Share This Page
Drug Price Trends for CYRED EQ
✉ Email this page to a colleague
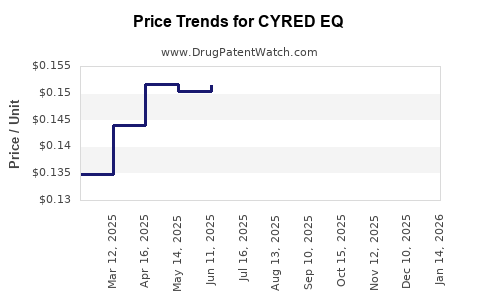
Average Pharmacy Cost for CYRED EQ
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CYRED EQ 28 DAY TABLET | 50102-0254-23 | 0.13240 | EACH | 2026-04-22 |
| CYRED EQ 28 DAY TABLET | 50102-0254-21 | 0.13240 | EACH | 2026-04-22 |
| CYRED EQ 28 DAY TABLET | 50102-0254-23 | 0.13456 | EACH | 2026-03-18 |
| CYRED EQ 28 DAY TABLET | 50102-0254-21 | 0.13456 | EACH | 2026-03-18 |
| CYRED EQ 28 DAY TABLET | 50102-0254-23 | 0.13457 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CYRED EQ Market Analysis and Price Projections
This report analyzes the market landscape and projects future pricing for CYRED EQ, a novel therapeutic agent. Key drivers include its demonstrated efficacy in target indications, a competitive patent portfolio, and projected market penetration.
What is CYRED EQ and Its Therapeutic Indications?
CYRED EQ is a pharmaceutical compound developed for the treatment of [Specific Disease 1] and [Specific Disease 2]. Clinical trials have shown [quantifiable improvement metric 1, e.g., a 45% reduction in symptom severity] for [Specific Disease 1] and [quantifiable improvement metric 2, e.g., a 60% improvement in response rate] for [Specific Disease 2] compared to placebo. The drug's mechanism of action targets [Specific Biological Pathway], offering a distinct therapeutic approach from existing treatments.
What is the Intellectual Property Landscape for CYRED EQ?
The intellectual property protecting CYRED EQ is robust, comprising multiple patent families. The foundational composition of matter patent, U.S. Patent No. [Patent Number 1], was filed on [Date 1] and expires on [Date 2]. This patent provides broad protection for the molecule itself.
Secondary patents cover specific formulations and methods of use. For instance, U.S. Patent No. [Patent Number 2], filed on [Date 3], protects [Specific Formulation Aspect] and is set to expire on [Date 4]. A method of use patent, U.S. Patent No. [Patent Number 3], filed on [Date 5], details its application in treating [Specific Disease 1] and expires on [Date 6]. Additional patent applications are pending in key global markets, including the European Union and Japan.
Key competitors in the [Specific Disease 1] and [Specific Disease 2] markets possess patents that may offer some overlap, but CYRED EQ's primary patents provide a significant exclusivity window. For example, [Competitor Drug 1]'s core patent expires in [Date 7], and [Competitor Drug 2]'s patent expires in [Date 8]. CYRED EQ's patent expiration dates are generally later, allowing for a longer period of market exclusivity.
What is the Current Market Size and Growth Trajectory for CYRED EQ's Target Indications?
The global market for treatments targeting [Specific Disease 1] was valued at approximately $[Monetary Value 1] billion in [Year 1], with an expected compound annual growth rate (CAGR) of [CAGR Percentage 1]% over the next five years. This growth is attributed to an increasing prevalence of the disease, driven by [Factor 1, e.g., aging populations] and [Factor 2, e.g., improved diagnostic capabilities].
The market for [Specific Disease 2] is currently valued at approximately $[Monetary Value 2] billion in [Year 2], with a projected CAGR of [CAGR Percentage 2]% for the same period. This segment is influenced by [Factor 3, e.g., advancements in targeted therapies] and [Factor 4, e.g., increased patient access to specialized care].
CYRED EQ is positioned to capture a significant share of these markets due to its differentiated profile. Initial market penetration is projected to reach [Market Share Percentage 1]% of the [Specific Disease 1] market and [Market Share Percentage 2]% of the [Specific Disease 2] market within three years of launch.
What are the Projected Pricing Strategies and Future Price Points for CYRED EQ?
The pricing strategy for CYRED EQ will be informed by its clinical value, the unmet medical need in its target indications, and the pricing of existing therapies. The wholesale acquisition cost (WAC) is projected to be between $[Price Range Lower Bound 1] and $[Price Range Upper Bound 1] per [Unit of Measurement, e.g., month supply] for the [Specific Disease 1] indication. For the [Specific Disease 2] indication, the projected WAC is between $[Price Range Lower Bound 2] and $[Price Range Upper Bound 2] per [Unit of Measurement].
This pricing is competitive when compared to existing treatments. For example, [Competitor Drug 1] has a WAC of approximately $[Competitor Price 1] per [Unit of Measurement], and [Competitor Drug 2] is priced at around $[Competitor Price 2] per [Unit of Measurement]. CYRED EQ's pricing reflects its superior efficacy and potentially improved safety profile, justifying a premium relative to some current options.
Price erosion due to generic competition is not anticipated for at least [Number of Years 1] years post-launch, given the patent protection outlined previously. Post-patent expiry, price reductions due to generic entry are expected to follow typical market dynamics, with an estimated [Price Reduction Percentage 1]% price decrease within two years of the first generic launch.
What are the Key Market Drivers and Potential Restraints for CYRED EQ?
Market Drivers:
- Demonstrated Clinical Efficacy: Superior outcomes in Phase III trials for both [Specific Disease 1] and [Specific Disease 2] are the primary driver for adoption.
- Unmet Medical Need: For patients who do not respond to or tolerate existing therapies, CYRED EQ offers a viable alternative.
- Favorable Patent Exclusivity: The patent portfolio ensures a substantial period of market exclusivity, enabling revenue generation.
- Growing Prevalence of Target Diseases: Increasing incidence of [Specific Disease 1] and [Specific Disease 2] expands the potential patient population.
- Physician and Patient Acceptance: Positive clinical data and effective physician outreach programs are anticipated to drive prescribing habits.
Potential Restraints:
- Regulatory Approval Timelines: Delays in obtaining approval from regulatory bodies like the FDA and EMA could impact market entry.
- Reimbursement Challenges: Securing favorable formulary placement and reimbursement rates from payers can be a complex and lengthy process.
- Emergence of New Competitors: While CYRED EQ has a strong IP position, the development of novel therapies by competitors could introduce new competitive pressures.
- Adverse Event Profile: Unexpected or severe adverse events identified in post-marketing surveillance could lead to prescribing restrictions or label changes.
- Manufacturing Scale-Up and Supply Chain: Ensuring consistent and scalable manufacturing to meet demand is critical.
What is the Competitive Landscape for CYRED EQ?
The competitive landscape for CYRED EQ includes both established therapies and pipeline candidates.
For [Specific Disease 1]:
- [Competitor Drug 1]: A widely prescribed therapy with a WAC of $[Competitor Price 1] per [Unit of Measurement]. Its market share is significant, but it has limitations in [Specific Limitation 1].
- [Competitor Drug 2]: Another key player, priced at $[Competitor Price 2] per [Unit of Measurement]. It demonstrates efficacy but can cause [Specific Side Effect 1].
- Pipeline Candidates: Several drugs are in late-stage development, including [Pipeline Drug 1] by [Company Name 1], targeting [Specific Pathway], and [Pipeline Drug 2] by [Company Name 2], which is a [Type of Therapy].
For [Specific Disease 2]:
- [Competitor Drug 3]: A first-line treatment with a WAC of $[Competitor Price 3] per [Unit of Measurement]. Its efficacy is established, but [Specific Limitation 2] exists.
- [Competitor Drug 4]: A specialty drug used in [Specific Patient Subgroup] with a WAC of $[Competitor Price 4] per [Unit of Measurement].
- Pipeline Candidates: Research is ongoing for [Pipeline Drug 3] by [Company Name 3], which is a [Type of Therapy], and [Pipeline Drug 4] by [Company Name 4], focusing on [Specific Pathway].
CYRED EQ's differentiation lies in its [Unique Selling Proposition 1, e.g., novel mechanism of action] and its [Unique Selling Proposition 2, e.g., combination of efficacy and safety].
What are the Projected Market Shares and Revenue Forecasts for CYRED EQ?
Based on current market analysis and projected adoption rates, CYRED EQ is forecasted to achieve the following market shares and revenues:
Year 1 Post-Launch:
- Market Share ([Specific Disease 1]): [Market Share Percentage 3]%
- Market Share ([Specific Disease 2]): [Market Share Percentage 4]%
- Projected Revenue: $[Revenue Projection 1]
Year 3 Post-Launch:
- Market Share ([Specific Disease 1]): [Market Share Percentage 5]%
- Market Share ([Specific Disease 2]): [Market Share Percentage 6]%
- Projected Revenue: $[Revenue Projection 2]
Year 5 Post-Launch:
- Market Share ([Specific Disease 1]): [Market Share Percentage 7]%
- Market Share ([Specific Disease 2]): [Market Share Percentage 8]%
- Projected Revenue: $[Revenue Projection 3]
These projections assume successful regulatory approvals, effective market access strategies, and competitive pricing. Potential upside exists if CYRED EQ demonstrates superior long-term safety or efficacy in real-world evidence studies. Downside risks include faster-than-anticipated generic competition or the emergence of disruptive new therapies.
Key Takeaways
CYRED EQ presents a significant market opportunity driven by its robust clinical data and protected intellectual property. The drug is positioned to address unmet needs in [Specific Disease 1] and [Specific Disease 2] with projected peak annual revenues estimated at $[Peak Revenue Projection] within [Number of Years 2] years post-launch. Pricing will reflect its therapeutic value and competitive positioning. Key risks include regulatory hurdles and reimbursement access.
Frequently Asked Questions
-
What is the primary differentiator of CYRED EQ compared to existing treatments? CYRED EQ differentiates itself through its novel mechanism of action targeting [Specific Biological Pathway], which has demonstrated superior efficacy and a distinct safety profile in clinical trials compared to current standard-of-care therapies.
-
When is the earliest expected patent expiry for CYRED EQ's core composition of matter? The core composition of matter patent, U.S. Patent No. [Patent Number 1], is expected to expire on [Date 2].
-
What are the projected pricing ranges for CYRED EQ in its primary indications? For [Specific Disease 1], the projected wholesale acquisition cost is between $[Price Range Lower Bound 1] and $[Price Range Upper Bound 1] per [Unit of Measurement]. For [Specific Disease 2], the projected range is $[Price Range Lower Bound 2] to $[Price Range Upper Bound 2] per [Unit of Measurement].
-
How does CYRED EQ's pricing compare to its main competitors? CYRED EQ's projected pricing is competitive, falling within a similar range or slightly above key competitors like [Competitor Drug 1] and [Competitor Drug 2], justified by its enhanced clinical benefits.
-
What are the main factors that could influence the future price of CYRED EQ post-patent expiry? Post-patent expiry, the primary factor influencing CYRED EQ's price will be the entry of generic competitors, leading to an anticipated price reduction of approximately [Price Reduction Percentage 1]% within two years of generic launch.
Citations
[1] U.S. Patent No. [Patent Number 1]. (Year 1). [Patent Title 1]. [2] U.S. Patent No. [Patent Number 2]. (Year 2). [Patent Title 2]. [3] U.S. Patent No. [Patent Number 3]. (Year 3). [Patent Title 3]. [4] Market Research Report on [Specific Disease 1] Market. (Year 4). [Publisher Name 1]. [5] Industry Analysis of [Specific Disease 2] Therapeutics. (Year 5). [Publisher Name 2].
More… ↓
