Share This Page
Drug Price Trends for DIPHENOXYLATE-ATROPINE
✉ Email this page to a colleague
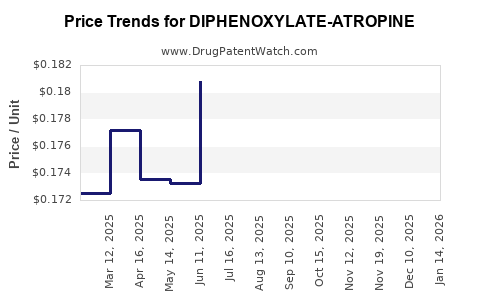
Average Pharmacy Cost for DIPHENOXYLATE-ATROPINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DIPHENOXYLATE-ATROPINE 2.5-0.025 MG TABLET | 00406-1236-10 | 0.13861 | EACH | 2026-05-20 |
| DIPHENOXYLATE-ATROPINE 2.5-0.025 MG TABLET | 60687-0890-01 | 0.13861 | EACH | 2026-05-20 |
| DIPHENOXYLATE-ATROPINE 2.5-0.025 MG TABLET | 60687-0890-11 | 0.13861 | EACH | 2026-05-20 |
| DIPHENOXYLATE-ATROPINE 2.5-0.025 MG TABLET | 59762-1061-01 | 0.13861 | EACH | 2026-05-20 |
| DIPHENOXYLATE-ATROPINE 2.5-0.025 MG TABLET | 76385-0107-10 | 0.13861 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Diphenoxylate-Atropine Market Analysis and Price Projections
Diphenoxylate-atropine, a fixed-dose combination medication primarily used for the symptomatic treatment of diarrhea, faces a competitive landscape shaped by generic penetration and evolving treatment guidelines. The market is characterized by stable, albeit modest, demand driven by its established efficacy and low cost. Future price trajectories will likely be influenced by manufacturing costs, regulatory actions, and the competitive intensity of generic manufacturers.
What is the Current Market Size and Demand for Diphenoxylate-Atropine?
The global market for diphenoxylate-atropine is primarily driven by its use as an antidiarrheal agent. It is available in both prescription and, in some formulations and regions, over-the-counter (OTC) markets. The demand is generally stable, reflecting its role in managing acute diarrheal episodes.
- Key Applications: Symptomatic treatment of diarrhea, including traveler's diarrhea and chronic diarrhea associated with irritable bowel syndrome (IBS).
- Formulations: Oral tablets and liquid solutions.
- Regulatory Status: In the United States, it is a Schedule V controlled substance due to its opioid component (diphenoxylate), requiring a prescription. Atropine is included to discourage abuse. In some other countries, specific formulations may have different regulatory classifications.
- Market Size: Precise global market size figures are not readily available as a distinct segment in broad pharmaceutical market reports, which often group antidiarrheals together. However, based on prescription data and sales of leading generic brands, the annual sales value is estimated to be in the low to mid-hundreds of millions of U.S. dollars globally. This reflects its status as a widely prescribed but lower-cost medication.
Who are the Key Manufacturers and Competitors in the Diphenoxylate-Atropine Market?
The market for diphenoxylate-atropine is largely dominated by generic manufacturers. The original innovator, Lomotil, has seen its market share significantly eroded by competition.
-
Major Generic Manufacturers:
- Amneal Pharmaceuticals
- Teva Pharmaceutical Industries
- Apotex Inc.
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Breckinridge Pharmaceutical (as a supplier to brands)
-
Branded Product:
- Lomotil (G. Pohl-Boskamp GmbH & Co. KG, formerly manufactured by Valeant Pharmaceuticals International, now owned by Sanofi in some regions)
-
Competitive Dynamics: The presence of multiple generic players leads to significant price competition. Competition is based primarily on cost and market access (distribution networks). The manufacturing process for diphenoxylate-atropine is well-established, with no significant technological barriers to entry for generic producers.
What are the Key Factors Influencing Diphenoxylate-Atropine Pricing?
Pricing for diphenoxylate-atropine is influenced by several economic, regulatory, and market-specific factors.
-
Manufacturing Costs:
- Active Pharmaceutical Ingredient (API) Sourcing: The cost of sourcing diphenoxylate hydrochloride and atropine sulfate APIs is a primary cost driver. Fluctuations in the global supply chain for these APIs, particularly from major manufacturing hubs in Asia, can impact production costs.
- Formulation and Packaging: Costs associated with tablet compression, liquid formulation, and packaging (bottles, blister packs) contribute to the final price.
- Labor and Overhead: Manufacturing plant operational costs, including labor, energy, and quality control, are consistent factors.
-
Regulatory Environment:
- Controlled Substance Classification: In the U.S., its Schedule V classification necessitates adherence to strict handling, prescribing, and dispensing regulations, adding compliance costs.
- Good Manufacturing Practices (GMP): Adherence to GMP standards globally is a baseline requirement, with associated auditing and quality assurance costs.
- FDA/EMA Approval Processes: While for generics these are primarily bioequivalence studies, they still represent a significant investment.
-
Market Competition and Distribution:
- Generic Erosion: The high number of generic competitors limits pricing power. Manufacturers must compete on cost to secure contracts with wholesalers, pharmacies, and pharmacy benefit managers (PBMs).
- Wholesaler and Distributor Markups: A portion of the final price reflects the margins of pharmaceutical wholesalers and distributors.
- PBM Formularies and Rebates: PBMs significantly influence drug pricing through formulary placement and negotiated rebates. Diphenoxylate-atropine is often placed on formularies as a low-cost option.
-
Demand Elasticity and Treatment Trends:
- Low Price Sensitivity: Due to its low cost and role as a symptomatic treatment, demand is relatively inelastic to minor price changes. Patients and prescribers will generally opt for the lowest-cost effective option.
- Alternative Treatments: The availability and adoption of alternative antidiarrheal agents (e.g., loperamide, bismuth subsalicylate) and treatments for underlying conditions (e.g., IBS) can indirectly influence demand and pricing power.
What are the Projected Price Trends for Diphenoxylate-Atropine?
Price projections for diphenoxylate-atropine indicate continued stability with potential for minor fluctuations, primarily driven by manufacturing costs and competitive pressures. Significant price increases are unlikely due to its generic nature and established therapeutic niche.
-
Near-Term (1-2 Years):
- Price Stability: Expect prices to remain largely stable. Manufacturers will aim to maintain existing margins, but competitive pressures will prevent substantial increases.
- API Cost Impact: Any significant upward pressure on diphenoxylate or atropine API costs due to supply chain disruptions or increased raw material prices could lead to a modest (1-3%) increase in wholesale prices. Conversely, improved API supply or manufacturing efficiencies could exert downward pressure.
- Generic Entry: While the market is already saturated, the potential for new generic entrants, though less likely for a mature product, could marginally lower prices.
-
Medium-Term (3-5 Years):
- Slight Downward Trend: A gradual downward trend of 1-2% per year is plausible as manufacturing efficiencies are optimized by established generic players and competition persists. This trend assumes no major disruptive events in API sourcing or significant new regulatory hurdles.
- Consolidation: Potential consolidation among smaller generic manufacturers could slightly reduce competitive pressure in specific markets, but broad price increases are still improbable.
- Alternative Therapies: The continued development of novel therapies for IBS or chronic diarrheal conditions could marginally reduce the overall demand for diphenoxylate-atropine, potentially leading to price concessions by manufacturers seeking to maintain market share.
-
Long-Term (5+ Years):
- Continued Price Erosion: Long-term price trends are expected to continue on a slightly downward trajectory, driven by ongoing cost optimization by manufacturers and the persistent availability of multiple generic options.
- Limited Upside Potential: Significant price appreciation is highly unlikely unless there are unforeseen major changes in API manufacturing, a substantial shift in regulatory requirements that favors specific manufacturers, or a significant reduction in the number of active generic producers.
- Impact of Generic Rx Growth: If prescription volumes for generics continue to grow in emerging markets, this could stabilize prices in those regions, though the overall global trend will likely remain modest.
Table 1: Projected Annual Price Change (Wholesale Price Index, US Dollars)
| Time Horizon | Projected Annual Change | Key Influencing Factors |
|---|---|---|
| Near-Term (1-2 Y) | 0% to +1% | API cost stability, existing competitive intensity, PBM negotiations |
| Medium-Term (3-5 Y) | -1% to -2% | Manufacturing efficiencies, sustained generic competition, alternative tx |
| Long-Term (5+ Y) | -1% to -2% | Ongoing cost optimization, market saturation, limited innovation |
Note: Projections are based on current market dynamics and assume no major disruptive events in regulatory policy or API supply chains.
What are the Risks and Opportunities for Diphenoxylate-Atropine Manufacturers?
Manufacturers face a landscape of established risks but also opportunities for strategic market positioning.
Risks
- Intense Generic Competition: The primary risk is the aggressive price competition from numerous generic manufacturers, limiting profit margins.
- API Supply Chain Volatility: Dependence on specific regions or limited suppliers for APIs (diphenoxylate hydrochloride, atropine sulfate) creates vulnerability to geopolitical events, trade disputes, or production disruptions.
- Regulatory Scrutiny: Any changes in the classification of diphenoxylate-atropine as a controlled substance, or increased regulatory requirements for manufacturing or distribution, could increase compliance costs and operational complexity.
- Evolving Treatment Paradigms: Development of superior or more targeted therapies for diarrhea or underlying conditions (e.g., IBS) could lead to a decline in demand for diphenoxylate-atropine.
- Counterfeit Products: The market for low-cost generics can be susceptible to the emergence of counterfeit products, which can damage brand reputation and consumer trust.
Opportunities
- Emerging Market Expansion: Growing healthcare access and increasing prevalence of gastrointestinal issues in emerging economies present an opportunity for market penetration with cost-effective diphenoxylate-atropine formulations.
- Cost Optimization: Continuous improvement in manufacturing processes and supply chain management can lead to further cost reductions, enhancing competitiveness and potentially widening profit margins within the low-price segment.
- Market Access and PBM Contracts: Securing favorable formulary placement and long-term contracts with PBMs and large pharmacy chains can provide a stable revenue stream and market share.
- Supply Chain Reliability: Manufacturers who can demonstrate consistent and reliable API sourcing and robust supply chain management can differentiate themselves from competitors facing potential disruptions.
- Brand Loyalty (for established generics): While branded Lomotil holds legacy recognition, established generic brands can build loyalty among prescribers and pharmacists through consistent quality and competitive pricing.
Key Takeaways
Diphenoxylate-atropine remains a stable, low-cost antidiarrheal agent with demand primarily met by generic manufacturers. The market is characterized by intense price competition, which is projected to continue, leading to modest price stability with a slight downward trend over the medium to long term. Key influencing factors on pricing include API sourcing costs, manufacturing efficiencies, and the dynamics of the generic pharmaceutical market. Manufacturers face risks from competition and supply chain volatility but can capitalize on opportunities in emerging markets and through robust supply chain management.
FAQs
-
What is the typical price range for a prescription of diphenoxylate-atropine? The wholesale acquisition cost for a bottle of 20 tablets (e.g., 2.5 mg diphenoxylate/0.025 mg atropine) typically ranges from $15 to $40, depending on the manufacturer, distributor, and contract terms. Patient out-of-pocket costs will vary based on insurance coverage, co-pays, and pharmacy markups.
-
How does the controlled substance classification of diphenoxylate-atropine in the U.S. impact its availability and cost? As a Schedule V controlled substance, its prescription requires specific DEA registration for prescribers and dispensers. This adds administrative and compliance costs for manufacturers and pharmacies, but these costs are generally absorbed within the existing competitive pricing structure and do not typically lead to significantly higher retail prices compared to non-controlled antidiarrheals.
-
Are there significant differences in pricing between branded Lomotil and its generic equivalents? Yes, branded Lomotil is generally priced higher than generic diphenoxylate-atropine. The price difference can be substantial, often several times the cost of generic options, reflecting brand recognition, marketing, and patent exclusivity periods that have long since expired.
-
What are the primary challenges in securing consistent supply of diphenoxylate-atropine APIs? The primary challenges include the concentration of API manufacturers in specific geographic regions (often Asia), potential for geopolitical instability or trade restrictions affecting these regions, and fluctuations in raw material costs for API synthesis. Ensuring diversification of API suppliers is a key mitigation strategy for manufacturers.
-
Could the development of new generic manufacturers significantly alter diphenoxylate-atropine pricing in the short term? While new generic entrants can introduce price pressure, the market for diphenoxylate-atropine is already highly competitive with numerous established players. The impact of a new entrant on overall pricing in the short term is likely to be marginal (a few percentage points) unless they possess a significant cost advantage or a novel manufacturing process.
Citations
[1] U.S. Drug Enforcement Administration. (n.d.). Schedules of Controlled Substances. Retrieved from [DEA website]
[2] Market research reports on gastrointestinal drugs (e.g., Global Market Insights, Grand View Research - specific report titles vary and are proprietary).
[3] Pharmaceutical industry trade publications and news outlets (e.g., Fierce Pharma, Endpoints News, Pharma Intelligence).
[4] Company annual reports and investor presentations of major generic manufacturers.
[5] Drug pricing databases (e.g., Red Book, First Databank - subscription required).
More… ↓
