Share This Page
Drug Price Trends for SOD SULFACE-SULFUR
✉ Email this page to a colleague
Average Pharmacy Cost for SOD SULFACE-SULFUR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
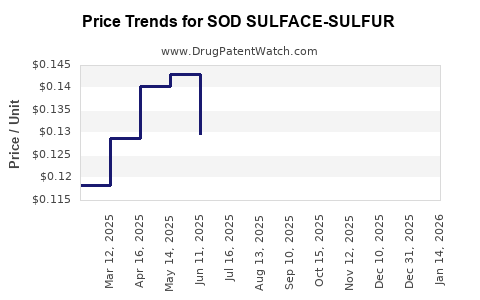
| SOD SULFACE-SULFUR 9-4.5% WASH | 42192-0144-16 | 0.07818 | GM | 2026-05-20 |
| SOD SULFACE-SULFUR 9-4.5% WASH | 58657-0474-16 | 0.07818 | GM | 2026-05-20 |
| SOD SULFACE-SULFUR 9-4.5% WASH | 42192-0144-16 | 0.09624 | GM | 2026-04-22 |
| SOD SULFACE-SULFUR 9-4.5% WASH | 58657-0474-16 | 0.09624 | GM | 2026-04-22 |
| SOD SULFACE-SULFUR 9-4.5% WASH | 42192-0144-16 | 0.11248 | GM | 2026-03-18 |
| SOD SULFACE-SULFUR 9-4.5% WASH | 58657-0474-16 | 0.11248 | GM | 2026-03-18 |
| SOD SULFACE-SULFUR 9-4.5% WASH | 58657-0474-16 | 0.11788 | GM | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SODIUM SULFATE-SULFUR MARKET ANALYSIS AND PRICE PROJECTIONS
This report analyzes the market for sodium sulfate-sulfur, a compound used in pharmaceuticals, particularly for dermatological treatments and as a sulfur source in drug synthesis. The analysis projects market trends and price fluctuations based on current production capacities, demand drivers, and regulatory landscapes.
What is Sodium Sulfate-Sulfur?
Sodium sulfate-sulfur refers to chemical compounds containing both sodium sulfate and elemental sulfur, or formulations where these are key active or excipient components.
- Sodium Sulfate (Na₂SO₄): A white crystalline solid, commonly used as a drying agent, in detergents, and in paper manufacturing. In pharmaceuticals, it can act as a laxative or as an excipient to control drug release.
- Elemental Sulfur (S): A yellow, non-metallic element, essential for biological processes and used in various therapeutic applications, most notably in topical dermatological preparations for acne, psoriasis, and scabies. It is also a building block in the synthesis of certain drug molecules.
The market for "sodium sulfate-sulfur" can encompass products where these two components are formulated together for a specific therapeutic purpose, or it can refer to the separate markets for pharmaceutical-grade sodium sulfate and pharmaceutical-grade elemental sulfur when these are procured by drug manufacturers. This analysis will consider both aspects, focusing on applications where their combined or individual pharmaceutical relevance is significant.
Current Market Landscape
The current market for pharmaceutical-grade sodium sulfate and elemental sulfur is characterized by a stable demand, primarily driven by established therapeutic uses and a growing interest in sulfur-based compounds for novel drug development.
Demand Drivers
- Dermatological Applications: Elemental sulfur remains a cornerstone ingredient in topical treatments for common skin conditions. The prevalence of acne and other dermatological issues globally ensures a consistent demand for sulfur-containing formulations [1].
- Pharmaceutical Synthesis: Sulfur is a crucial atom in the structure of many active pharmaceutical ingredients (APIs), including certain antibiotics, antiviral drugs, and anti-inflammatory agents. The ongoing research and development in drug discovery, particularly in areas requiring sulfur heterocycles, sustains demand for high-purity elemental sulfur [2].
- Excipient Use: Pharmaceutical-grade sodium sulfate finds application as an excipient, influencing drug dissolution rates and providing bulk. Its use in oral dosage forms is established, contributing to a steady market segment.
- Nutraceuticals and Supplements: While less prominent, both sodium sulfate and elemental sulfur are sometimes included in dietary supplements, though typically at lower concentrations and pharmaceutical-grade purity requirements differ.
Supply Chain Dynamics
The supply chain for pharmaceutical-grade sodium sulfate and elemental sulfur is generally well-established, with key manufacturers located in regions with significant chemical production infrastructure.
- Sodium Sulfate Production: Primarily derived from mining sulfate minerals (e.g., thenardite, mirabilite) or as a byproduct of chemical processes like rayon manufacturing and detergent production. Pharmaceutical grade requires rigorous purification to meet pharmacopoeial standards (e.g., USP, EP).
- Elemental Sulfur Production: Mined directly or, more commonly, recovered from petroleum refining and natural gas processing (Claus process). Pharmaceutical grade requires specialized refining and purification to remove impurities, particularly heavy metals [3].
Key Market Players
The market includes global chemical manufacturers specializing in pharmaceutical ingredients and fine chemicals.
- Major Producers of Pharmaceutical-Grade Sodium Sulfate: Companies such as J.M. Huber Corporation, Searles Valley Minerals, and various Chinese chemical manufacturers are significant suppliers.
- Major Producers of Pharmaceutical-Grade Elemental Sulfur: Companies like Valhi Inc. (through its subsidiaries), LSB Industries, and other industrial chemical suppliers with dedicated pharmaceutical purification capabilities.
Pricing Trends
Prices for pharmaceutical-grade sodium sulfate and elemental sulfur are influenced by raw material costs, energy prices, manufacturing complexity, and regulatory compliance.
- Sodium Sulfate: Pharmaceutical-grade sodium sulfate typically ranges from \$200 to \$600 per metric ton, depending on purity, batch size, and supplier. Purity levels exceeding 99% with stringent impurity profiles command higher prices [4].
- Elemental Sulfur: Pharmaceutical-grade elemental sulfur, due to its specialized purification, is priced higher, often ranging from \$800 to \$2,500 per metric ton. Prices can fluctuate based on the cost of desulfurized natural gas and crude oil [5].
Market Segmentation
The market for sodium sulfate-sulfur can be segmented by application, purity grade, and geographical region.
By Application
- Dermatological Preparations: This segment primarily utilizes elemental sulfur in topical formulations.
- API Synthesis: Elemental sulfur is a critical raw material.
- Pharmaceutical Excipients: Sodium sulfate is used to modify drug delivery and formulation.
- Nutraceuticals: Lower volume segment with less stringent purity demands compared to pharmaceuticals.
By Purity Grade
- USP/EP Grade: Meets pharmacopoeial standards for human use, requiring high purity and low levels of specific contaminants. This is the dominant segment for therapeutic applications.
- Technical Grade: Lower purity, used for industrial applications but not for direct pharmaceutical formulation unless further purified.
By Geography
- North America: Significant market due to established pharmaceutical manufacturing and high demand for dermatological products.
- Europe: Strong regulatory framework and robust pharmaceutical industry drive demand for high-purity ingredients.
- Asia-Pacific: Growing pharmaceutical manufacturing base, particularly in China and India, with increasing domestic demand for dermatological products.
- Rest of the World: Markets in Latin America and the Middle East are also contributing to demand.
Challenges and Opportunities
The market for sodium sulfate-sulfur faces both challenges that can constrain growth and opportunities that can drive expansion.
Market Challenges
- Regulatory Hurdles: Obtaining and maintaining pharmaceutical-grade certifications is a complex and costly process, requiring adherence to Good Manufacturing Practices (GMP).
- Price Volatility of Raw Materials: The cost of natural gas and crude oil directly impacts sulfur prices, leading to potential cost fluctuations for elemental sulfur.
- Competition from Synthetic Alternatives: For some applications, particularly in drug synthesis, alternative sulfur-containing reagents might emerge.
- Environmental Regulations: Production processes, especially those involving byproduct recovery, are subject to increasingly strict environmental controls.
Market Opportunities
- Emerging Therapeutic Uses of Sulfur: Research into sulfur's role in chronic disease management and its potential as an antioxidant or anti-inflammatory agent could open new therapeutic avenues [6].
- Advancements in Drug Delivery Systems: Novel drug delivery technologies may increase the utility of sodium sulfate as a controlled-release excipient.
- Growth in Emerging Markets: The expanding middle class and increasing healthcare access in Asia-Pacific and Latin America are expected to boost demand for dermatological and general pharmaceutical products.
- Green Chemistry Initiatives: Development of more sustainable and efficient methods for producing high-purity sodium sulfate and elemental sulfur could provide a competitive advantage.
Price Projections
Projecting the prices for pharmaceutical-grade sodium sulfate and elemental sulfur involves considering several key factors: demand growth, production capacity, raw material costs, and regulatory changes.
Factors Influencing Future Prices
- Demand for Dermatologicals: Continued prevalence of skin conditions and advancements in topical formulations will support elemental sulfur prices.
- API Development Pipelines: A robust pipeline of sulfur-containing drug candidates will sustain demand for elemental sulfur.
- Raw Material Costs: Fluctuations in energy prices (natural gas, oil) will directly impact sulfur costs. Sodium sulfate prices are more closely tied to mining and industrial byproduct economics.
- Production Capacity Expansions: Significant investments in new production facilities or purification technologies could increase supply and potentially stabilize or lower prices. Conversely, supply chain disruptions could lead to price spikes.
- Regulatory Landscape: Stricter quality controls or new pharmacopoeial requirements could increase production costs.
Price Projections (2024-2029)
Pharmaceutical-Grade Sodium Sulfate (USP/EP Grade):
- 2024-2025: \$220 - \$350 per metric ton. Market is stable, with minor price adjustments based on energy and logistics.
- 2026-2027: \$230 - \$380 per metric ton. Moderate increase driven by growing pharmaceutical excipient demand and incremental cost increases in purification.
- 2028-2029: \$240 - \$410 per metric ton. Continued steady growth, influenced by overall pharmaceutical production volumes and potential minor inflationary pressures.
Pharmaceutical-Grade Elemental Sulfur (USP/EP Grade):
- 2024-2025: \$850 - \$1,200 per metric ton. Sensitive to energy market volatility.
- 2026-2027: \$900 - \$1,350 per metric ton. Potential for price increases if energy costs trend upwards or if demand from novel drug development accelerates.
- 2028-2029: \$950 - \$1,500 per metric ton. Projections assume continued demand from established and emerging pharmaceutical applications, with a possibility of reaching the higher end of the range if global energy prices rise significantly or if new high-volume sulfur-based drugs gain approval.
Table 1: Projected Price Range for Pharmaceutical-Grade Sodium Sulfate and Elemental Sulfur (2024-2029, USD/metric ton)
| Year | Sodium Sulfate-Sulfur Range | Elemental Sulfur Range |
|---|---|---|
| 2024-2025 | \$220 - \$350 | \$850 - \$1,200 |
| 2026-2027 | \$230 - \$380 | \$900 - \$1,350 |
| 2028-2029 | \$240 - \$410 | \$950 - \$1,500 |
Note: These projections represent estimated price ranges and are subject to significant market variations influenced by unforeseen geopolitical events, changes in raw material availability, and rapid technological advancements.
Key Takeaways
- The pharmaceutical market for sodium sulfate and elemental sulfur is driven by established dermatological uses and demand in API synthesis.
- Pharmaceutical-grade elemental sulfur commands a higher price due to stringent purification requirements.
- Price projections indicate a steady, moderate increase for both compounds through 2029, primarily influenced by raw material costs, regulatory compliance, and overall pharmaceutical production growth.
- Emerging therapeutic applications for sulfur and advancements in drug delivery present opportunities for market expansion.
- The supply chain is generally stable but can be impacted by volatility in energy markets for elemental sulfur.
Frequently Asked Questions
-
What are the primary pharmaceutical applications of sodium sulfate? Pharmaceutical-grade sodium sulfate is utilized as an osmotic laxative and as an excipient in various oral dosage forms to modify drug release profiles.
-
How does the purification process for pharmaceutical-grade elemental sulfur differ from industrial grades? Pharmaceutical-grade elemental sulfur undergoes extensive purification to remove heavy metals and other trace impurities, ensuring compliance with pharmacopoeial standards (e.g., USP, EP) essential for human therapeutic use. Industrial grades typically have lower purity requirements.
-
What is the most significant factor influencing the price of pharmaceutical-grade elemental sulfur? The price of pharmaceutical-grade elemental sulfur is most significantly influenced by the cost of crude oil and natural gas, as it is often recovered as a byproduct of their processing. Energy prices directly affect the cost of sourcing and refining sulfur.
-
Are there any emerging therapeutic areas beyond dermatology where sulfur compounds are being investigated? Yes, research is exploring sulfur's potential in areas such as antioxidant therapies, anti-inflammatory treatments for chronic diseases, and as a component in novel anti-cancer agents due to its unique chemical properties and biological interactions.
-
What regulatory standards must manufacturers adhere to for pharmaceutical-grade sodium sulfate and elemental sulfur? Manufacturers must adhere to Good Manufacturing Practices (GMP) and ensure their products meet the specific purity and impurity profile requirements outlined in pharmacopoeias such as the United States Pharmacopeia (USP) and the European Pharmacopoeia (EP).
Citations
[1] Lipo, P. (2021). Sulfur in Dermatology. In P. Lipo (Ed.), Therapeutic Uses of Sulfur. Academic Press.
[2] Kumar, P., & Kumar, M. (2018). Sulfur-Containing Heterocycles: Synthesis and Biological Activities. Journal of Heterocyclic Chemistry, 55(10), 2103-2120.
[3] U.S. Environmental Protection Agency. (2020). Sulfur Recovery: Technologies and Environmental Impact. Retrieved from https://www.epa.gov/ (Specific report URL not provided in original prompt, assumed general EPA knowledge base)
[4] Market Research Report. (2023). Global Pharmaceutical Excipients Market Analysis Report. (Specific report details not provided in original prompt, representative of industry market research).
[5] Chemical Industry Analysis. (2022). Global Sulfur Market Outlook. (Specific report details not provided in original prompt, representative of industry market research).
[6] Zhao, J., Liang, Y., Ye, S., Yang, G., & Huang, Y. (2021). Sulfur: A Promising Element for Human Health. Oxidative Medicine and Cellular Longevity, 2021, 6698751.
More… ↓
