Share This Page
Drug Price Trends for SOFOSBUVIR-VELPATASVIR
✉ Email this page to a colleague
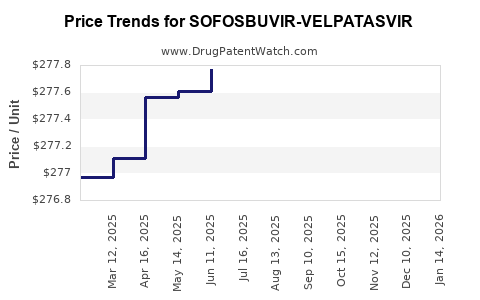
Average Pharmacy Cost for SOFOSBUVIR-VELPATASVIR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SOFOSBUVIR-VELPATASVIR 400-100 | 72626-2701-01 | 278.15922 | EACH | 2026-03-18 |
| SOFOSBUVIR-VELPATASVIR 400-100 | 72626-2701-01 | 278.09736 | EACH | 2026-02-18 |
| SOFOSBUVIR-VELPATASVIR 400-100 | 72626-2701-01 | 278.12293 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Sofosbuvir-Velpatasvir
What is the current market landscape for Sofosbuvir-Velpatasvir?
Sofosbuvir-Velpatasvir is a combination antiviral used to treat hepatitis C virus (HCV) infections across multiple genotypes. Approved by the FDA in 2016 under the brand name Epclusa by Gilead Sciences, the drug quickly gained market acceptance due to its broad-spectrum activity.
Market Size and Growth
The global hepatitis C drug market was valued at approximately $11 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 3.5% between 2023 and 2030, reaching nearly $15 billion.
Key Drivers:
- Increased diagnosis rates globally.
- Expanded access to direct-acting antivirals (DAAs).
- Improved treatment adherence and outcomes.
Regional Distribution:
- North America holds approximately 45% of the market.
- Europe accounts for nearly 25%.
- Asia-Pacific has seen the fastest growth, driven by increasing awareness and generic availability.
Competition Landscape
Main competitors include:
- Harvoni (Ledipasvir/Sofosbuvir)
- Mavyret (Glecaprevir/Pibrentasvir)
- Epclusa (Sofosbuvir-Velpatasvir)
- Vosevi (Sofosbuvir-Velpatasvir-Voxilaprevir)
Gilead maintains dominant market share through extensive clinical trials and broad national reimbursement coverage.
What are current pricing strategies?
Pricing in Major Markets
- United States: List price for a 12-week pack is approximately $74,760 (USD). Actual prices paid are typically lower post-negotiation and discounts.
- Europe: Prices range from €40,000 to €50,000 per treatment course, depending on the country and negotiated agreements.
- India: Generic versions priced at $300 to $900 for the full course.
Price Trends
- Prices declined by approximately 85% in the U.S. from 2015 to 2022 due to patent challenges and increased generic production.
- Several countries have negotiated value-based pricing models to improve affordability.
- Gilead's patent protections for Epclusa are scheduled to expire in key markets by 2030, opening the market for generics.
What are the key factors influencing future pricing?
Patent Expiry and Generic Competition
Patent expiration in major markets will facilitate generic entry, resulting in price erosion. For instance:
| Market | Patent Expiry Year | Estimated Generic Entry Year | Expected Price Reduction |
|---|---|---|---|
| United States | 2030 | 2032 | Up to 90% |
| European Union | 2030 | 2032 | Up to 85% |
| India | 2024 | Already present | 95% reduction |
Regulatory Policies
Countries adopting price caps or referencing international prices influence domestic pricing strategies.
Reimbursement Policies
Reimbursement coverage significantly impacts market penetration and pricing. Countries with national health systems tend to negotiate prices more aggressively.
Development Costs and Pharmaco-economic Value
The cost of research, clinical trials, and manufacturing influence baseline pricing. The high efficacy and simplicity of the therapy support premium pricing in some regions post-patent expiry.
What are the projections for the next five years?
| Year | Expected Market Size | Anticipated Price Range (USD) | Key Drivers |
|---|---|---|---|
| 2023 | ~$12 billion | $75,000 per course | Continued adoption, competition limited in some regions |
| 2024 | ~$12.2 billion | $70,000 – $80,000 | Patent expiry in India, generic entry in other markets |
| 2025 | ~$12.4 billion | $50,000 – $70,000 | Increased generic availability, price negotiations |
| 2026 | ~$12.7 billion | $30,000 – $60,000 | Market saturation, broad generic access |
| 2027 | ~$13 billion | $20,000 – $50,000 | Widespread generic penetration |
Summative insights
- The market for Sofosbuvir-Velpatasvir remains substantial but is vulnerable to price erosion due to generic competition, especially post-patent expiry.
- Gilead’s pricing strategies adapt to regional policies; overall prices are downward trending.
- Price reductions of up to 90% are expected over the next five years in mature markets.
- Generics and biosimilars will be critical players influencing the future price landscape.
Key Takeaways
- The hepatitis C antiviral market is mature, with heavy competition and significant generic entry expected after patent cliffs.
- Prices in developed countries have decreased sharply since introduction, with further reductions projected.
- Gilead’s patent protections in the U.S. and Europe extend until approximately 2030, delaying generic competition.
- Emerging markets exhibit much lower prices due to local generics and negotiated pricing models.
- Future pricing will balance patent protections, regulatory policies, and the cost-to-value ratio of the therapy.
FAQs
-
When will generic versions of Sofosbuvir-Velpatasvir be available in the U.S.?
Patent expiry is scheduled for 2030, with generic entry likely around 2032. -
How much can prices decrease after patent expiry?
Prices could decline by up to 90% in markets with low-cost generics and biosimilars. -
What factors influence drug pricing in developing countries?
Patent status, government negotiations, health policies, and local manufacturing capacity. -
Are there ongoing efforts to reduce treatment costs further?
Yes, countries are adopting value-based pricing, expanding generic access, and negotiating discounts. -
What is the market potential for Sofosbuvir-Velpatasvir beyond hepatitis C?
No significant alternative indications are currently under development.
References
[1] Gilead Sciences. (2022). Epclusa (sofosbuvir/velpatasvir) pricing and reimbursement details. Gilead.com.
[2] Global Data. (2023). Hepatitis C market analysis and forecast. GlobalData.com.
[3] IQVIA. (2022). The Global Use of Medicines in 2022. IQVIA Institute.
[4] World Health Organization. (2023). Hepatitis C Fact Sheet. WHO.int.
[5] European Medicines Agency. (2023). Patent expirations and generic approval timelines. EMA.europa.eu.
More… ↓
