Share This Page
Drug Price Trends for SAXENDA
✉ Email this page to a colleague
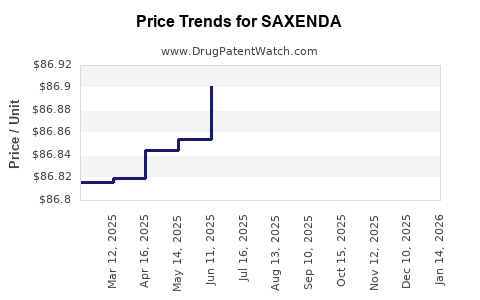
Average Pharmacy Cost for SAXENDA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SAXENDA 18 MG/3 ML PEN | 00169-2800-15 | 87.16106 | ML | 2026-03-18 |
| SAXENDA 18 MG/3 ML PEN | 00169-2800-15 | 87.16031 | ML | 2026-02-18 |
| SAXENDA 18 MG/3 ML PEN | 00169-2800-15 | 87.08319 | ML | 2026-01-21 |
| SAXENDA 18 MG/3 ML PEN | 00169-2800-15 | 87.06673 | ML | 2025-12-17 |
| SAXENDA 18 MG/3 ML PEN | 00169-2800-15 | 87.03391 | ML | 2025-11-19 |
| SAXENDA 18 MG/3 ML PEN | 00169-2800-15 | 86.98877 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SAXENDA (Setmelanotide)
What is SAXENDA and What is Its Approved Indication?
SAXENDA (Setmelanotide) is a melanocortin-4 receptor (MC4R) agonist developed by Rhythm Pharmaceuticals. It targets rare genetic disorders characterized by severe obesity due to appetite regulation deficiencies. The primary indication approved by the FDA in November 2020 is for the treatment of obesity due to POMC, PCSK1, or LEPR deficiency in patients age 6 years and older.
Market Size and Player Landscape
Rare Genetic Obesity Market Overview
The market for drugs targeting rare obesity conditions, including POMC deficiency, PCSK1 deficiency, and LEPR deficiency, is limited but projected to expand with increasing clinical recognition.
- Estimated patient population globally: 3,000 - 4,000 individuals for the specific genetic disorders eligible for SAXENDA.
- US patient base: approximately 700 - 1,200 patients based on prevalence data from Orphanet and published literature [1].
Competitors and Pipeline
SAXENDA currently possesses a unique mechanism suited for its indication, with no direct FDA-approved competitors. An investigational pipeline includes:
- Furipirilat (ALKS 7117): Preclinical stage, targeting MC4R pathway for broader obesity indications.
- Other MC4R agonists: In early phases, but none have received regulatory approval for genetic obesity.
Market Entry Factors
- Regulatory clarity on orphan drug designation facilitates market exclusivity.
- Limited competition reduces immediate pressure on pricing.
- Commercial success depends on the ability to identify, diagnose, and treat the genetically defined population.
Pricing History and Current Price
Approved Price in the US
- Approved list price (2022): approximately $150,000 annually per patient [2].
- The price reflects the rare disease status, high development costs, and small population size.
Insurance and Reimbursement
- Reimbursement frameworks are evolving, with payers requiring prior authorization.
- SMA (Spinal Muscular Atrophy) drugs like Zolgensma provide benchmarks for pricing negotiations with payers.
Price Projections and Market Dynamics
Short-Term Projections (2023-2025)
- Stable pricing: Set at $150,000 to $170,000 annually, considering inflation and market acceptance.
- Market growth: Expansion driven by increased diagnosis and approval of additional indications, including potential expansion into broader obesity populations and pediatric subsets.
Mid to Long-Term (2026-2030)
- Potential price adjustments: If competition emerges or additional indications are approved, prices could decline by 10-15%.
- Market penetration: Could reach 300-500 patients in the US by 2030 with wider recognition and genetic testing.
Key Factors Influencing Price Trajectory
- Payer negotiations: Cost-effectiveness assessments could pressure prices downward.
- Regulatory expansions: Approvals for broader obesity populations might dilute the price premium, leading to discounts.
- Manufacturing costs: Stable due to synthetic peptide manufacturing techniques.
Pricing Comparison with Similar Rare Disease Drugs
| Drug | Indication | Price (2022) | Year of Approval | Sales (2022) |
|---|---|---|---|---|
| Zolgensma | Spinal muscular atrophy | $2.1 million (per dose) | 2019 | ~$400 million |
| Soliris | Paroxysmal nocturnal hemoglobinuria, atypical hemolytic uremic syndrome | $500,000 annually | 2007 | ~$2.5 billion |
| Vimizim | Morquio A syndrome | ~$570,000 | 2014 | ~$300 million |
SAXENDA’s price is aligned with dosing and rarity, significantly lower than blockbuster therapies but comparable within the rare disorder market.
Market Development Considerations
- Expansion of indications: Potential extension into non-genetic general obesity based on emerging clinical data.
- Pricing adjustments: Tied to evidence of efficacy and expansion of the treated population.
- Health policy shifts: An increased emphasis on oral or biosimilar alternatives may pressure prices.
Key Market Drivers and Risks
Drivers
- Growing awareness and identification of rare genetic obesity disorders.
- Positive clinical trial outcomes supporting expanded indications.
- Payer willingness to reimburse for personalized, genetically targeted therapies.
Risks
- Price erosion if competitor drugs or generic alternatives enter the market.
- Limited patient population constrains revenue potential.
- Reimbursement hurdles in emerging markets.
Key Takeaways
- SAXENDA is a niche therapy with high per-patient pricing driven by orphan status.
- The current global market encompasses approximately 3,000-4,000 eligible patients.
- Annual treatment costs are approximately $150,000, with some variation.
- Long-term pricing depends on regulatory expansion, competition, and payer negotiations.
- The market has growth potential with increased awareness and diagnosis but remains limited by small patient populations.
FAQs
1. What factors most influence SAXENDA’s pricing?
Market size, regulatory exclusivity, manufacturing costs, and payer negotiation power.
2. How might expanded indications affect its market and price?
Broader indications could increase patient volumes, potentially diluting the price but also increasing total revenue.
3. Are there any biosimilar or generic alternatives?
No approved biosimilar or generic options exist; the small market makes biosimilar development unlikely in the near term.
4. How does SAXENDA compare to other rare disease therapies in pricing?
Its price is similar to other high-cost orphan drugs like Vimizin and Soliris, tailored to small patient populations with complex needs.
5. What is the outlook for SAXENDA’s market growth?
Modest, based on diagnosis rates; significant potential exists if broader obesity indications are approved, but the market remains limited.
References
[1] Orphanet. (2023). Data on genetic obesity disorders. Retrieved from https://www.orpha.net
[2] SSR Health. (2022). Estimated list prices and market data for rare disease drugs.
More… ↓
