Share This Page
Drug Price Trends for METHYLPHENIDATE ER(LA)
✉ Email this page to a colleague
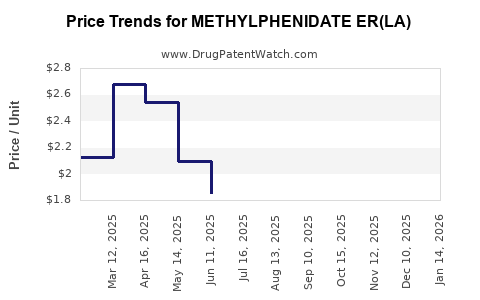
Average Pharmacy Cost for METHYLPHENIDATE ER(LA)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METHYLPHENIDATE ER(LA) 10 MG CP | 00781-2383-01 | 2.79956 | EACH | 2026-03-18 |
| METHYLPHENIDATE ER(LA) 10 MG CP | 75907-0049-01 | 2.79956 | EACH | 2026-03-18 |
| METHYLPHENIDATE ER(LA) 20 MG CP | 00093-5346-01 | 1.91012 | EACH | 2026-03-18 |
| METHYLPHENIDATE ER(LA) 10 MG CP | 70010-0012-01 | 2.79956 | EACH | 2026-03-18 |
| METHYLPHENIDATE ER(LA) 60 MG CP | 75907-0053-30 | 10.57109 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Methylphenidate ER (LA)
What is the current market landscape for methylphenidate ER (LA)?
Methylphenidate ER (LA) is a long-acting formulation of methylphenidate used primarily for attention deficit hyperactivity disorder (ADHD) and narcolepsy. It is marketed under brand names such as Concerta and Metadate ER, with multiple generic versions available. The global ADHD drug market was valued at approximately $13 billion in 2022 and targeted growth at a compound annual growth rate (CAGR) of 6.5% through 2028.[1]
The United States accounts for approximately 80% of methylphenidate market sales, driven by high diagnosis rates of ADHD and the widespread adoption of sustained-release formulations. The drug's favorable pharmacokinetic profile makes it a preferred option over immediate-release counterparts.
Who are the key players and patent statuses?
Major brand holders include Janssen Pharmaceuticals, which markets Concerta. Several generic manufacturers have entered the market following patent expirations, notably in the late 2010s. As of 2023, the primary patent for Concerta expired in 2017, leading to increased generic competition. Janssen retains exclusivity on certain extended-release patents until 2030, limiting generics' market share.
What are the main drivers influencing market growth?
Increase in ADHD diagnoses: Global diagnoses are rising, partly due to heightened awareness and better screening. The CDC reports a prevalence of 9.4% among children in the U.S.[2].
Off-label use: Growing off-label prescription for cognitive enhancement fuels demand.
Formulation improvements: Innovations yielding better compliance and fewer side effects sustain market relevance.
Regulatory approvals: New formulations, like OROS-based release mechanisms, expand accessible treatment options.
How is the pricing structured globally?
Brand-name drugs: In the U.S., a month’s supply of Concerta (18 mg) costs approximately $370–$420 retail, depending on pharmacy discounts.
Generics: Prices typically range between $90–$220 for the same dosage.[3]
Europe and Asia: Prices vary significantly based on healthcare systems, ranging from €50 to €150 per month.
What are the price projections and future trends?
Short-term (next 2 years): Prices for branded methylphenidate ER are expected to decline marginally due to increasing generic competition and biosimilar entries. The average price for a 30-day supply in the U.S. will likely fall to $300–$350.
Mid-term (3–5 years): Further generic penetration could reduce prices by an additional 20%. Patent protections on newer delivery systems, if extended, may temporarily stabilize or elevate prices of specific branded versions.
Long-term (beyond 5 years): Introduction of novel formulations or alternative active ingredients, such as lisdexamfetamine, may serve as substitutes, affecting methylphenidate ER (LA) pricing. Market consolidation and changes in prescribing guidelines will also influence pricing dynamics.
How might market shifts affect these projections?
- Regulatory changes: More stringent regulations on controlled substances could impact distribution and prices.
- Patent disputes: Legal challenges to existing patents could accelerate generic entry.
- New formulations: Extended-release versions with improved profiles might command premium pricing, delaying generic dominance.
- Global expansion: Markets in Asia-Pacific and Latin Americamay see increased adoption, creating opportunities for price increases in less mature markets.
Key Takeaways
- The methylphenidate ER (LA) market is mature, with significant generic competition following patent expirations.
- U.S. prices for branded versions range from approximately $370–$420 monthly, with generics priced at $90–$220.
- Market growth is driven by rising ADHD diagnoses and shifting prescribing practices, though pricing is contracting due to generics.
- Future pricing will depend on patent protection duration, formulation innovations, and regulatory factors.
- Overall, prices are expected to decline modestly over the next 3–5 years, with the potential for stabilization in branded markets through new formulations.
FAQs
1. How does the patent landscape affect methylphenidate ER (LA) pricing?
Expired patents have led to widespread generic entry, significantly reducing prices. Patent extensions on formulations could delay generics, maintaining higher prices temporarily.
2. Will new formulations impact market prices?
Yes. Formulations with improved delivery mechanisms or reduced side effects may command higher prices, moderating generic price declines.
3. How do international markets compare in price and access?
Pricing varies widely. Countries with centralized health systems or price controls, like the UK or Canada, pay less than the U.S. market. Emerging markets experience lower prices but face access barriers.
4. Are there significant biosimilars or alternatives to methylphenidate ER (LA)?
Biosimilars are limited, but other stimulant medications like amphetamine-based drugs are competitors, potentially influencing methylphenidate’s market share and pricing.
5. What regulatory developments could influence future prices?
Changes in controlled substance regulations, approval processes for generics, or market entry of new drugs could impact supply chains and prices.
References
[1] IQVIA, "Global ADHD Market Data," 2022.
[2] CDC, "Attention-Deficit/Hyperactivity Disorder (ADHD)," 2022.
[3] GoodRx, "Comparison of drug prices," 2023.
More… ↓
