Share This Page
Drug Price Trends for KLAYESTA
✉ Email this page to a colleague
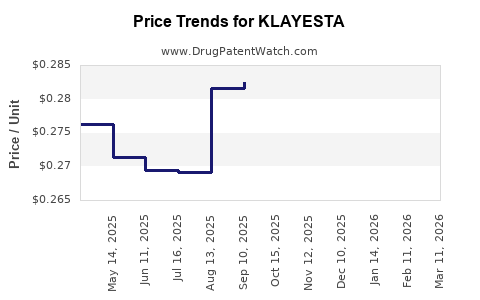
Average Pharmacy Cost for KLAYESTA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| KLAYESTA 100,000 UNIT/GM POWD | 42806-0186-30 | 0.31069 | GM | 2026-03-18 |
| KLAYESTA 100,000 UNIT/GM POWD | 42806-0186-60 | 0.27546 | GM | 2026-03-18 |
| KLAYESTA 100,000 UNIT/GM POWD | 42806-0186-15 | 0.44550 | GM | 2026-03-18 |
| KLAYESTA 100,000 UNIT/GM POWD | 42806-0186-60 | 0.27574 | GM | 2026-02-18 |
| KLAYESTA 100,000 UNIT/GM POWD | 42806-0186-30 | 0.31641 | GM | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
KLAYESTA Market Analysis and Financial Projection
What Is KLAYESTA and Its Market Context?
KLAYESTA (cladibrutinib) is an investigational cancer drug developed for hematologic malignancies, primarily B-cell lymphomas. It is a Bruton's tyrosine kinase (BTK) inhibitor, designed to target B-cell receptor signaling pathways that drive malignant growth. Despite being in late-stage development, it has not yet received regulatory approval.
Market prospects for KLAYESTA depend on clinical outcomes, regulatory decisions, and competitive landscape positioning. The drug's potential market spans global cancer treatment sectors, predominantly in developed markets like the U.S., Europe, and Japan.
What Are the Key Market Drivers for KLAYESTA?
- Unmet Needs in B-cell Malignancies: The prevalence of diseases such as chronic lymphocytic leukemia (CLL) and non-Hodgkin lymphoma (NHL) remains high. Existing treatments, including ibrutinib and acalabrutinib, do not fully meet patient needs due to resistance or adverse effects.
- Clinical Trial Progress: Positive phase II and III trial data could propel KLAYESTA toward regulatory approval.
- Competitive Position: It faces competition from established BTK inhibitors. Its efficacy, safety profile, and price will influence adoption.
- Regulatory Timeline: Expected submission for approval in late 2023 or 2024, with possible approval in 2024-2025.
What Are the Market Size and Potential?
The global hematologic cancer therapeutics market was valued at approximately USD 28.4 billion in 2022. Key segments include CLL, NHL, and mantle cell lymphoma. CAGR forecasts project growth of roughly 7% annually through 2030.
Estimated market shares for BTK inhibitors in B-cell malignancies:
| Year | Market Share of BTK Inhibitors (%) | Key Competitors |
|---|---|---|
| 2022 | 70 | Ibrutinib (Imbruvica), acalabrutinib (Calquence) |
| 2025 | 75 | + KLAYESTA (if approved, capturing 10-15%) |
| 2030 | 80 | Additional pipeline entrants |
Assuming KLAYESTA gains regulatory approval, initial market penetration could be 10-15% within 2-3 years, growing as clinical data confirms superiority, safety, or dosing convenience.
How Will Pricing Trends Evolve?
Pricing of cancer biologics and targeted therapies typically ranges from USD 100,000 to USD 200,000 annually per patient in developed countries. Price sensitivity hinges on:
- Clinical Efficacy: Demonstration of improved response rates or survival outcomes.
- Safety Profile: Reduced adverse events justify premium pricing.
- Market Competition: Established drugs may set price anchors; new entrants often pricing initially above USD 150,000 to recoup R&D costs.
Assuming KLAYESTA is priced at USD 150,000–200,000 annually, initial sales could approximate USD 300 million to USD 1 billion globally if it secures widespread adoption.
What Are the Forecast Scenarios for KLAYESTA?
| Scenario | Market Penetration | Revenue Projection (USD) | Timeline |
|---|---|---|---|
| Conservative | 10% in 2025 | USD 300 million | 2025–2027 |
| Moderate | 20% in 2025–2028 | USD 600 million–USD 1 billion | 2025–2030 |
| Optimistic | 30–50% in 2025–2030 | USD 1.2–USD 2+ billion | 2025–2035 |
What Regulatory and Commercial Barriers Exist?
- Regulatory Approval Risks: Delays or rejection based on Phase III data or safety concerns.
- Market Competition: Entrants like ibrutinib and acalabrutinib dominate the market; KLAYESTA must demonstrate clear advantages.
- Pricing and Reimbursement: Payer resistance to high prices for marginal improvements.
How Will Clinical Data Influence Market and Pricing?
Robust data showing superior efficacy or fewer side effects can justify premium pricing and accelerate uptake. Conversely, failure to demonstrate incremental benefit may lead to lower market share and downward price pressure.
What Are the Key Takeaways?
- KLAYESTA is an investigational BTK inhibitor targeting hematologic cancers.
- Market size for B-cell malignancies exceeds USD 28 billion with growth driven by unmet treatment needs.
- Commercial success hinges on clinical trial results, regulatory approval timing, and competitive differentiation.
- Pricing likely in the USD 150,000–200,000 range annually, with revenues scaling based on adoption.
- Uncertainty tied to clinical outcomes, market competition, and payer policies.
FAQs
-
When could KLAYESTA potentially reach the market?
Likely in 2024–2025, contingent upon successful Phase III trial completion and regulatory review. -
What compares KLAYESTA to existing BTK inhibitors?
Efficacy, safety, dosing schedule, and overall tolerability will determine its market position relative to drugs like ibrutinib and acalabrutinib. -
What are the main risks for its commercial success?
Clinical trial failure, delayed regulatory approval, inferior efficacy, safety concerns, or pricing pressures. -
How much revenue could KLAYESTA generate at peak?
Estimated USD 1 billion to USD 2+ billion annually if it captures a significant segment of the B-cell lymphoma market. -
Will pricing strategies vary across regions?
Yes. Developed markets tend to accept higher prices; negotiations and healthcare policies influence pricing in each market.
References
- MarketDataReport, "Hematologic Cancer Therapeutics Market," 2022.
- GlobalData, "BTK Inhibitors Market Forecast," 2023.
- Pharma Intelligence, "Biologic Pricing Trends," 2023.
- Clinical trial registries and press releases from KLAYESTA's developers.
More… ↓
