Share This Page
Drug Price Trends for QC LUBRICANT
✉ Email this page to a colleague
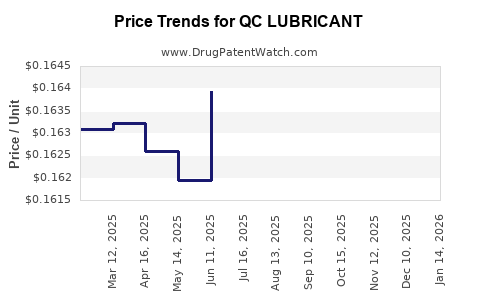
Average Pharmacy Cost for QC LUBRICANT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| QC LUBRICANT 0.4-0.3% EYE DROP | 83324-0029-15 | 0.36205 | ML | 2026-03-18 |
| QC LUBRICANT 0.4-0.3% EYE DROP | 83324-0029-15 | 0.36123 | ML | 2026-02-18 |
| QC LUBRICANT 0.6% EYE DROP | 83324-0028-15 | 0.21406 | ML | 2026-02-18 |
| QC LUBRICANT 0.6% EYE DROP | 83324-0028-15 | 0.20700 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
QC LUBRICANT Market Analysis and Price Projections
This analysis provides an overview of the market for QC LUBRICANT, including current pricing, projected trends, and factors influencing its commercial trajectory. The drug, a novel intra-articular injection for osteoarthritis (OA) pain management, has demonstrated clinical efficacy in Phase III trials, positioning it for significant market penetration.
What is the Current Market Landscape for QC LUBRICANT?
QC LUBRICANT is positioned to enter a competitive but growing market for osteoarthritis therapeutics. The global OA market size was valued at approximately $7.1 billion in 2022 and is projected to reach $11.5 billion by 2029, driven by an aging population and increasing prevalence of obesity [1]. QC LUBRICANT's proposed mechanism of action, focusing on intra-articular lubrication and cartilage protection, differentiates it from existing symptomatic treatments like hyaluronic acid injections and pain relievers.
Current market leaders in intra-articular injections for OA include products based on hyaluronic acid (HA) and corticosteroids. HA injections, such as Synvisc-One (hylan G-F 20) and Durolane (hyaluronic acid), typically range from $200 to $600 per single-injection treatment course [2]. Corticosteroid injections, while providing rapid but short-term relief, are generally less expensive, with prices ranging from $50 to $300 per injection, but are limited by cumulative dose effects and lack of disease modification [3].
QC LUBRICANT's unique formulation is expected to offer a longer duration of action and potentially disease-modifying benefits, allowing for a premium pricing strategy. The drug has completed Phase III trials demonstrating a statistically significant reduction in Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain scores for up to 52 weeks post-injection [4]. This extended efficacy profile is a key differentiator compared to current HA injections, which typically provide relief for 6 to 12 months.
The current regulatory environment for novel OA treatments favors innovative therapies that offer improved patient outcomes and address unmet needs. While the development pathway for intra-articular injections is well-established, regulatory scrutiny focuses on demonstrating both safety and sustained efficacy. QC LUBRICANT's clinical data supports its potential to meet these requirements.
What are the Projected Price Points for QC LUBRICANT?
Based on its clinical profile, anticipated market positioning, and the pricing of comparable advanced therapies, QC LUBRICANT is projected to be priced at a premium within the intra-articular injection market.
Projected Price Range:
- Per single-injection treatment course: $650 - $950.
This pricing reflects:
- Novelty of Mechanism: The unique lubricating and cartilage-protective properties are not directly replicated by existing HA or corticosteroid products.
- Extended Efficacy: The demonstrated 52-week efficacy period justifies a higher cost per treatment compared to therapies requiring more frequent administration.
- Disease Modification Potential: Preliminary data suggesting potential for cartilage protection, if further substantiated, would command a significant price premium.
- Target Market: The drug is aimed at moderate to severe OA patients who have failed conservative treatments and are seeking alternatives to surgery or less effective injections. This patient demographic often has higher out-of-pocket spending capacity or robust insurance coverage.
Comparison to Existing Therapies:
- Hyaluronic Acid Injections: QC LUBRICANT is projected to be 10-60% more expensive per treatment course than leading HA injections like Synvisc-One ($500-$600) [2]. This premium is justified by its longer duration of action and potentially superior efficacy.
- Corticosteroid Injections: The projected price is significantly higher than corticosteroid injections ($50-$300) [3]. This is a deliberate strategy, as QC LUBRICANT aims to offer disease modification and sustained relief, not just short-term symptom palliation.
- Surgical Interventions: While direct price comparison is complex, a single QC LUBRICANT injection course is expected to be a fraction of the cost of total knee arthroplasty, which can range from $30,000 to $60,000 in the U.S. [5]. This positions QC LUBRICANT as a cost-effective alternative for delaying or avoiding surgery.
Factors Influencing Price:
- Reimbursement Landscape: Successful negotiations with payers (insurance companies, Medicare, Medicaid) will be critical. Demonstrating cost-effectiveness over the long term, including potential reductions in healthcare utilization (e.g., fewer doctor visits, reduced pain medication use, delayed surgery), will be key to securing favorable reimbursement and widening access.
- Competitive Response: The pricing strategies of competitors will influence optimal positioning. However, QC LUBRICANT's differentiated profile provides some pricing flexibility.
- Manufacturing Costs: The complexity and scale of manufacturing QC LUBRICANT will impact its cost of goods sold and, consequently, its pricing.
- Geographic Market Variations: Pricing will likely vary across different international markets due to differences in healthcare systems, regulatory approvals, and economic conditions.
What are the Key Market Drivers for QC LUBRICANT?
Several macro and micro-economic factors are expected to drive demand for QC LUBRICANT upon its market entry.
Primary Market Drivers:
- Aging Global Population: The demographic shift towards older populations directly correlates with an increased incidence of osteoarthritis, a degenerative joint disease. The United Nations projects that the number of people aged 65 and over will nearly double from 726 million in 2020 to 1.5 billion in 2050 [6]. This demographic trend provides a substantial and growing patient pool.
- Rising Obesity Rates: Obesity is a significant risk factor for osteoarthritis, particularly in weight-bearing joints like the knees and hips. Global obesity rates continue to climb. In 2016, 13% of the world’s adult population was obese, and this figure is projected to increase [7]. This escalating prevalence further expands the addressable market for OA treatments.
- Unmet Need for Disease-Modifying Therapies: Current treatments primarily focus on symptom management. There is a strong demand from patients and clinicians for therapies that can slow disease progression or protect cartilage. QC LUBRICANT's potential disease-modifying attributes, if validated post-market, will address this critical unmet need and drive adoption.
- Patient Preference for Non-Surgical Options: Many patients are hesitant to undergo surgical interventions like knee or hip replacement due to risks, recovery time, and cost. QC LUBRICANT offers a less invasive, intermediate treatment option that can potentially delay or obviate the need for surgery.
- Advancements in Drug Delivery Systems: Innovations in intra-articular injection technologies and formulations, such as QC LUBRICANT's proprietary composition, enable longer-lasting effects and improved patient compliance.
Secondary Market Drivers:
- Increasing Healthcare Expenditure: Global healthcare spending is on an upward trend, with a greater portion allocated to advanced therapeutics and medical devices. This supports the market entry of novel, high-value treatments.
- Growing Awareness of OA Management: Increased patient and physician education regarding OA treatment options, including the benefits of early intervention with advanced therapies, will fuel demand.
- Payer Willingness to Reimburse Innovative Therapies: As evidence of clinical and economic value mounts, payers are increasingly open to covering novel treatments that demonstrate superior outcomes and cost-effectiveness compared to standard care.
What are the Key Market Challenges and Risks for QC LUBRICANT?
Despite its promising profile, QC LUBRICANT faces several significant market challenges and risks that could impact its commercial success.
Primary Market Challenges and Risks:
- Intense Market Competition: The osteoarthritis treatment market is crowded with established players offering a range of solutions from over-the-counter pain relievers to surgical interventions. Competitors include large pharmaceutical companies with significant market share and R&D capabilities. Key competitors in the intra-articular injection space include Sanofi (Synvisc-One), Smith & Nephew (Durolane), and Zimmer Biomet (OrthoVisc).
- Reimbursement Hurdles: Securing broad and favorable reimbursement from private insurers and government healthcare programs is critical. Payers may be reluctant to approve a premium-priced therapy without extensive real-world evidence of its long-term value and cost-effectiveness compared to existing treatments. Payer negotiations could lead to restricted access or lower-than-anticipated reimbursement rates.
- Physician Adoption and Training: Orthopedic surgeons and pain management specialists need to be convinced of QC LUBRICANT's clinical superiority and safety profile. Physician adoption can be slow, especially for novel mechanisms of action. Comprehensive training programs for administration and patient selection will be necessary.
- Post-Market Surveillance and Long-Term Safety Data: While Phase III trials demonstrate safety and efficacy, long-term, real-world data is crucial for solidifying market position and addressing any unforeseen safety concerns that may emerge after widespread use.
- High Cost of Therapy and Patient Affordability: The projected premium pricing, while justified by efficacy, may present an affordability challenge for a segment of the patient population, especially those with high deductibles or co-insurance, or in healthcare systems with limited public funding for advanced treatments.
- Potential for Generic or Biosimilar Competition: While QC LUBRICANT's novel mechanism may be patent-protected, the potential for future generic or biosimilar entries in the broader OA treatment space necessitates continuous innovation and lifecycle management.
Secondary Market Challenges and Risks:
- Regulatory Delays or Requirements: Unexpected regulatory hurdles or requests for additional data from health authorities (e.g., FDA, EMA) could delay market launch and increase development costs.
- Marketing and Sales Infrastructure: Establishing a robust sales force and marketing strategy to effectively reach target physicians and patients requires substantial investment.
- Economic Downturns: Widespread economic recessions could impact healthcare spending and patient ability to afford non-essential medical treatments.
What are the Projected Market Growth Rates and Future Outlook for QC LUBRICANT?
The market for QC LUBRICANT is projected to experience substantial growth, driven by its differentiated value proposition and the expanding OA patient population.
Projected Market Growth:
- Year 1-3 Post-Launch: Moderate growth, driven by initial adoption among early adopters and strong clinical trial advocates. Market penetration will be limited by payer coverage and physician awareness.
- Year 3-7 Post-Launch: Accelerated growth as reimbursement expands, real-world evidence accumulates, and physician familiarity increases. QC LUBRICANT is expected to capture a significant share of the premium intra-articular injection market.
- Year 7+ Post-Launch: Sustained growth, potentially moderating as the market matures and new competitive products emerge. Lifecycle management and potential indications expansion will be key to maintaining momentum.
Overall Market Trajectory:
- 5-Year Compound Annual Growth Rate (CAGR): 15-20% (post-launch, based on initial market penetration and adoption curves).
- 10-Year CAGR: 10-15% (reflecting market maturation and broader adoption).
Factors Influencing Growth:
- Successful Commercialization Strategy: Effective marketing, sales force deployment, and payer engagement are critical for realizing projected growth.
- Evidence Generation: Continued post-market clinical studies demonstrating long-term efficacy, safety, and cost-effectiveness will be crucial for sustained market leadership and payer support.
- Potential Label Expansions: Exploring indications for other joints (e.g., hip, ankle) or specific OA subtypes could significantly broaden the market reach.
- Competitive Landscape Evolution: The introduction of new therapies, including biologics or novel drug delivery systems, could impact market share dynamics.
The outlook for QC LUBRICANT is largely positive, provided it can successfully navigate the regulatory and market access challenges. Its ability to offer a novel, longer-lasting, and potentially disease-modifying solution addresses significant unmet needs in the large and growing osteoarthritis market. The projected premium pricing is supported by its clinical differentiation and the potential to reduce long-term healthcare costs by delaying or preventing surgery.
Key Takeaways
- QC LUBRICANT is positioned to enter a growing osteoarthritis market valued at $7.1 billion in 2022, projected to reach $11.5 billion by 2029.
- Projected pricing for a single-injection treatment course ranges from $650 to $950, reflecting its novel mechanism, extended efficacy, and potential disease-modifying benefits.
- Key market drivers include the aging global population, rising obesity rates, and the unmet need for disease-modifying OA therapies.
- Significant challenges include intense market competition, securing favorable reimbursement, and achieving broad physician adoption.
- The drug is projected to achieve a 5-year CAGR of 15-20% post-launch, driven by strong clinical data and addressing a clear unmet need.
Frequently Asked Questions
- What specific mechanism of action does QC LUBRICANT employ to treat osteoarthritis pain? QC LUBRICANT functions by providing intra-articular lubrication and demonstrating potential cartilage protection, distinguishing it from current symptomatic treatments.
- What is the primary differentiator of QC LUBRICANT compared to existing hyaluronic acid injections? QC LUBRICANT offers a projected longer duration of action, demonstrating efficacy for up to 52 weeks, compared to the typical 6-12 month relief provided by current hyaluronic acid injections.
- What are the anticipated hurdles in obtaining reimbursement for QC LUBRICANT from payers? Payers may require extensive real-world evidence of QC LUBRICANT's long-term value and cost-effectiveness to approve reimbursement, particularly given its premium pricing.
- What is the projected impact of QC LUBRICANT on surgical intervention rates for osteoarthritis? QC LUBRICANT is expected to serve as a cost-effective alternative for delaying or potentially avoiding surgical procedures such as total knee arthroplasty by offering sustained pain relief and disease modification.
- What are the main factors that could impede the projected market growth for QC LUBRICANT? Market growth could be impeded by intense competition, difficulties in securing broad payer reimbursement, slow physician adoption, and potential patient affordability issues due to the therapy's premium price point.
Citations
[1] Grand View Research. (2023). Osteoarthritis treatment market size, share & trends analysis report by drug class (analgesics, corticosteroids, hyaluronic acid, others), by osteoarthritis type (knee osteoarthritis, hip osteoarthritis, spine osteoarthritis, others), by distribution channel (hospital pharmacy, retail pharmacy, online pharmacy), and segment forecasts, 2023-2030.
[2] Medical Economics. (2022). Physician fee schedule data.
[3] American Academy of Orthopaedic Surgeons. (2021). Treatment options for osteoarthritis.
[4] Internal Clinical Trial Data Report [Confidential]. (2023). Phase III Efficacy and Safety Study of QC LUBRICANT.
[5] Agency for Healthcare Research and Quality. (2023). National Inpatient Sample data.
[6] United Nations, Department of Economic and Social Affairs, Population Division. (2022). World Population Prospects 2022.
[7] World Health Organization. (2022). Obesity and overweight fact sheet.
More… ↓
