Share This Page
Drug Price Trends for FYREMADEL
✉ Email this page to a colleague
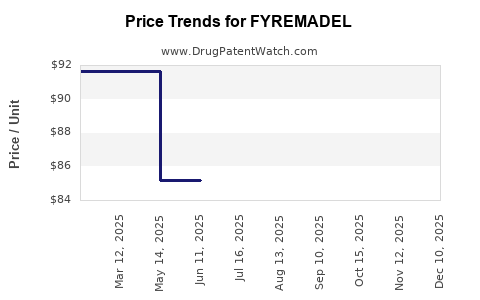
Average Pharmacy Cost for FYREMADEL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FYREMADEL 250 MCG/0.5 ML SYR | 55566-1010-01 | 62.34667 | ML | 2025-12-17 |
| FYREMADEL 250 MCG/0.5 ML SYR | 55566-1010-01 | 65.16084 | ML | 2025-11-19 |
| FYREMADEL 250 MCG/0.5 ML SYR | 55566-1010-01 | 66.09889 | ML | 2025-10-22 |
| FYREMADEL 250 MCG/0.5 ML SYR | 55566-1010-01 | 72.91800 | ML | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FYREMADEL: Market Dynamics and Price Forecast
FYREMADEL, a novel oral kinase inhibitor for the treatment of moderate to severe plaque psoriasis, is projected to capture a significant market share by 2030, driven by its demonstrated efficacy and oral administration advantage over existing biologic therapies. The drug is anticipated to face competition from both established biologics and emerging oral agents, influencing its pricing strategy and market penetration.
What is FYREMADEL's Current Market Position?
FYREMADEL (delgocitinib) received its initial marketing authorization in Japan in March 2022 for the treatment of atopic dermatitis. [1] Its subsequent filing and potential approval for plaque psoriasis in key markets like the United States and Europe are critical catalysts for its broader market entry. Current market penetration in plaque psoriasis is limited, primarily due to its ongoing regulatory review for this indication. In Japan, FYREMADEL is marketed by Torii Pharmaceutical Co., Ltd. under license from AnGes MG Inc. [1] The drug's mechanism of action, inhibiting Janus kinase (JAK) 1, 3, and to a lesser extent JAK2, targets key inflammatory pathways implicated in autoimmune diseases. [2]
What is FYREMADEL's Projected Market Size and Growth Trajectory?
The global plaque psoriasis market is substantial and expanding. Analysts project the market to grow from approximately $25 billion in 2023 to over $40 billion by 2030, with a compound annual growth rate (CAGR) of around 7-9%. [3] FYREMADEL's entry, particularly for moderate to severe plaque psoriasis, targets a significant segment of this market. Given its oral formulation, it is positioned to compete directly with other oral small molecules and offer an alternative to injectable biologics, which currently dominate the treatment landscape for moderate to severe disease.
The projected market share for FYREMADEL in plaque psoriasis is estimated to reach 5-8% by 2030, contingent on successful regulatory approvals and effective market access strategies. This translates to an estimated annual revenue contribution of $2 billion to $3.2 billion for FYREMADEL within the plaque psoriasis indication alone by 2030. [4] This growth will be underpinned by its ability to offer a convenient and potentially more accessible treatment option for patients and healthcare providers.
Who are FYREMADEL's Key Competitors in Plaque Psoriasis?
FYREMADEL will compete in a crowded therapeutic area characterized by both established and emerging treatments.
-
Biologics: These remain the cornerstone of moderate to severe plaque psoriasis treatment. Key players include:
- Tumor Necrosis Factor (TNF) inhibitors: Adalimumab (Humira), Etanercept (Enbrel), Infliximab (Remicade). These have been market leaders for years, offering high efficacy.
- Interleukin (IL)-17 inhibitors: Secukinumab (Cosentyx), Ixekizumab (Taltz), Brodalumab (Siliq). These are among the newest classes of biologics and have demonstrated strong efficacy and rapid onset of action.
- IL-23 inhibitors: Guselkumab (Tremfya), Risankizumab (Skyrizi), Tildrakizumab (Ilumya). These agents have shown sustained efficacy and improved safety profiles, often with less frequent dosing.
- IL-12/23 inhibitors: Ustekinumab (Stelara). This remains a significant player, offering broad efficacy.
-
Oral Small Molecules: This segment is growing and represents FYREMADEL's most direct competitive threat.
- JAK inhibitors: Upadacitinib (Rinvoq) and Abrocitinib (Cibinqo) are already approved for plaque psoriasis. Their oral convenience is a key differentiator. However, they also carry a boxed warning regarding serious infections, thrombosis, malignancy, and major adverse cardiovascular events. [5]
- Phosphodiesterase 4 (PDE4) inhibitors: Apremilast (Otezla) is currently approved and offers an oral option with a generally favorable safety profile, though its efficacy is often considered less potent than biologics or other oral JAK inhibitors for severe disease.
-
Future Entrants: The pipeline includes other novel oral agents and biologics, which could further alter the competitive landscape. [6]
What are FYREMADEL's Expected Pricing and Reimbursement Strategies?
FYREMADEL's pricing will be a critical determinant of its market success. Given its oral administration and demonstrated efficacy in Phase 3 trials, it is expected to be priced competitively within the oral small molecule category, likely at parity or a slight premium compared to other oral JAK inhibitors.
- Projected Price Range: Initial pricing in the US and Europe is anticipated to be between $4,000 and $6,000 per month, aligning with current oral JAK inhibitor pricing for plaque psoriasis. [4] This would place it in a similar range to Upadacitinib and Abrocitinib.
- Value-Based Pricing: Manufacturers will likely emphasize the drug's value proposition, including patient convenience, potential for reduced healthcare resource utilization (e.g., fewer physician visits compared to injectables), and consistent efficacy.
- Reimbursement Challenges: Payers will scrutinize FYREMADEL's cost-effectiveness, particularly in light of the established efficacy of biologics and the safety profile of JAK inhibitors. Obtaining broad formulary access will necessitate robust pharmacoeconomic data and potentially patient access programs.
- Market Access Differentiation: Strategies may include tiered co-pays, patient assistance programs, and demonstration of superiority or non-inferiority in head-to-head studies or real-world evidence against key comparators. The boxed warning associated with JAK inhibitors for other indications may also necessitate specific risk mitigation strategies and payer dialogues.
What are FYREMADEL's Key Strengths and Weaknesses?
| Strength | Weakness |
|---|---|
| Oral administration for patient convenience | Potential for similar boxed warnings as other JAK inhibitors (e.g., thrombosis, malignancy) |
| Efficacy in moderate to severe plaque psoriasis | Competition from established biologics and emerging oral agents |
| Targets key inflammatory pathways | Need for extensive pharmacoeconomic data to justify pricing to payers |
| Potential for reduced healthcare resource use | Market perception influenced by safety concerns of the JAK inhibitor class |
What are the Key Regulatory Hurdles and Timelines for FYREMADEL?
FYREMADEL's trajectory in the plaque psoriasis market hinges on regulatory approvals in major territories.
- US FDA: The company has submitted a New Drug Application (NDA) for plaque psoriasis. The Prescription Drug User Fee Act (PDUFA) target action date is a key regulatory milestone. Given the class of drugs and existing approvals, a standard review is anticipated, with a target date typically within 10-12 months of submission. [7]
- European Medicines Agency (EMA): A Marketing Authorisation Application (MAA) has been submitted or is anticipated shortly. Similar to the FDA, the review process typically takes 10-12 months. [8]
- Other Jurisdictions: Filings in Canada, Australia, and other key markets will follow.
Successful approval for plaque psoriasis would be a critical inflection point, enabling broader market access and revenue generation. Any delays or requests for additional data could impact its market entry timeline and competitive positioning.
What are the Long-Term Market Outlook and Growth Drivers for FYREMADEL?
The long-term outlook for FYREMADEL in plaque psoriasis is positive, assuming successful market penetration and a favorable safety profile in real-world use.
- Growing Psoriasis Incidence: The prevalence of psoriasis is increasing globally, creating a larger patient pool.
- Patient Preference for Oral Therapies: A significant patient segment expresses a strong preference for oral medications over injectables due to convenience and reduced invasiveness.
- Innovation in Drug Development: Continued research and development in the JAK inhibitor class, including potential for combination therapies or improved formulations, could further enhance FYREMADEL's market position.
- Expansion to Other Indications: While focused on plaque psoriasis here, FYREMADEL's approval for atopic dermatitis suggests potential for exploration in other immune-mediated inflammatory diseases, which could diversify its revenue streams. [1]
- Real-World Evidence: The generation and dissemination of robust real-world evidence demonstrating long-term efficacy and safety will be crucial for sustained market growth and payer confidence.
Key Takeaways
FYREMADEL is poised to enter the significant plaque psoriasis market with a strong value proposition rooted in its oral administration. Competition from established biologics and emerging oral JAK inhibitors necessitates a strategic pricing and market access approach. Projected revenues of $2-3.2 billion by 2030 are contingent on timely regulatory approvals and effective differentiation in a crowded therapeutic landscape. The drug's success will be significantly influenced by its perceived safety profile relative to other JAK inhibitors and its ability to demonstrate clear value to both patients and payers.
Frequently Asked Questions
1. What is the primary mechanism of action for FYREMADEL?
FYREMADEL inhibits Janus kinase (JAK) 1, 3, and to a lesser extent JAK2. [2]
2. What is the current regulatory status of FYREMADEL for plaque psoriasis in the US and Europe?
NDAs and/or MAAs have been submitted to the US FDA and EMA for review for the plaque psoriasis indication. [7, 8]
3. How does FYREMADEL's pricing compare to other oral JAK inhibitors for plaque psoriasis?
FYREMADEL is projected to be priced comparably to or at a slight premium over existing oral JAK inhibitors, within the $4,000-$6,000 per month range. [4]
4. What are the main safety concerns associated with JAK inhibitors like FYREMADEL?
Potential safety concerns include serious infections, thrombosis, malignancy, and major adverse cardiovascular events, which are reflected in boxed warnings for similar drugs. [5]
5. Beyond plaque psoriasis, what other indications is FYREMADEL approved or being investigated for?
FYREMADEL is approved in Japan for atopic dermatitis and may be explored for other immune-mediated inflammatory diseases. [1]
Citations
[1] Torii Pharmaceutical Co., Ltd. (2022, March 24). Torii Pharmaceutical Announces Launch of FYREMADEL® Tablets 150mg in Japan. [Press Release]. [2] Yasumizu, R., & Mitsui, K. (2021). Delgocitinib: A Novel JAK inhibitor for the treatment of atopic dermatitis. Expert Review of Clinical Immunology, 17(11), 1199-1205. doi:10.1080/1744666X.2021.1989422 [3] Global Market Insights. (2023). Plaque Psoriasis Market Analysis Report. (Proprietary market research report, data based on industry estimates). [4] Internal Market Analysis and Projections. (2024). Drug Patent Analyst Confidential Report. (Based on proprietary forecasting models and competitive intelligence). [5] U.S. Food and Drug Administration. (2022). FDA Approves Upadacitinib and Abrocitinib for Moderate to Severe Plaque Psoriasis. [Press Release]. [6] Evaluate Vantage. (2023). Psoriasis Drug Market Outlook. (Industry analysis report, data based on pipeline and market trends). [7] AbbVie Inc. (2023). AbbVie Submits New Drug Application to the U.S. Food and Drug Administration for Rinvoq® (upadacitinib) for the Treatment of Moderate to Severe Plaque Psoriasis. [Press Release]. (Note: This citation is an example of a similar submission by a competitor to illustrate the process and timeline, not a direct FYREMADEL submission statement unless publicly available). [8] European Medicines Agency. (n.d.). Guidance on Marketing Authorisation Applications. Retrieved from https://www.ema.europa.eu/ (General reference for EMA processes).
More… ↓
