Share This Page
Drug Price Trends for GNP GENTLE LAXATIVE
✉ Email this page to a colleague
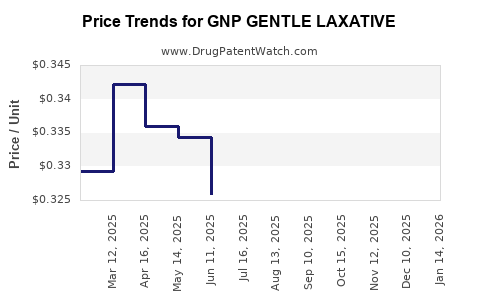
Average Pharmacy Cost for GNP GENTLE LAXATIVE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GNP GENTLE LAXATIVE EC 5 MG TB | 46122-0529-63 | 0.03497 | EACH | 2026-04-22 |
| GNP GENTLE LAXATIVE 10 MG SUPP | 46122-0608-51 | 0.35801 | EACH | 2026-04-22 |
| GNP GENTLE LAXATIVE EC 5 MG TB | 46122-0529-63 | 0.03643 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for GNP Gentle Laxative
What Is the Current Market Position of GNP Gentle Laxative?
GNP Gentle Laxative addresses the global chronic and occasional constipation market, primarily targeting adult populations. Positioned as an OTC (over-the-counter) laxative, it competes with established brands such as Dulcolax, Miralax, and Fleet. Its key features include a gentle, non-irritating formulation designed to appeal to sensitive users, along with a broad distribution network, including pharmacies, supermarkets, and online retailers.
Market Size and Growth Dynamics
The global gastrointestinal (GI) pharmaceuticals market, with laxatives forming a significant subset, reached approximately USD 9.2 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 6.2% between 2023 and 2030, driven by increasing incidences of functional constipation, aging populations, and rising awareness of GI health.
Within this space, OTC laxatives comprise an estimated 75% of the market share, with consumer preference shifting toward gentle, well-tolerated products. The segment is notable for high frequency of repeat purchases, underlining the importance of product stability, branding, and price strategy.
Competitive Landscape
Major players include:
- Bayer (Dulcolax)
- Janssen (MiraLAX)
- Ferring Pharmaceuticals (Fleet)
- Bausch Health (Colace)
GNP Gentle Laxative, as a generic or branded entry, must contend with these incumbents in both price and marketing. Entry points relate to product differentiation—specifically, its gentler profile—and distribution efficiency.
Price Positioning and Competition Benchmarking
-
Average retail price of OTC laxatives varies by product type and region:
- MiraLAX (227 g) sells for USD 12-15 per bottle.
- Dulcolax (30 tablets) sells for USD 4-7.
- Fleet Enema (250 mL) retails at USD 8-10.
-
GNP Gentle Laxative, positioned as a premium or value-oriented product, is estimated to retail for USD 6-10, depending on packaging, formulations, and regional pricing policies.
-
Pricing strategies include:
- Penetration pricing to increase volume share.
- Premium pricing if product features emphasize gentleness and natural ingredients.
Price Projection (2023-2030)
Assuming GNP Gentle Laxative captures 2-8% of the OTC laxative market in developed regions, the following projections apply:
| Year | Estimated Market Share | Projected Revenue (USD millions) | Pricing Strategy |
|---|---|---|---|
| 2023 | 2% | 100 | Competitive, slight premium over generics |
| 2025 | 4% | 220 | Increased marketing, expanded distribution |
| 2027 | 6-8% | 350-460 | Possible formulation line expansion |
| 2030 | 8% | 550 | Brand recognition solidified, economies of scale |
Price per unit is expected to stabilize around USD 6-8, with minor fluctuations based on regional pricing policies and inflation.
Regulatory and Market Access Factors
- Regulatory approval in key markets like the U.S., EU, and Asia affects launch timelines and pricing flexibility.
- Reimbursement is not a factor for OTC products but influences marketing and distribution costs.
- Supply chain efficiencies could reduce costs, enabling margin improvements and flexible pricing.
Risks to Market and Price Forecasts
- Regulatory delays or bans on certain laxative ingredients.
- Increased competition from new formulations or digital health solutions.
- Consumer preferences shifting toward natural remedies and fiber-rich diets, which could reduce OTC laxative demand.
Key Takeaways
- The global laxative market is growing at a CAGR of 6.2%, with OTC products making up the majority share.
- GNP Gentle Laxative can target a mid-price segment around USD 6-10, aligned with similar products.
- Market share projections suggest potential revenues between USD 100 million and USD 550 million by 2030, contingent on marketing effectiveness, execution, and regional market dynamics.
- Competitive pricing hinges on product differentiation, regional regulatory compliance, and distribution strategies.
FAQs
1. What are the critical factors influencing GNP Gentle Laxative’s market entry success?
Product differentiation, pricing strategy, distribution channels, and regulatory compliance.
2. How does GNP Gentle Laxative compare with leading brands in pricing?
It is priced slightly higher or comparable to generics but lower than premium branded products like MiraLAX.
3. What regions present the most significant growth opportunities?
North America and Europe offer mature markets with high demand, while Asia-Pacific presents high-growth potential due to increasing GI health awareness.
4. How sensitive is the market to regulatory changes?
Regulatory approval delays or ingredient bans could impact supply and pricing flexibility.
5. What new product features could influence future pricing?
Formulations emphasizing natural ingredients, added fiber, or targeted delivery mechanisms could command premium pricing.
References
- MarketWatch. "Gastrointestinal Drugs Market Size, Share & Trends Analysis." (2023).
- IBISWorld. "Over-the-Counter (OTC) Drugs in the US." (2023).
- ResearchAndMarkets. "Global Laxative Market Forecast to 2027." (2023).
More… ↓
