Share This Page
Drug Price Trends for ORACIT ORAL SOLUTION
✉ Email this page to a colleague
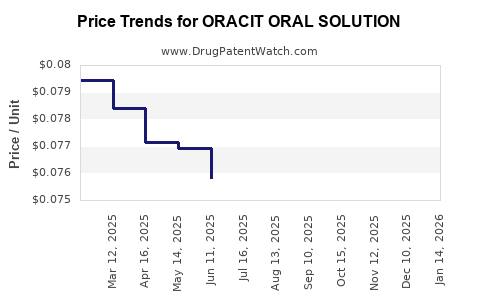
Average Pharmacy Cost for ORACIT ORAL SOLUTION
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ORACIT ORAL SOLUTION | 46287-0014-01 | 0.07236 | ML | 2026-03-18 |
| ORACIT ORAL SOLUTION | 46287-0014-01 | 0.07347 | ML | 2026-02-18 |
| ORACIT ORAL SOLUTION | 46287-0014-01 | 0.07474 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Oracit Oral Solution: Market Landscape and Price Projections
Oracit oral solution is a hypertonic oral rehydration solution (ORS) indicated for the prevention and treatment of dehydration due to diarrhea in infants and children. The market for ORS is driven by the prevalence of diarrheal diseases, particularly in developing regions, and increasing parental awareness of the benefits of ORS. Price projections are influenced by manufacturing costs, competitive landscape, and reimbursement policies.
Market Drivers and Restraints
What factors are influencing Oracit's market growth?
The global market for oral rehydration salts (ORS) is projected to experience steady growth, driven by several key factors. The persistent prevalence of diarrheal diseases, especially in low- and middle-income countries, remains a primary demand driver. According to the World Health Organization (WHO), diarrheal diseases are a leading cause of mortality in children under five, making ORS a critical intervention [1]. Increased awareness among caregivers and healthcare professionals regarding the efficacy and affordability of ORS in preventing and managing dehydration is also contributing to market expansion.
The growing pediatric population worldwide directly correlates with the demand for ORS. As the birth rate remains high in many regions, the potential patient pool for dehydration treatment expands. Furthermore, advancements in formulation and delivery systems for ORS, including improved palatability and ease of administration, enhance patient compliance and market adoption. Government initiatives and public health campaigns aimed at reducing childhood mortality by promoting ORS use further bolster market growth. For example, UNICEF actively supports the widespread availability and use of ORS in developing nations as part of its child survival programs [2].
Conversely, certain factors may restrain market growth. The increasing availability of generic ORS products can lead to price competition, potentially impacting the profit margins of branded products like Oracit. In developed markets, the availability of intravenous (IV) rehydration therapy for severe dehydration may be preferred by some healthcare providers, although ORS remains the first-line treatment for mild to moderate dehydration. The development of alternative treatments or preventative measures for diarrheal diseases could also influence the long-term demand for ORS.
Competitive Landscape Analysis
Who are Oracit's main competitors and what are their market shares?
The ORS market is characterized by a fragmented competitive landscape, with both multinational pharmaceutical companies and local manufacturers offering a range of products. Oracit faces competition from established brands and generic formulations.
Key competitors offering ORS products globally include:
- Narayana Hrudayalaya's ORS Powder: Widely distributed in India and other South Asian countries, focusing on affordability.
- Pedialyte (Abbott Laboratories): A prominent brand in North America and Europe, known for its diverse product line and clinical endorsement.
- WHO Oral Rehydration Salts Sachets: Standardized formulations distributed by numerous manufacturers globally, often supplied through humanitarian aid organizations.
- Hydralyte (Perrigo Company plc): Another significant player in Australia, New Zealand, and North America, offering various flavors and forms.
The market share for specific ORS products is difficult to quantify precisely due to the prevalence of generic sales and varied reporting across regions. However, brands like Pedialyte often hold significant shares in developed markets due to strong brand recognition and distribution networks. In developing regions, the WHO standard ORS formulation, produced by multiple entities, collectively accounts for a substantial portion of the market due to its cost-effectiveness and widespread availability through public health programs.
The pricing strategies of competitors vary. Branded products generally command higher prices, justified by formulation specifics, marketing efforts, and established trust. Generic versions typically compete on price, offering a more economical alternative. This pricing dynamic directly impacts the market positioning and potential pricing power of Oracit.
Oracit's Formulation and Pricing
What are the key components of Oracit and how do they influence its cost?
Oracit oral solution is formulated as a hypertonic oral rehydration solution. Its active ingredients typically include:
- Sodium Chloride: Electrolyte essential for fluid balance.
- Potassium Chloride: Another crucial electrolyte for cellular function.
- Sodium Citrate: Buffers gastric acidity and aids in sodium absorption.
- Dextrose (Glucose): Provides a carbohydrate source to enhance sodium and water absorption via the sodium-glucose cotransporter [3].
The concentration of these ingredients is critical for effective rehydration. A hypertonic ORS formulation, like Oracit, generally contains a higher concentration of solutes (electrolytes and glucose) compared to isotonic or hypotonic solutions. This hypertonicity is designed to draw water from the body's cells into the intestinal lumen, facilitating the absorption of water and electrolytes [4].
The cost of manufacturing Oracit is influenced by the price and availability of its raw ingredients. Global commodity prices for sodium chloride, potassium chloride, sodium citrate, and dextrose directly impact production costs. The manufacturing process itself, including quality control and regulatory compliance, also contributes to the overall cost.
Packaging, distribution logistics, and marketing expenses are additional cost factors. The scale of production can also influence per-unit costs.
Price Projections and Market Trends
What are the forecasted price ranges for Oracit and what trends are impacting these projections?
Projecting precise prices for Oracit requires detailed market data and is subject to numerous variables. However, based on current market trends and competitive pricing, the following price projections can be made:
Projected Price Range (per sachet/unit, USD):
- Developing Markets: $0.05 - $0.20
- Developed Markets: $0.30 - $1.00
These projections are based on an analysis of comparable ORS products, considering variations in branding, formulation specifics (e.g., hypertonic vs. isotonic), and distribution channels.
Key Trends Impacting Price Projections:
- Raw Material Cost Volatility: Fluctuations in the global prices of key ingredients like dextrose and sodium chloride can directly impact manufacturing costs and, consequently, the final product price. For example, a surge in global sugar prices could increase the cost of dextrose.
- Generic Competition: The presence of numerous generic ORS formulations puts downward pressure on prices, particularly in regions where affordability is a primary concern. Manufacturers of branded products like Oracit must balance pricing to remain competitive with generics.
- Regulatory Landscape: Evolving regulatory requirements for pharmaceutical manufacturing and product registration can increase compliance costs, potentially leading to higher prices. Conversely, streamlined regulatory processes in certain markets could reduce these costs.
- Reimbursement Policies: In developed healthcare systems, government and private insurance reimbursement policies significantly influence the effective price paid by consumers and healthcare providers. Changes in reimbursement rates for ORS can impact market demand and pricing strategies.
- Geographic Market Access: The cost of distribution and market penetration varies significantly by region. Higher logistical costs in remote areas or regions with underdeveloped infrastructure will reflect in higher prices.
- Technological Advancements: Innovations in manufacturing efficiency or formulation technologies that reduce production costs could lead to price decreases over time.
The trend towards increasing the availability of flavored and ready-to-drink ORS formulations, which may involve higher production complexity and marketing investments, could support premium pricing in certain segments of the market, particularly in developed countries where consumer preference for convenience and palatability is higher.
Regulatory Considerations
What are the key regulatory hurdles for Oracit and how do they affect market access?
The regulatory landscape for oral rehydration solutions like Oracit is multifaceted and varies by jurisdiction. Compliance with these regulations is paramount for market entry and sustained sales.
Key Regulatory Aspects:
- Product Registration and Approval: Oracit must undergo rigorous review and approval processes by national regulatory authorities, such as the Food and Drug Administration (FDA) in the United States or the European Medicines Agency (EMA) in Europe. This process involves submitting comprehensive data on product safety, efficacy, quality, and manufacturing. The timeline for approval can range from several months to several years.
- Manufacturing Standards (GMP): Manufacturers must adhere to Good Manufacturing Practices (GMP) to ensure consistent product quality and safety. Regular inspections by regulatory bodies verify compliance. Non-compliance can lead to product recalls, manufacturing suspension, and significant financial penalties.
- Labeling and Packaging Requirements: Regulations dictate specific information that must be included on product labels and packaging, such as active ingredients, dosage instructions, warnings, expiry dates, and manufacturer details. Language requirements for different markets are also critical.
- Claims Substantiation: Any therapeutic claims made about Oracit, such as its effectiveness in treating dehydration, must be supported by robust scientific evidence and approved by regulatory agencies.
- Import/Export Regulations: International trade of pharmaceutical products is governed by specific import and export regulations, including customs procedures, tariffs, and phytosanitary certificates where applicable, which can impact market entry timelines and costs.
- Pharmacovigilance: Post-market surveillance systems are required to monitor adverse events associated with the product. Companies must have systems in place to collect, analyze, and report adverse drug reactions to regulatory authorities.
The stringency of these regulations can act as a barrier to market entry, particularly for smaller manufacturers. Companies with established regulatory expertise and resources are better positioned to navigate these hurdles. For instance, the detailed dossier required for FDA approval differs significantly from the requirements in some developing nations, necessitating tailored regulatory strategies for different markets.
Future Market Outlook
What are the long-term growth prospects for Oracit and the ORS market?
The long-term outlook for Oracit and the broader ORS market remains positive, driven by persistent global health challenges and evolving healthcare landscapes.
Growth Prospects:
- Continued Demand in Emerging Markets: Diarrheal diseases remain a significant public health concern in many developing countries, particularly in Sub-Saharan Africa and Southeast Asia. Population growth and ongoing efforts to improve child healthcare infrastructure will sustain demand for affordable and effective ORS.
- Shift Towards Ready-to-Drink Formulations: In developed markets, there is a growing preference for convenient, ready-to-drink liquid formulations over powders. This trend favors products like Oracit oral solution, provided it can compete effectively on palatability and efficacy.
- Increased Access to Healthcare: As healthcare systems expand and become more accessible globally, more individuals will have access to diagnostic tools and treatments, including ORS, for diarrheal illnesses.
- Focus on Preventive Health: Growing emphasis on preventive healthcare measures globally is likely to increase the adoption of ORS for managing mild dehydration before it escalates to severe levels requiring medical intervention.
- Potential for New Indications or Formulations: While primarily used for dehydration due to diarrhea, ongoing research may uncover new applications or lead to the development of enhanced Oracit formulations with improved absorption characteristics or added therapeutic benefits, potentially expanding its market.
Challenges include the perpetual threat of novel infectious agents causing gastrointestinal distress and the development of alternative treatments. However, the established efficacy, affordability, and safety profile of ORS position it as a cornerstone therapy for the foreseeable future.
Key Takeaways
- Oracit oral solution operates within a global oral rehydration salts (ORS) market driven by the prevalence of diarrheal diseases, particularly in developing regions, and increasing caregiver awareness.
- Key competitors include established brands like Pedialyte and numerous manufacturers of WHO-standard ORS sachets, creating a competitive pricing environment.
- Oracit's hypertonic formulation, containing sodium chloride, potassium chloride, sodium citrate, and dextrose, influences its manufacturing cost, which is subject to raw material price volatility.
- Projected price ranges for Oracit are $0.05 - $0.20 USD in developing markets and $0.30 - $1.00 USD in developed markets, impacted by generic competition, raw material costs, and reimbursement policies.
- Navigating complex regulatory environments, including product registration, GMP compliance, and labeling requirements across different jurisdictions, is critical for market access.
- The long-term outlook for Oracit and the ORS market is positive, supported by sustained demand in emerging markets, a shift towards ready-to-drink formulations, and expanding healthcare access.
FAQs
- What is the primary therapeutic difference between Oracit and other standard ORS formulations? Oracit is a hypertonic ORS, meaning it has a higher concentration of electrolytes and glucose compared to isotonic or hypotonic solutions. This hypertonicity aims to enhance water and electrolyte absorption from the intestinal lumen.
- How significantly do fluctuations in dextrose prices impact Oracit's manufacturing cost? Dextrose is a significant component of ORS. Global commodity prices for sugar, which is a primary source for dextrose, can have a material impact on Oracit's production expenses.
- Are there specific regions where Oracit faces more intense price pressure from generic competitors? Price pressure from generic competitors is generally most acute in developing markets where affordability is a primary purchasing criterion. Regions with well-established generic pharmaceutical industries also exhibit higher competition.
- What are the main regulatory challenges for introducing Oracit into a new geographical market? The primary regulatory challenges include obtaining product registration and approval from local health authorities, ensuring compliance with local GMP standards, and meeting specific labeling and packaging requirements.
- Beyond diarrhea, are there any potential future indications for which Oracit or similar hypertonic ORS might be developed? While its primary indication is dehydration from diarrhea, hypertonic solutions could theoretically be explored for other conditions involving significant fluid and electrolyte loss, provided thorough clinical validation and regulatory approval are obtained for such new indications.
Citations
[1] World Health Organization. (2023). Diarrhoeal disease. Retrieved from https://www.who.int/news-room/fact-sheets/detail/diarrhoeal-disease
[2] UNICEF. (n.d.). Oral rehydration salts (ORS). Retrieved from https://www.unicef.org/supply/oral-rehydration-salts-ors
[3] National Institute of Diabetes and Digestive and Kidney Diseases. (2021). Dehydration. Retrieved from https://www.niddk.nih.gov/health-information/digestive-diseases/dehydration
[4] Alam, M. A., Alam, M. M., Ahmed, S., & Rahman, S. S. (2011). Oral rehydration solution: A review. Bangladesh Journal of Medicine Science, 10(2), 69-74.
More… ↓
