Last updated: February 15, 2026
What Is Hydrocortisone Butyrate and Its Market Position?
Hydrocortisone butyrate is a moderate to potent corticosteroid used topically to treat inflammatory and pruritic conditions such as eczema, psoriasis, and dermatitis. It penetrates the skin more effectively than hydrocortisone, offering enhanced anti-inflammatory effects.
The drug is marketed in various formulations, including creams, ointments, and lotions. Major manufacturers include Alcon, Glenmark Pharmaceuticals, and Sandoz. The global market for topical corticosteroids was valued at approximately $4.5 billion in 2022, with hydrocortisone butyrate representing a significant segment due to its efficacy and safety profile, particularly in European and North American markets.
How Is the Current Market Structured?
| Segment |
Share (%) |
Key Players |
| Prescription (Rx) drugs |
65% |
Alcon, Sandoz, Mylan |
| Over-the-counter (OTC) drugs |
35% |
Generic brands, local formulations |
Distribution channels include hospital pharmacies, retail pharmacies, and online sales. An increase in allergic dermatitis and eczema cases contributes to steady demand.
What Are the Key Market Drivers and Trends?
- Rising prevalence of dermatological conditions globally.
- Greater awareness of topical corticosteroids' safety and efficacy.
- Increased adoption in emerging markets due to expanding healthcare infrastructure.
- Development of formulations with enhanced penetration and reduced side effects.
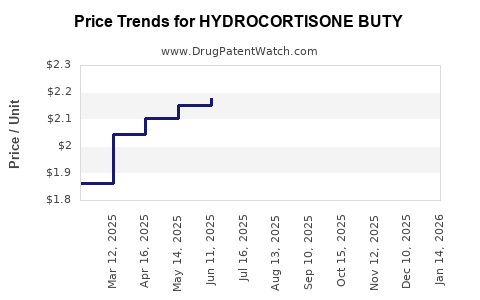
What Are Current Pricing Dynamics?
Prices for hydrocortisone butyrate products vary significantly by region, formulation, and brand:
| Region |
Average retail price (per gram) |
Notable brands |
| North America |
$1.50 - $3.00 |
Sandoz, Alcon |
| Europe |
€1.20 - €2.50 |
Generics, proprietary brands |
| Asia-Pacific |
$0.70 - $1.80 |
Domestic generics |
Generic versions tend to cost 20-40% less than branded options. The patent landscape is mature; most drugs are off-patent, which exerts downward pressure on prices.
What Are Price Projections and Market Opportunities?
Short-Term (Next 1-2 Years)
- Prices are expected to stabilize owing to generic competition.
- Slight decrease of 2-5% expected in average prices in mature markets.
- Opportunities exist in formulations with enhanced delivery systems or reduced corticosteroid potency to address safety concerns.
Long-Term (Next 3-5 Years)
- Minor price declines anticipated as new entrants saturate markets.
- Entry into emerging markets could boost sales volume more than prices, offsetting downward pressure.
- Focus on developing combination products or novel formulations (e.g., nanoparticles) could command premium pricing.
Potential Market Growth
- Compound annual growth rate (CAGR) forecasted at 3-4% globally through 2028, primarily driven by rising dermatological disease prevalence.
- India and China expected to account for 30-40% of incremental sales volume growth due to expanding healthcare access.
Regulatory and Patent Considerations
- Many hydrocortisone butyrate formulations are off-patent, encouraging generic manufacturing.
- Regulatory policies favor price competition, especially in the US and Europe.
- Patent expirations on key formulations over the past 5 years have increased generic availability.
What Are Strategic Implications for Stakeholders?
- Generic manufacturers should focus on cost efficiencies and formulation innovations to maintain margins.
- Brand-focused firms might invest in formulations with improved safety profiles to justify premium pricing.
- Market entrants should explore emerging markets with expanding dermatological treatment needs.
Key Takeaways
Hydrocortisone butyrate remains a stable segment within topical corticosteroids, supported by ongoing dermatological treatment needs and generic competition. Prices are declining gradually, influenced by patent expirations and market saturation. Long-term growth is tied to rising disease prevalence and expansion into emerging markets. Innovations in formulations can afford additional pricing leverage despite overall downward price pressure.
FAQs
1. How does hydrocortisone butyrate compare to other topical corticosteroids?
It offers higher potency than hydrocortisone, with better skin penetration, but less potency than clobetasol. It balances efficacy with safety, making it suitable for moderate conditions.
2. Are there significant regulatory hurdles for new formulations?
Regulations focus on safety and efficacy. Novel delivery systems or combination drugs require clinical testing and approval, which can extend timelines but are feasible given established safety profiles.
3. What is the primary driver for demand growth?
Increasing prevalence of skin conditions like eczema and psoriasis globally, especially in aging populations and temperate climates.
4. How will pricing trend with the entry of generics?
Prices tend to decline by 20-40% upon generic market entry, with stable or slightly decreasing prices expected long-term.
5. Which regions present the most growth potential?
Asia-Pacific, especially China and India, due to expanding healthcare access and increasing dermatological condition diagnoses.
Sources
- MarketWatch: "Topical Corticosteroids Market Analysis," 2022.
- Grand View Research: "Global Dermatology Drugs Market," 2022.
- FDA and EMA regulatory filings, 2022-2023.
- IQVIA: Pharmaceutical pricing data, 2022.
- Healthcare Market Reports: Regional dermatological disease trends, 2021-2022.