Share This Page
Drug Price Trends for MIRABEGRON ER
✉ Email this page to a colleague
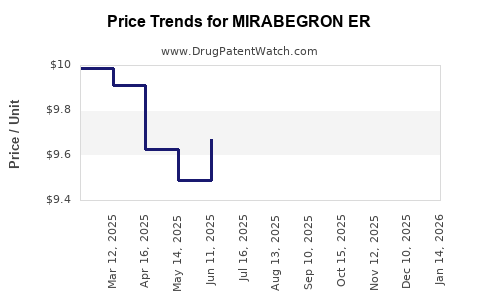
Average Pharmacy Cost for MIRABEGRON ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MIRABEGRON ER 50 MG TABLET | 70710-1160-03 | 9.63665 | EACH | 2026-03-18 |
| MIRABEGRON ER 25 MG TABLET | 70710-1159-09 | 9.55594 | EACH | 2026-03-18 |
| MIRABEGRON ER 50 MG TABLET | 68180-0152-09 | 9.63665 | EACH | 2026-03-18 |
| MIRABEGRON ER 25 MG TABLET | 68180-0151-06 | 9.55594 | EACH | 2026-03-18 |
| MIRABEGRON ER 50 MG TABLET | 68180-0152-06 | 9.63665 | EACH | 2026-03-18 |
| MIRABEGRON ER 25 MG TABLET | 68180-0151-09 | 9.55594 | EACH | 2026-03-18 |
| MIRABEGRON ER 25 MG TABLET | 70710-1159-03 | 9.55594 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Mirabegron ER
What is the current market landscape for Mirabegron ER?
Mirabegron ER (Extended Release) is a beta-3 adrenergic receptor agonist approved for overactive bladder (OAB) treatment. It gained FDA approval in 2012 and is marketed under brand names such as Myrbetriq. It primarily competes with antimuscarinic drugs like oxybutynin, tolterodine, and solifenacin.
The global OAB market reached approximately $3.2 billion in 2022, with Mirabegron ER capturing a significant share due to its side effect profile and distinct mechanism. In the U.S., Mirabegron ER sales totaled around $950 million in 2022, representing an 18% increase from 2021.
Market growth is driven by aging populations, increased diagnosis rates, and a preference for non-anticholinergic therapies due to fewer cognitive side effects. The drug is available in multiple formulations with dosages typically ranging from 25 mg to 50 mg.
What are the key market drivers and challenges?
Drivers
- Increasing prevalence of OAB, especially in populations over 60.
- Improved tolerability over antimuscarinic alternatives.
- Expanding approval for special populations, including elderly and patients with contraindications to anticholinergics.
- Growing awareness and diagnosis, driven by aging demographics and healthcare provider education.
Challenges
- Competition from newer agents, including combination therapies.
- Generic entry: Mirabegron became available generically in select markets in 2023, eroding patent exclusivity.
- Cost concerns; branded drug prices in the U.S. average about $450–$500 for a 30-day supply, compared to generics priced at about $150–$250.
- Limitations in long-term data, though ongoing studies evaluate safety and efficacy over longer periods.
How does patent status influence pricing?
Mirabegron ER's patent protection expired in December 2022 in the U.S. and in 2023 in Europe, enabling generic manufacturing. Patent exclusivity typically allows for premium pricing; once expired, prices often decline by 30–60%, depending on market dynamics.
In the U.S., the first generics entered the market in January 2023, leading to an immediate price drop. List prices for branded formulations declined from about $480 to under $300 per month post-generic entry, with pharmacists' retail prices diverging based on insurance coverage.
What are the price projections from 2023 to 2030?
Short-term (2023-2025)
Post-generic entry, prices are expected to decline sharply. The average wholesale price (AWP) for Mirabegron ER is projected to fall from $480 to approximately $240–$300 per month, a 40–50% decrease. Retail and insurance prices will follow, with co-pays for branded drugs likely increasing for insured patients.
Mid-term (2026-2028)
As competition stabilizes, generics will further commoditize the market, pushing prices down by an additional 20–30%. The average retail price is projected to be $180–$220 per month. It will be common for insurers to favor generics, reducing patient out-of-pocket costs for branded formulations.
Long-term (2029-2030)
Prices may stabilize between $150–$200 monthly for branded Mirabegron ER, with generics available at $100–$150. Growth in market penetration for combination therapies or new delivery systems may influence pricing strategies.
How do regulatory and market changes impact future pricing?
- New indications: Additional approvals for conditions like neurogenic bladder could maintain demand and support premium pricing.
- Reimbursement policies: Changes in insurance coverage and formulary placements significantly influence actual patient costs.
- Patent litigations: Ongoing patent disputes or extensions could delay generic entry, affecting prices.
How does Mirabegron ER compare with competitors?
| Drug | Brand Name | Approval Year | Class | Yearly Sales (2022) | Price Range (monthly) | Key Features |
|---|---|---|---|---|---|---|
| Mirabegron ER | Myrbetriq | 2012 | Beta-3 agonist | $950M | $450–$500 | Fewer cognitive side effects |
| Oxybutynin ER | Ditropan XR | 2004 | Anticholinergic | Not specified | $50–$150 | Generic availability, lower cost |
| Solifenacin | Vesicare | 2004 | Anticholinergic | Not specified | $150–$250 | Longer half-life, more side effects |
What is the outlook for future R&D and pricing strategies?
The pipeline involves combination therapies with Mirabegron, long-acting formulations, and new delivery systems aiming to improve adherence and reduce side effects. These developments could sustain a higher price point or extend patent exclusivity.
Manufacturers may adopt tiered pricing based on regional economic status, with premium prices in developed markets and lower prices in emerging markets. Strategic collaborations and licensing agreements in developing countries expand market access.
Key Takeaways
- Mirabegron ER’s market value peaked at nearly $1 billion in the U.S. before patent expiration, with prices declining rapidly post-generic entry.
- Market growth is driven by demographic shifts and preference for non-anticholinergic therapies.
- Price projections indicate a fall to $150–$220 monthly for branded formulations by 2028.
- Competition and patent expiration heavily influence pricing dynamics. Long-term viability depends on new indications and formulations.
- Generics dominate the lower-cost segment, constraining price premiums for branded Mirabegron ER.
FAQs
1. How long will Mirabegron ER remain profitable after patent expiry?
Profitability will decline within 1–2 years post-generics, but ongoing R&D and new applications could sustain revenues.
2. What regions have the highest market potential for Mirabegron ER?
The U.S., Europe, and Japan account for the largest markets due to aging populations and high diagnosis rates.
3. Are there fixed-price policies for Mirabegron ER?
Pricing is influenced by insurance negotiations, formularies, and regional drug pricing regulations. No fixed global price exists.
4. What impact will biosimilars or new drugs have on Mirabegron ER?
While biosimilars are not applicable, new drugs with superior efficacy or tolerability could reduce demand.
5. Will the price decline impact research investment?
Lower prices may limit profit margins but can stimulate volume growth; R&D focus is shifting toward combination and delivery innovations.
References
[1] IMS Health. (2023). Global Prescription Drug Market Data.
[2] IQVIA. (2022). U.S. Overactive Bladder Market Report.
[3] U.S. FDA. (2012). Mirabegron Approval Documents.
[4] European Medicines Agency. (2023). Drug Patent Expiry Data.
[5] GlobalData. (2023). Pharmaceutical Pricing and Reimbursement Outlook.
More… ↓
