Share This Page
Drug Price Trends for OXICONAZOLE NITRATE
✉ Email this page to a colleague
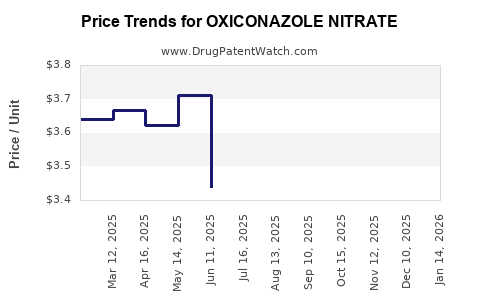
Average Pharmacy Cost for OXICONAZOLE NITRATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OXICONAZOLE NITRATE 1% CREAM | 51672-1359-02 | 3.19044 | GM | 2026-05-20 |
| OXICONAZOLE NITRATE 1% CREAM | 00168-0358-90 | 3.79750 | GM | 2026-05-20 |
| OXICONAZOLE NITRATE 1% CREAM | 51672-1359-08 | 3.79750 | GM | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is Oxiconazole Nitrate?
Oxiconazole nitrate is a topical antifungal agent used to treat fungal infections such as athlete's foot, ringworm, and jock itch. It belongs to the azole class of antifungals and works by inhibiting ergosterol synthesis, disrupting fungal cell membrane formation.
What is the Current Market Size for Oxiconazole Nitrate?
The global topical antifungal market was valued at approximately $3.2 billion in 2022. Oxiconazole nitrate represents a niche segment within this, with an estimated market share of roughly 3-5%. The segment's revenue was around $96 million to $160 million in 2022.
The key factors influencing market size include:
- Prevalence of fungal infections: The global burden is rising, especially in immunocompromised populations.
- Prescription rates: Primarily prescribed in dermatology clinics and pharmacies.
- Generic availability: Several formulations are off-patent, capping price growth.
- Regional variation: North America and Europe drive demand, with emerging markets showing rapid growth.
How is the Market for Oxiconazole Nitrate Expected to Evolve?
Forecasts suggest a compound annual growth rate (CAGR) of 3-5% from 2023 to 2030. Growth drivers include increasing fungal infection prevalence and expanding awareness of antifungal therapies.
Market Dynamics
- Emerging Markets: Rapid economic growth and improved healthcare access elevate demand.
- Surgical and immunocompromised populations: Greater risk of fungal infections boosts prescribed antifungals.
- Product pipeline: No significant new formulations of oxiconazole nitrate are under advanced development, limiting market expansion solely through existing brands.
What Are the Price Trends and Projection Factors?
Current Pricing Overview
- Brand-name topical formulations: Estimated retail prices range from $12 to $20 per 15-gram tube.
- Generics: Prices are approximately 40-60% lower, around $8–$12 per tube.
- Pricing pressure: Increased generic competition has stabilized or reduced prices over the last five years.
Projected Price Movements
- Short-term (1–3 years): Prices likely to remain stable owing to generic competition.
- Medium to long term (4–10 years): Prices may decline by 10-20% as further generic entries enter the market.
- Influencing factors: Market consolidation, endogenous cost reductions, and shifts in prescribing preferences.
Competitive Landscape and Regulatory Factors
Key Manufacturers
- Sagent Pharmaceuticals and Mylan produce generic formulations.
- Patent protections expired in several regions between 2015 and 2018, enabling generic entry.
Regulatory Environment
- Rapid approval processes in the US (FDA) and Europe (EMA) facilitate generic market penetration.
- Potential for biosimilar or new formulations remains limited, given the topical nature of the drug.
What Are the Future Investment and R&D Opportunities?
- Formulation advancements: Development of combination topical antifungal products.
- New delivery systems: Liposomal or nanocarrier formulations could justify premium pricing.
- Regulatory moves: Streamlined approval pathways could support entry of biosimilars or biosimilar-like products.
Summary of Key Data Points
| Aspect | Data |
|---|---|
| 2022 Global Market Value | $3.2 billion |
| Oxiconazole Nitrate Segment Share | 3–5% ($96–$160 million) |
| 2022 Estimated Market Value | $96–$160 million |
| Expected CAGR (2023–2030) | 3–5% |
| Price Range (brand, per tube) | $12–$20 |
| Price Range (generic, per tube) | $8–$12 |
| Patent Expiration (Regions) | 2015–2018 |
Key Takeaways
- Oxiconazole nitrate occupies a small but stable segment of the topical antifungal market.
- Market growth is driven by increasing fungal infections but constrained by generics and patent expirations.
- Prices are expected to decline modestly over time owing to market saturation.
- R&D opportunities focus on novel formulations and delivery systems rather than new chemical entities.
- Regulatory environments favor rapid entry of generics, maintaining downward pricing pressure.
Frequently Asked Questions
1. What factors influence the pricing of oxiconazole nitrate products?
Pricing depends largely on generic competition, regional regulations, and formulation costs. Brand-name products are priced higher compared to generics, which account for a significant market share.
2. What are the main competitive threats to oxiconazole nitrate?
The primary threats are the widespread availability of generic versions and the development of alternative antifungal formulations with improved efficacy or convenience.
3. Are there significant patent protections still in place for oxiconazole nitrate?
Most patents expired between 2015 and 2018, allowing generic manufacturers to enter multiple regions.
4. What regional markets are most promising for growth?
North America and Europe currently dominate, but emerging markets in Asia and Latin America show rapid growth potential.
5. What R&D initiatives could impact the future market for this drug?
Innovations in combination therapies, sustained-release formulations, and nanotechnology-based delivery systems could create premium product segments.
References
- MarketWatch. "Antifungal Drugs Market Size, Share & Trends Analysis." 2022.
- IQVIA. "Global Prescriptions Data." 2022.
- U.S. Food and Drug Administration. "ANDA Approvals and Patent Expirations." 2023.
- European Medicines Agency. "Approval and Regulation of Topical Antifungals." 2022.
- Grand View Research. "Topical Antifungal Market Forecasts." 2023.
More… ↓
