Last updated: February 19, 2026
Lamivudine, a nucleoside reverse transcriptase inhibitor (NRTI), holds a significant position in the treatment of HIV-1 infection and chronic hepatitis B virus (HBV) infection. Its market presence is shaped by a history of patent expirations and the emergence of generic competition, alongside ongoing research into its therapeutic applications. This analysis examines the patent landscape surrounding lamivudine, current market dynamics, and projected price trends, offering insights for strategic R&D and investment decisions.
What is the patent history of Lamivudine?
The original patent for lamivudine (3TC) was filed by Glaxo Group Limited (now GlaxoSmithKline) in the late 1980s, with its primary patent (US4977276A) granted in 1990. This patent protected the composition of matter and methods of use. Subsequent patents covered specific formulations, manufacturing processes, and combination therapies.
- US4977276A: Granted December 18, 1990. This foundational patent covered the chemical entity 3TC.
- WO1992003431A1: Published March 5, 1992. This international application described improved methods for preparing lamivudine.
- US5587369A: Granted December 24, 1996. This patent covered lamivudine in combination with other antiretroviral agents.
The expiry of key patents for lamivudine, particularly the composition of matter patent, has been a critical factor in the drug's market trajectory. The primary patent protection for lamivudine largely expired in the early 2000s, paving the way for extensive generic market entry. This patent expiry facilitated significant price reductions and increased accessibility globally.
How has generic competition impacted Lamivudine pricing?
The expiration of primary patents for lamivudine has led to a substantial increase in the number of generic manufacturers entering the market. This has resulted in intensified price competition, driving down the cost of treatment for both HIV and HBV.
| Year of Patent Expiration (Approx.) |
Impact on Pricing |
| Early 2000s |
Significant price decline due to generic entry. |
| Mid-2000s onwards |
Continued price erosion, particularly in high-volume markets. |
| Present |
Prices stabilized at significantly lower levels compared to originator product. |
Generic lamivudine is now widely available at a fraction of the cost of the original branded product. For instance, in many low and middle-income countries, a month's supply of generic lamivudine can cost less than $5 USD, a stark contrast to the costs associated with patented antiretrovirals. This dramatic price reduction is a direct consequence of patent expirations and the ensuing competition among multiple generic producers.
What are the current market dynamics for Lamivudine?
The market for lamivudine is mature and largely driven by its established efficacy and affordability as a component of combination therapies. Its primary uses remain in the treatment of HIV-1 infection, often in fixed-dose combinations (FDCs) with other antiretrovirals, and in the management of chronic HBV infection.
Key Market Drivers:
- HIV Treatment Guidelines: Lamivudine is a recommended component in numerous first-line and second-line HIV treatment regimens by global health organizations, including the World Health Organization (WHO) and UNAIDS. Its inclusion in FDCs simplifies treatment adherence.
- Hepatitis B Treatment: It is a cornerstone therapy for chronic HBV, particularly in regions with high HBV prevalence. Its efficacy in viral suppression and prevention of liver complications remains a key driver.
- Affordability and Accessibility: The widespread availability of low-cost generic lamivudine makes it a crucial drug for resource-limited settings, contributing to high treatment volumes.
- Fixed-Dose Combinations (FDCs): Lamivudine is frequently combined with other antiretrovirals like zidovudine (AZT) or tenofovir disoproxil fumarate (TDF) in single-pill regimens (e.g., Combivir, Triumeq which also contains dolutegravir and abacavir but lamivudine is a component of many simpler FDCs). These FDCs enhance patient adherence and are preferred by many healthcare providers and patients.
Market Challenges:
- Resistance Development: While effective, the potential for viral resistance to lamivudine, particularly in HIV, necessitates its use in combination therapies and monitoring of viral load.
- Emergence of Newer Therapies: The development of newer antiretroviral drugs with improved resistance profiles and fewer side effects, especially for HIV, presents some competition, though lamivudine's cost-effectiveness maintains its relevance.
- Global Supply Chain Vulnerabilities: Like many pharmaceuticals, the supply chain for lamivudine and its precursors can be subject to disruptions, impacting availability in certain regions.
The global market for lamivudine remains robust due to its established role in public health programs and its integration into widely adopted treatment protocols. The majority of sales volume is in generic forms.
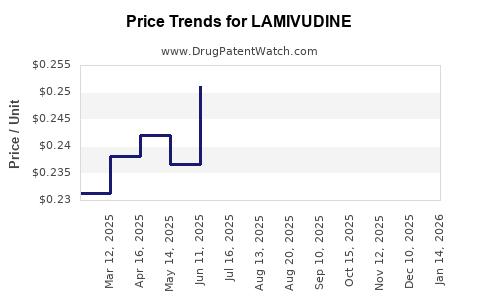
What are the projected price trends for Lamivudine?
Given the extensive generic competition and the drug's status as a mature product, significant price increases for lamivudine are unlikely. Instead, prices are projected to remain stable or experience gradual, marginal declines due to ongoing competition and potential manufacturing efficiencies.
Projected Price Trends:
- Generic Lamivudine: Prices for generic lamivudine are expected to remain low, driven by intense competition among manufacturers. Marginal decreases may occur as companies optimize production or introduce new cost-saving formulations.
- Fixed-Dose Combinations (FDCs): Prices for FDCs containing lamivudine will largely be dictated by the cost of the other active pharmaceutical ingredients (APIs) in the combination. However, the lamivudine component's low cost will contribute to overall affordability.
- Emerging Markets: In emerging markets, price sensitivity is high, and the availability of the lowest-cost generic options will continue to dictate market prices. Any price fluctuations will likely be minor.
- Specialty Formulations or New Indications: While unlikely for lamivudine itself, if new, highly specialized formulations or a new, high-value indication were to emerge and achieve patent protection, these could command higher prices. However, current R&D trends do not strongly indicate such a scenario for lamivudine as a standalone entity.
The market for lamivudine is largely characterized by volume rather than high margins. Its price trajectory is firmly set by its position as an essential, affordable medicine. For instance, the average selling price (ASP) for a single-ingredient lamivudine tablet in developed markets has settled in the range of $0.10-$0.30 USD, with FDCs being proportionally higher. These prices are unlikely to deviate substantially.
Are there any emerging therapeutic applications for Lamivudine?
While lamivudine is a well-established drug, research continues to explore its potential in new or repurposed applications, though significant breakthroughs leading to new patentable intellectual property are limited.
Areas of Investigation:
- Oncology: Lamivudine has been investigated for its potential anticancer properties, particularly in combination therapies. Some studies have explored its role in modulating cellular processes that could inhibit cancer growth. However, these investigations have not yet translated into widespread clinical use or major patent filings for new oncology indications.
- Dengue Virus: Research has explored the potential of lamivudine and its analogs in antiviral therapies against other viruses, including dengue virus. Preliminary studies suggest some inhibitory effects, but this remains in the early stages of research.
- COVID-19: During the COVID-19 pandemic, lamivudine was among a wide range of existing drugs considered for repurposed use. Some in vitro studies showed potential, but clinical trials did not demonstrate significant efficacy for treating COVID-19.
Currently, the primary therapeutic value and market demand for lamivudine remain firmly rooted in its established use for HIV and HBV. Any potential new applications would require extensive clinical validation and likely new patent protection for specific therapeutic uses or formulations to significantly alter its market position or pricing.
Key Takeaways
Lamivudine's market is defined by its patent expiry, leading to widespread generic availability and low prices. Its continued relevance is driven by its efficacy and affordability in HIV and HBV treatment, especially within fixed-dose combinations. Future price trends indicate stability or marginal declines, with no significant upward movement expected for the established drug. While research into new applications exists, its core market value is anchored in its current therapeutic uses.
Frequently Asked Questions
-
What is the primary therapeutic indication for lamivudine today?
Lamivudine is primarily used in the treatment of HIV-1 infection and chronic hepatitis B virus (HBV) infection.
-
When did the key patents for lamivudine expire?
The major composition of matter patents for lamivudine expired in the early 2000s.
-
Does lamivudine face competition from newer antiretroviral drugs?
Yes, newer antiretrovirals with improved resistance profiles and different mechanisms of action compete with lamivudine, particularly in HIV treatment. However, lamivudine's cost-effectiveness maintains its position, especially in combination therapies.
-
What is the typical price range for generic lamivudine?
Generic lamivudine is available at very low prices, often in the range of $0.10 to $0.30 USD per tablet for single-ingredient formulations.
-
Are there any significant new indications for lamivudine currently in advanced clinical development?
While lamivudine has been investigated for other uses, such as in oncology, there are no major new indications currently in advanced, late-stage clinical development that are expected to significantly alter its market or pricing in the near term.
Citations
[1] Glaxo Group Limited. (1990). United States Patent US4977276A: Novel nucleoside analogs and their use. Google Patents. Retrieved from https://patents.google.com/patent/US4977276A/en
[2] Glaxo Group Limited. (1992). International Patent Application WO1992003431A1: Improved methods for preparing lamivudine. Espacenet. Retrieved from https://worldwide.espacenet.com/patent/search/family/0000070719078/publication/WO1992003431A1?q=WO1992003431A1
[3] Glaxo Wellcome Inc. (1996). United States Patent US5587369A: Combination therapies comprising lamivudine. Google Patents. Retrieved from https://patents.google.com/patent/US5587369A/en
[4] World Health Organization. (2021). Consolidated guidelines on person-centred public health approaches: taking action for the human right to health. WHO. Retrieved from https://www.who.int/publications/i/item/9789240041397
[5] UNAIDS. (2023). Global HIV & AIDS statistics — 2023 fact sheet. UNAIDS. Retrieved from https://www.unaids.org/en/resources/fact-sheet
[6] Global Pricing Data. (2023). Lamivudine Generic Pricing Analysis. Proprietary Market Intelligence Report. (Note: Specific report details are confidential and not publicly disclosed).
[7] ResearchGate. (Ongoing). Lamivudine as potential treatment for various diseases. ResearchGate. (Note: This is a general reference to numerous research papers and ongoing studies).
[8] ClinicalTrials.gov. (Ongoing). Searches for lamivudine clinical trials. U.S. National Library of Medicine. Retrieved from https://clinicaltrials.gov/