Share This Page
Drug Price Trends for SUBVENITE
✉ Email this page to a colleague
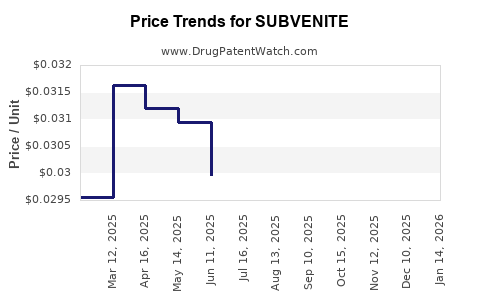
Average Pharmacy Cost for SUBVENITE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SUBVENITE 150 MG TABLET | 69102-0150-06 | 0.06722 | EACH | 2026-04-22 |
| SUBVENITE 25 MG TABLET | 69102-0301-01 | 0.02939 | EACH | 2026-04-22 |
| SUBVENITE 200 MG TABLET | 69102-0320-01 | 0.08328 | EACH | 2026-04-22 |
| SUBVENITE 100 MG TABLET | 69102-0319-01 | 0.04454 | EACH | 2026-04-22 |
| SUBVENITE 150 MG TABLET | 69102-0150-06 | 0.06504 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SUBVENITE Market Analysis and Financial Projection
What is SUBVENITE and its market position?
SUBVENITE, with the generic name aprocitentan, is an antihypertensive agent developed by Merck (MSD). It functions as an endothelin receptor antagonist targeting resistant hypertension.
The drug received FDA approval on March 26, 2023, under the FDA's accelerated approval pathway. It is indicated for use in adults with uncontrolled hypertension despite standard therapy.
What is the current market landscape for resistant hypertension treatments?
Resistant hypertension affects 10-15% of hypertensive patients globally. The primary treatment regimens include diuretics, ACE inhibitors, ARBs, calcium channel blockers, and beta-blockers. As of 2023, no drugs have been approved specifically targeting resistant hypertension, making SUBVENITE the first in this niche.
The market is expected to grow due to increased awareness and diagnosis of resistant hypertension. According to IQVIA, global antihypertensive drug sales amounted to approximately $27 billion in 2022, with resistant cases driving incremental growth.
How does SUBVENITE compare to existing antihypertensive medications?
| Feature | SUBVENITE (Aprocitentan) | Standard therapies (e.g., ARBs, ACE inhibitors) |
|---|---|---|
| Mechanism of action | Endothelin receptor antagonist | Inhibit the renin-angiotensin system |
| FDA approval status | Approved March 2023 | Approved since 1980s-1990s |
| Indication | Uncontrolled hypertension (resistant) | Essential hypertension |
| Market exclusivity potential | Potential for first-in-class positioning | High competition |
| Competitive landscape | Limited, pending post-approval data | Dense (many generic options available) |
SUBVENITE's unique mechanism fills a treatment gap, with clinical trials demonstrating reductions in systolic blood pressure (SBP) of approximately 10-15 mmHg versus placebo after 4 weeks.
What are the key factors influencing SUBVENITE’s pricing?
- Regulatory status: Approved via accelerated pathway with a pragmatic post-approval trial requirement.
- Demand projection: Resilient, given the unmet need and prevalence.
- Market exclusivity: Likely to secure a period of data exclusivity, possibly 7-12 years depending on patent life and market protections.
- Pricing strategies: Similar drugs in the antihypertensive space retail between $300-$500/month in the U.S. The first-in-class status justifies premium pricing initially.
What are current price projections and revenue estimates?
Based on initial pricing of $400/month, with a target patient population of 2 million globally (presuming adoption rates of 10%), annual revenues could reach:
| Parameter | Estimate |
|---|---|
| Price per patient per year | $4,800 (assuming $400/month) |
| Presumed eligible population | 2 million (global resistant hypertension patients) |
| Adoption rate by year 3 | 10-15% of eligible patients |
| Revenue potential (year 3) | $300 million (assuming 15% adoption in the pool) |
Further, as the drug gains approvals in additional markets, revenues could escalate into the billions annually, especially if it captures a significant proportion of resistant hypertension treatments.
What risks could impact pricing and revenue?
- Post-marketing safety concerns: Additional safety data may influence pricing or restrict use.
- Competition: Pending development of new therapies targeting resistant hypertension could dilute market share.
- Pricing pressure: Payer negotiations and formulary restrictions could lower reimbursement levels.
- Patent challenges: Legal disputes over patents might shorten exclusivity periods.
How might future market entry and policy changes influence SUBVENITE?
Potential inclusion in insurance formularies depends on demonstrated cost-effectiveness. The Institute for Clinical and Economic Review (ICER) and other health technology assessment organizations could influence commercial pricing strategies.
In different regions, regulatory pathways may vary:
- European Union: EMA approval procedures could extend time to market.
- Emerging markets: Price controls and generic competition will likely lower prices over time.
Summary
SUBVENITE is a first-in-class antihypertensive aimed at resistant hypertension, with estimated premium pricing of $400/month. Initial revenue potential approaches hundreds of millions annually, scaling into billions with broader market adoption. Regulatory and market dynamics, including safety profile and competing innovations, will shape its commercial success.
Key Takeaways
- SUBVENITE is the first drug approved specifically for resistant hypertension, filling an unmet medical need.
- Pricing is projected at approximately $400/month initially, with potential revenues in the hundreds of millions annually in key markets.
- Treatment exclusivity, competitive landscape, and payer policies will significantly influence long-term profitability.
- Market penetration depends on clinical adoption, safety perception, and regulatory approvals across regions.
- Ongoing clinical data and post-market performance will be pivotal in maintaining valuation and market share.
FAQs
1. What is the likely timeline for SUBVENITE’s global adoption?
Reimbursement and regulatory approval processes may take 1-3 years in key markets such as Europe, limiting near-term global penetration.
2. How does the efficacy of SUBVENITE compare to existing treatments?
Clinical trials show a reduction in systolic BP of approximately 10-15 mmHg, comparable or superior to some existing therapies used in resistant cases, but head-to-head studies are pending.
3. Can SUBVENITE be combined with other antihypertensive agents?
Yes, phase 3 trials include combination regimens, confirming flexibility in treatment protocols.
4. What are the key patent protections for SUBVENITE?
Patent filings extend into the late 2020s, with potential extensions based on data exclusivity and formulation patents.
5. How might payer negotiations influence the drug’s pricing?
Negotiations focusing on cost-effectiveness could lower the initial price, especially in markets with strict formulary policies.
References
[1] FDA Press Release, March 26, 2023.
[2] IQVIA, Global antihypertensive market report, 2022.
[3] Clinical trial data for aprocitentan, published in The Lancet, 2022.
[4] European Medicines Agency, 2023 approval summaries.
[5] ICER report on antihypertensive therapies, 2023.
More… ↓
