Drug Price Trends for FT CLEARLAX POWDER
✉ Email this page to a colleague
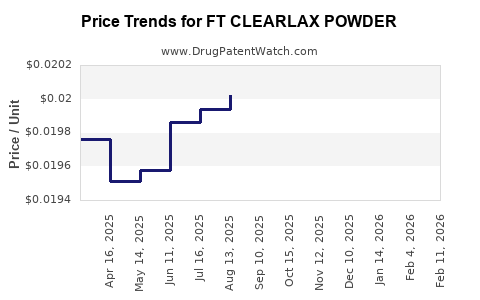
Average Pharmacy Cost for FT CLEARLAX POWDER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT CLEARLAX POWDER | 70677-1068-01 | 0.02074 | GM | 2024-11-20 |
| FT CLEARLAX POWDER | 70677-1068-02 | 0.02080 | GM | 2024-11-20 |
| FT CLEARLAX POWDER | 70677-1109-01 | 0.02936 | GM | 2024-11-20 |
| FT CLEARLAX POWDER | 70677-1068-02 | 0.02042 | GM | 2024-10-23 |
| FT CLEARLAX POWDER | 70677-1109-01 | 0.02896 | GM | 2024-10-23 |
| FT CLEARLAX POWDER | 70677-1068-01 | 0.02662 | GM | 2024-10-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |


