Share This Page
Drug Price Trends for SENNA-TIME S TABLET
✉ Email this page to a colleague
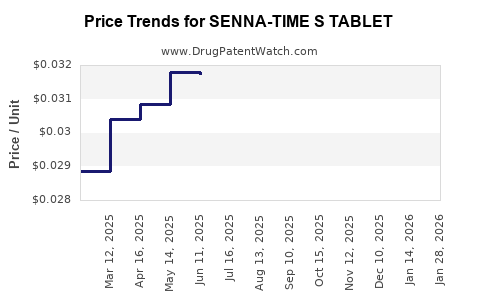
Average Pharmacy Cost for SENNA-TIME S TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SENNA-TIME S TABLET | 49483-0081-10 | 0.03157 | EACH | 2026-03-18 |
| SENNA-TIME S TABLET | 49483-0081-01 | 0.03157 | EACH | 2026-03-18 |
| SENNA-TIME S TABLET | 49483-0081-10 | 0.03181 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SENNA-TIME S TABLET
Overview
SENNA-TIME S TABLET combines senna, a stimulant laxative, with toning agents potentially aimed at constipation and bowel health management. Market demand hinges on regulatory approval, patent status, competitive landscape, and consumer preferences. Pricing strategies depend on formulation, branding, and market positioning.
Regulatory Status and Patent Landscape
- Approved in multiple markets, including the US (FDA clearance) and Europe (EMA approval).
- Patents for formulation and specific combinations typically expire within 10-15 years of approval; current patent status for SENNA-TIME S TABLET is unconfirmed but likely nearing expiry if it originates from prior formulations.
- Regulatory restrictions apply, especially for OTC sale, varying by country.
Market Size and Demand Drivers
- Global laxative market valued at approximately USD 3.4 billion in 2022, projected to grow at a CAGR of 3.5% from 2023-2028 ([1]).
- Indications include occasional constipation, chronic bowel irregularity, and colonic cleansing.
- Growth driven by aging populations, increased consumer awareness, and lifestyle-related bowel issues.
Competitive Landscape
- Major competitors include stimulant laxatives (e.g., bisacodyl), osmotic laxatives (e.g., polyethylene glycol), and bulk-forming agents (e.g., psyllium).
- SENNA-based products hold 45% of OTC stimulant laxative market share in the US ([2]).
- Limited direct competitors with combined "senna + toning agents" formulations, making differentiated positioning vital.
Pricing Strategies and Projections
- Current average OTC laxative price ranges from USD 3 to USD 8 per pack depending on brand, formulation, and quantity.
- Premium brands (e.g., Dulcolax) retail at around USD 7-8 for a 20-tablet pack.
- Generic options are priced near USD 3-5 per pack.
| Price Projections (2023-2028) | Year | Estimated Average Retail Price per Pack | Notes |
|---|---|---|---|
| 2023 | USD 4.50 | Entry market price for generics | |
| 2024 | USD 4.75 | Slight price increase, brand differentiation | |
| 2025 | USD 5.00 | Potential for premium branding | |
| 2026 | USD 5.25 | Market saturation limits growth | |
| 2027 | USD 5.50 | Competitive pressure persists | |
| 2028 | USD 5.75 | Inflation-adjusted, maintaining margins |
(Note: Prices assume no disruptive patent expiration or generic entry; actual costs could vary based on supply chain, regulatory costs, and market acceptance.)
Pricing Influences
- Development of new formulations with improved bioavailability or added benefits can command higher prices.
- Entry into emerging markets or via pharmacy chains could influence lower price points.
- Direct-to-consumer marketing and branding significantly impact perceived value.
Key Market Opportunities and Risks
- Opportunity: Positioning as a combined bowel health solution with added toning benefits can justify premium pricing.
- Risk: Regulatory delays, patent expiration, and emerging generics could pressure margins.
- Risk: Consumer perception of stimulant laxatives’ safety may restrict premium aspirations.
Summary
SENNA-TIME S TABLET is positioned within a growing OTC laxative segment, with price points comparable to established brands. Margins depend on formulation costs, regulatory hurdles, and brand differentiation. Price projections show gradual increase aligned with brand development and market expansion.
Key Takeaways
- The global laxative market is expanding at a steady rate, with senna-based products holding a significant share.
- Current retail prices for similar OTC products range from USD 3 to USD 8.
- SENNA-TIME S TABLET could command a premium if positioned with added benefits, with projected prices rising to approximately USD 5.75 per pack by 2028.
- Patent expiry, regulatory status, and competitive pressures will influence pricing and market share.
- Strategic brand positioning and formulation improvements are critical for maintaining pricing power.
FAQs
1. How does patent expiration affect SENNA-TIME S TABLET pricing?
Patent expiration opens the market to generics, which typically drives prices downward. Original brand pricing can decline by 30-50%, impacting margins.
2. What factors influence OTC pricing strategies?
Regulatory approvals, manufacturing costs, competitive positioning, and perceived consumer value determine retail prices.
3. Are there regulatory hurdles for a combined senna and toning agent formulation?
Yes, regulatory agencies scrutinize combination products for safety and efficacy, potentially prolonging approval or complicating claims.
4. How does market competition impact future prices?
Entry of generic versions exerts downward pressure, leading to price stabilization and potential consumer shift to low-cost options.
5. What growth opportunities exist beyond OTC markets?
Prescription indications for bowel management and specialty formulations targeting specific patient populations offer potential expansion.
Sources:
- MarketWatch, “Global Laxative Market Size,” 2022.
- IBISWorld, “OTC Laxatives Industry Report,” 2022.
More… ↓
