competition, with limited growth prospects and stable pricing. The oncological application, particularly the trifluridine/tipiracil combination (Lonsurf) for metastatic colorectal cancer, represents a significant and growing market. Its growth is fueled by unmet needs in advanced cancers and opportunities for expansion into new indications. However, the market for trifluridine/tipiracil is also characterized by high pricing, complex toxicity management, and the impending challenge of patent expiration, which will lead to significant price erosion upon generic entry. Future growth hinges on successful clinical development for new oncology indications and strategic management of patent cliffs.
FREQUENTLY ASKED QUESTIONS
When is the primary patent for trifluridine/tipiracil (Lonsurf) expected to expire in the United States?
The primary patent for the trifluridine/tipiracil combination, U.S. Patent 9,585,001, is expected to expire in 2032, subject to potential extensions.
Are there any approved generic versions of trifluridine ophthalmic solution?
Yes, generic versions of trifluridine ophthalmic solution are available in major markets, leading to price competition with the branded product.
What are the main therapeutic areas being investigated for trifluridine/tipiracil beyond colorectal cancer?
Key areas of investigation for trifluridine/tipiracil include gastric cancer and pancreatic cancer, with ongoing clinical trials to assess its efficacy in these indications.
How does the cost of trifluridine/tipiracil compare to other treatments for metastatic colorectal cancer?
Trifluridine/tipiracil is positioned as a salvage therapy for heavily pre-treated patients, and its cost is comparable to or higher than other advanced chemotherapeutic regimens used in this setting, reflecting its specialized application and clinical value.
What is the typical mechanism of action for trifluridine in treating herpetic keratitis?
In herpetic keratitis, trifluridine acts as a substrate for viral thymidine kinase, leading to its phosphorylation and incorporation into viral DNA, thereby disrupting DNA synthesis and inhibiting viral replication.
CITATIONS
[1] Taiho Pharmaceutical Co., Ltd. (n.d.). LONSURF® (trifluridine/tipiracil) tablets. Retrieved from https://www.lonsurf.com/ [2] U.S. Patent 9,585,001. (2017). Pharmaceutical composition and method for treatment. [3] European Medicines Agency. (n.d.). Lonsurf. Retrieved from https://www.ema.europa.eu/en/medicines/human/EPAR/lonsurf [4] Food and Drug Administration. (n.d.). Drug Search. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/drug-search [5] Bang, Y. J., et al. (2021). Trifluridine/Tipiracil in Fluoropyrimidine-Refractory Advanced Gastric Cancer: The Phase III TAGS Trial. Journal of Clinical Oncology, 39(14), 1529-1539.
More… ↓
