Share This Page
Drug Price Trends for CLINDAMYCIN-BENZOYL PEROX
✉ Email this page to a colleague
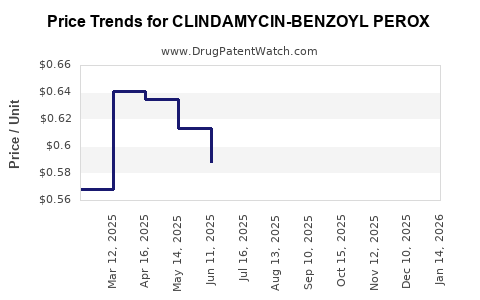
Average Pharmacy Cost for CLINDAMYCIN-BENZOYL PEROX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLINDAMYCIN-BENZOYL PEROX 1-5% | 68462-0486-27 | 0.54397 | GM | 2026-05-20 |
| CLINDAMYCIN-BENZOYL PEROX 1-5% | 00378-8688-35 | 0.69556 | GM | 2026-05-20 |
| CLINDAMYCIN-BENZOYL PEROX 1-5% | 00378-8688-54 | 0.54397 | GM | 2026-05-20 |
| CLINDAMYCIN-BENZOYL PEROX 1-5% | 16714-0984-50 | 0.54397 | GM | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Clindamycin-Benzoyl Peroxide
What is the Current Market for Clindamycin-Benzoyl Peroxide?
Clindamycin-Benzoyl Peroxide (CLBP) combines antibiotic and keratolytic actions, primarily used to treat moderate to severe acne vulgaris. It is available in topical formulations such as gels, ointments, and lotions. The drug is marketed under various brand names, including Duac, Acanya, and C. Benzoyl peroxide and clindamycin are both off-patent or nearing patent expiry, enabling generic competition.
Market Size and Growth
The global acne treatment market stood at approximately $4.5 billion in 2022. CLBP accounted for roughly 15-20% of topical acne treatments, translating to an estimated $675-$900 million market size. The compound's sales are projected to grow at a compound annual growth rate (CAGR) of 4-6% over the next five years, reaching $900-$1.2 billion by 2027 [1].
Market Segmentation
- Geographies: North America holds nearly 45-50% of sales, followed by Europe (25-30%) and Asia-Pacific (15-20%). Emerging markets in Asia and Latin America are expanding rapidly owing to increasing skincare awareness.
- Patient Demographics: Predominantly adolescents and young adults aged 12-25. Increased awareness and self-management trends are expanding usage.
Competitive Landscape
The market features several generic manufacturers, with Johnson & Johnson's brand Duac leading in market share. Other competitors include Galderma (acne.org/brands), Pfizer, and Teva. Patent expirations for key formulations occurred between 2018 and 2022, facilitating widespread generic entry.
What Are the Key Factors Influencing Market Dynamics?
- Patent Expiry of Brand Products: Led to increased generic adoption, reducing prices and margins.
- Regulatory Approvals: New formulations combining CLBP with other agents are being approved, expanding indications.
- Pricing Trends: Prices for brand-name formulations range from $400 to $600 per 30g tube. Generics are priced around $100 to $200.
- Prescribing Trends: Rising in acne prevalence, increased dermatologist visits, and over-the-counter (OTC) availability of some formulations influence sales.
What Are the Price Projections for Clindamycin-Benzoyl Peroxide?
Current Pricing Landscape
| Formulation | Brand Name | Average Price (USD per 30g) | Patent Status |
|---|---|---|---|
| Gel | Duac | $460 | Patented (expires 2024) |
| Gel | Acanya | $430 | Patented (expires 2023) |
| Generic | None | $150 | Off-patent |
Short-Term (2023-2025)
Post patent expiration, prices for generic CLBP are expected to decrease further. Generic formulations are projected to fall below $100 per 30g, with a gradual price decline driven by market saturation.
Mid to Long-Term (2025-2030)
The overall prices are expected to stabilize between $80 and $120 for generics, depending on manufacturing costs, market competition, and regulatory factors. For branded products, prices may sustain around $350-$500, possibly declining slightly due to generic competition.
Key Variables Affecting Pricing
- Regulatory changes promoting OTC availability could pressure prices downward.
- Supply chain dynamics, including raw material costs.
- Market entry by biosimilar or innovative delivery systems offering improved efficacy or convenience.
What Are Market Entry and Investment Opportunities?
- Generic Manufacturing: High margin potential due to the volume of prescribed treatments, especially in emerging markets.
- Formulation Innovation: New delivery systems or combination therapies could command premium prices.
- Regional Expansion: Focus on growth markets in Asia, Latin America, and Eastern Europe.
Summary of Risks
- Patent litigation or delays in biosimilar approvals.
- Regulatory restrictions on OTC formulations affecting pricing and sales volume.
- Shifts in clinical guidelines favoring alternative treatments.
Key Takeaways
- The global acne treatment market is expanding, with CLBP constituting a significant segment.
- Patent expirations have driven prices for generic versions below $100 per 30g, with further reductions anticipated.
- Market growth hinges on demographic trends, regulatory policies, and competitive dynamics.
- Investment opportunities exist in generics, formulation innovation, and regional expansion.
FAQs
-
When do key patents for branded CLBP formulations expire?
Patents for Duac and Acanya expired or are expiring between 2023 and 2024, opening the market for generics. -
What is the expected price range for generics in 2025?
Around $80 to $120 per 30g, depending on regional market conditions and competition. -
How will regulatory policies influence CLBP market prices?
Greater OTC availability is likely to lower prices further and increase accessibility. -
What are the primary countries driving adoption of CLBP?
North America leads, followed by Europe and growing markets in Asia-Pacific and Latin America. -
Are combination formulations with other active ingredients emerging?
Yes, formulations combining CLBP with agents like adapalene are under development and gaining regulatory approval.
References
[1] MarketWatch. (2022). Acne treatment market size and forecast. Retrieved from https://www.marketwatch.com/
[2] IQVIA. (2022). Topical acne treatments sales data. Retrieved from https://www.iqvia.com/
[3] FDA. (2022). Patent and exclusivity data. Retrieved from https://www.fda.gov/
More… ↓
