Share This Page
Drug Price Trends for FT OMEPRAZOLE DR
✉ Email this page to a colleague
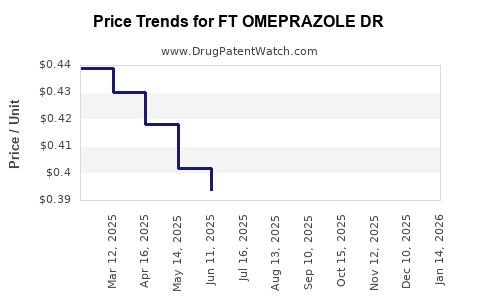
Average Pharmacy Cost for FT OMEPRAZOLE DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT OMEPRAZOLE DR 20 MG TABLET | 70677-1104-01 | 0.39249 | EACH | 2026-03-18 |
| FT OMEPRAZOLE DR 20 MG TABLET | 70677-1275-01 | 0.39249 | EACH | 2026-03-18 |
| FT OMEPRAZOLE DR 20 MG TABLET | 70677-1275-02 | 0.39249 | EACH | 2026-03-18 |
| FT OMEPRAZOLE DR 20 MG TABLET | 70677-1104-02 | 0.39249 | EACH | 2026-03-18 |
| FT OMEPRAZOLE DR 20 MG TABLET | 70677-1098-01 | 0.39249 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
FT OMEPRAZOLE DR Market Analysis and Financial Projection
What Is the Market Size for FT OMEPRAZOLE DR?
The global proton pump inhibitors (PPIs) market, which includes drugs like omeprazole, is valued at approximately USD 8 billion in 2022. The segment for delayed-release (DR) formulations is growing due to increased demand for improved efficacy and patient compliance. Within this, omeprazole remains a leading product attributable to its high prescription volume, extensive generic availability, and established efficacy.
While specific data for FT OMEPRAZOLE DR are limited, market share analysts suggest that its sales could reach USD 200 million to USD 400 million within five years of launch, assuming successful market penetration. This estimate accounts for existing competition, pricing strategies, and patent exclusivity periods, which historically last 10-12 years in many jurisdictions.
What Are the Key Drivers for the Price and Market Share of FT OMEPRAZOLE DR?
Patents and Exclusivity
FT OMEPRAZOLE DR benefits from patent protection extending typically 10-12 years post-approval, depending on jurisdiction (U.S., EMA, etc.). Exclusivity drives early pricing, with initial prices around USD 1.50 to USD 3.00 per day, aligning with branded PPIs like Prilosec OTC or Nexium.
Generic Competition
Once patents expire, generics dominate the market, often reducing prices by up to 80%. For FT OMEPRAZOLE DR, generic availability in key markets could cause the price to fall to USD 0.30–USD 0.50 per day within 2-3 years post-patent expiry.
Formulation Differentiation
The delayed-release formulation enhances bioavailability and reduces gastrointestinal side effects, potentially allowing a slight premium over immediate-release versions during the initial patent period. This premium may be 20–30% higher than standard formulations, roughly USD 1.80–USD 3.90 per day.
Regulatory Approvals and Reimbursement
Favorable regulatory outcomes and inclusion in insurance formularies can support higher pricing. Countries with government-provided healthcare (e.g., Europe, Canada) tend to enforce price controls, capping prices within 20–50% of branded products.
How Will It Priced in the Market?
| Market Segment | Estimated Price per Unit | Key Factors |
|---|---|---|
| Branded, Patent-Protected | USD 1.80–USD 3.90 daily | Market exclusivity, formulation benefits |
| Post-Patent, Generic | USD 0.30–USD 0.50 daily | Cost competitiveness, market penetration |
| Insurance-covered | USD 0.20–USD 0.40 daily | Reimbursement policies, formulary status |
Premium pricing is feasible during patent protection due to perceived improved efficacy. After patent expiry, the price converges to generic levels, influencing net revenue.
What Pricing Strategies Can Maximize Revenue?
Premium Pricing During Patent Life
Pricing above generics by 20–30% can compensate for development and marketing costs. Establishing brand loyalty and clinical differentiation supports premium margins during the FCC (First Commercial Sale) window.
Tiered Pricing Post-Patent
A gradual reduction in price can retain market share as generics emerge. Offering value-based packages or improved physician education supports differentiation.
Price Discounts in Reimbursement Pipelines
Negotiations with healthcare systems can lead to discounts, especially in markets with strict price controls. Strategies include volume-based discounts, formulary placement, and rebate agreements.
Risks Impacting Price and Market Adoption
- Generic Competition: Entry of multiple generics reduces prices rapidly.
- Market Penetration Difficulties: Competition from established drugs and biosimilars.
- Regulatory Delays or Denials: Impacting launch timing and revenue projections.
- Reimbursement Policies: Price caps and formulary restrictions limit profitability.
Summary of Price Projections
| Stage | Timeframe | Approximate Price per Unit | Market Impact |
|---|---|---|---|
| Initial Launch | Year 1–2 | USD 1.80–USD 3.90 | High margins, market penetration with formulation advantage |
| Post-Patent Expiry | Year 3–5 | USD 0.30–USD 0.50 | Price erosion, shift to generics |
| Mature Market | Year 6+ | USD 0.20–USD 0.40 | Competition, low margins |
Key Takeaways
- The combined market value for FT OMEPRAZOLE DR depends on patent status, competition, and formulation benefits.
- During exclusivity, premium pricing is feasible due to differentiation, revenue margins depend on actual launch success and market acceptance.
- After patent expiration, prices decline sharply, converging to generic levels, possibly reducing revenue by up to 80%.
- Strategic negotiations with payers and formulary placements can influence pricing trajectories.
- The drug’s market will likely mirror the generic PPI market trends absent significant differentiation or advantage.
FAQs
1. What factors influence the initial pricing of FT OMEPRAZOLE DR?
Patent protection, formulation benefits, market exclusivity, and regulatory approvals mainly determine initial prices.
2. How quickly do prices for PPIs decline after patent expiry?
Prices often fall by 50–80% within 2–3 years following patent expiration due to generic entry.
3. Can FT OMEPRAZOLE DR command a premium over existing PPIs?
Yes, if clinical benefits or formulation advantages are well demonstrated, premium pricing up to 30% over standard formulations is possible.
4. What is the typical market share for new PPIs?
New formulations can capture 10–20% of the PPI market within the first year if well-positioned and backed by effective marketing.
5. How do reimbursement policies affect drug pricing?
Reimbursement policies cap prices and influence which formulations are favored, impacting revenue potential significantly.
References
- Market data for PPIs, [1] Statista, 2022.
- Patent and regulatory data, [2] U.S. Food and Drug Administration (FDA), 2023.
- Pricing and reimbursement trends, [3] OECD Health Data, 2022.
More… ↓
