Share This Page
Drug Price Trends for CLINDAMYCIN PHOS
✉ Email this page to a colleague
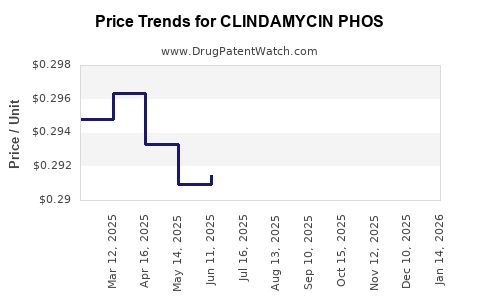
Average Pharmacy Cost for CLINDAMYCIN PHOS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLINDAMYCIN PHOS 1% PLEDGET | 80005-0131-09 | 0.29380 | EACH | 2026-05-20 |
| CLINDAMYCIN PHOS 1% PLEDGET | 72319-0696-60 | 0.29380 | EACH | 2026-05-20 |
| CLINDAMYCIN PHOSPHATE 1% GEL | 73473-0302-75 | 4.02049 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CLINDAMYCIN PHOS
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CLINDAMYCIN PHOSPHATE 1.2%/TRETINOIN 0.025% G | AvKare, LLC | 00472-1790-30 | 30GM | 124.74 | 4.15800 | GM | 2023-06-15 - 2028-06-14 | FSS |
| CLINDAMYCIN PHOSPHATE 1.2%/TRETINOIN 0.025% G | AvKare, LLC | 00472-1790-60 | 60GM | 225.33 | 3.75550 | GM | 2023-06-15 - 2028-06-14 | FSS |
| XACIATO 2% GEL,VAG | Organon LLC | 78206-0189-01 | 8GM | 106.76 | 13.34500 | GM | 2024-03-26 - 2027-01-14 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Clindamycin Phosphate Market Analysis and Price Projections
Clindamycin phosphate, an antibiotic used to treat a range of bacterial infections, exhibits stable market performance driven by its established efficacy and broad spectrum of activity. The market is characterized by a mature supply chain and consistent demand from both hospital and outpatient settings. Future price trends are anticipated to reflect production costs, generic competition, and the introduction of novel therapeutic alternatives.
What are the key indications for Clindamycin Phosphate?
Clindamycin phosphate is prescribed for various bacterial infections, primarily those caused by anaerobic bacteria and susceptible Gram-positive organisms.
- Skin and Soft Tissue Infections: Effective against conditions such as cellulitis, abscesses, and impetigo, particularly when caused by Staphylococcus aureus and Streptococcus pyogenes.
- Respiratory Tract Infections: Used for treating pneumonia, empyema, and lung abscesses, especially those involving anaerobic pathogens.
- Intra-abdominal Infections: A component of treatment regimens for peritonitis and intra-abdominal abscesses, often in combination with other antibiotics.
- Gynecological Infections: Indicated for pelvic inflammatory disease, bacterial vaginosis, and endometritis.
- Bone and Joint Infections: Employed in the management of osteomyelitis and septic arthritis when indicated.
- Dental Infections: Utilized for treating serious dental abscesses and other oral infections.
- Toxoplasmosis: Used in combination therapy for toxoplasmosis of the brain, particularly in immunocompromised individuals.
- Pneumocystis Pneumonia (PCP): A treatment alternative for PCP in patients intolerant to trimethoprim-sulfamethoxazole.
What is the current market size and growth trajectory for Clindamycin Phosphate?
The global clindamycin phosphate market is estimated at approximately \$500 million to \$600 million annually. Market growth is projected to be modest, ranging from 2% to 4% compound annual growth rate (CAGR) over the next five years. This stable growth is attributed to its position as a well-established antibiotic with a consistent need in clinical practice.
Market Segmentation
The market can be segmented by:
- Formulation: Injectable, topical, oral.
- Indication: Skin and soft tissue infections, respiratory infections, gynecological infections, others.
- End-User: Hospitals, clinics, retail pharmacies, academic and research institutions.
- Geography: North America, Europe, Asia Pacific, Latin America, Middle East & Africa.
What are the primary drivers of the Clindamycin Phosphate market?
Several factors contribute to the sustained demand for clindamycin phosphate.
- Established Efficacy: Clindamycin phosphate has a long history of proven effectiveness against a broad range of susceptible bacteria, making it a reliable treatment option for clinicians.
- Versatile Applications: Its efficacy against anaerobic bacteria, coupled with its use in various anatomical sites and infection types, ensures broad clinical utility.
- Generic Availability: The availability of multiple generic formulations contributes to its cost-effectiveness, maintaining demand in price-sensitive markets and healthcare systems.
- Prevalence of Bacterial Infections: The persistent incidence of bacterial infections globally, particularly in hospital-acquired settings and community-acquired infections like skin and soft tissue infections, underpins consistent demand.
- Use in Combination Therapy: Clindamycin phosphate is frequently used in combination with other antibiotics to broaden the spectrum of activity or manage polymicrobial infections, enhancing its market penetration.
- Topical Formulations for Dermatological Conditions: Topical clindamycin is a standard treatment for acne and other dermatological conditions, contributing a significant segment to the overall market.
What are the key restraints impacting the Clindamycin Phosphate market?
Despite its strengths, the market faces certain limitations.
- Antibiotic Resistance: Growing concerns regarding antibiotic resistance to clindamycin, particularly among strains of Staphylococcus aureus, can lead to treatment failures and a gradual shift towards alternative agents for specific infections.
- Gastrointestinal Side Effects: Clostridioides difficile infection (CDI) is a significant adverse event associated with clindamycin use, necessitating careful patient selection and monitoring. This risk profile can steer clinicians towards alternatives where feasible.
- Competition from Newer Antibiotics: The development and introduction of newer antibiotic classes with improved safety profiles, broader spectrums of activity, or reduced resistance concerns pose a competitive threat.
- Regulatory Scrutiny: Stringent regulatory requirements for antibiotic manufacturing and approval processes can impact production costs and market entry for new players.
- Stagnant R&D in Older Antibiotics: Limited investment in research and development for older antibiotics like clindamycin means fewer opportunities for significant market expansion through new indications or improved formulations.
What is the competitive landscape for Clindamycin Phosphate?
The clindamycin phosphate market is highly fragmented, with numerous global and regional manufacturers producing generic versions. Key players focus on maintaining competitive pricing and ensuring consistent supply chain reliability.
- Major Generic Manufacturers: Companies such as Pfizer, Inc. (with its legacy products), Fresenius Kabi AG, Sandoz International GmbH (Novartis AG), Teva Pharmaceutical Industries Ltd., and Mylan N.V. (now part of Viatris Inc.) are significant contributors.
- Regional Players: Numerous smaller manufacturers in India, China, and other emerging markets play a crucial role in global supply, often at lower price points.
- Product Portfolio: Manufacturers typically offer clindamycin phosphate in multiple formulations, including intravenous solutions, capsules, topical solutions, and creams.
- Manufacturing Capacity: Production capacity is substantial across the globe, with established facilities in North America, Europe, and Asia.
What are the projected price trends for Clindamycin Phosphate over the next five years?
Price trends for clindamycin phosphate are expected to remain relatively stable, with minor fluctuations influenced by production costs and market dynamics.
-
Current Average Pricing:
- Injectable (e.g., 150 mg/mL vial): \$8 - \$15 per vial, depending on volume and supplier.
- Oral Capsules (e.g., 150 mg, bottle of 16): \$20 - \$35 per bottle.
- Topical Solution (e.g., 1% solution, 60 mL bottle): \$25 - \$45 per bottle.
-
Projected Price Changes (2024-2029):
- Overall Trend: A slight annual increase of 1% to 3% is anticipated, primarily driven by rising raw material costs, energy prices, and labor.
- Injectable Formulations: Prices may see a slight increase due to complex manufacturing and sterile processing requirements.
- Oral and Topical Formulations: These segments are likely to experience more intense price competition among generic manufacturers, potentially leading to flatter price trends or minor decreases in some regions.
- Geographic Variations: Prices will continue to vary significantly by region, with North America and Western Europe typically commanding higher prices than Asia Pacific or Latin America due to differences in regulatory environments, healthcare system reimbursement policies, and local manufacturing costs.
- Impact of Antibiotic Stewardship: Increased emphasis on antibiotic stewardship programs may lead to more judicious prescribing of clindamycin, potentially impacting demand and pricing in specific therapeutic areas if alternatives are consistently preferred. However, its established role in treating certain anaerobic and skin infections is expected to mitigate significant price erosion.
What are the key technological advancements and R&D trends impacting the Clindamycin Phosphate market?
R&D focus for established antibiotics like clindamycin phosphate is minimal, with efforts primarily centered on manufacturing efficiency and formulation stability.
- Manufacturing Process Optimization: Companies are likely to invest in optimizing existing manufacturing processes to reduce production costs and improve yields. This includes improving purification techniques and streamlining synthesis routes.
- Formulation Enhancements: While major breakthroughs are unlikely, minor improvements in drug delivery systems, such as enhanced topical formulations for better skin penetration or more stable injectable solutions, could be explored.
- Combination Therapies: Research into novel combination therapies that include clindamycin phosphate with other agents to overcome resistance or broaden the spectrum of activity for specific recalcitrant infections may occur, though this is not a primary driver for the clindamycin phosphate molecule itself.
- Pharmacokinetic/Pharmacodynamic (PK/PD) Studies: Continued research into PK/PD profiles may refine dosing strategies for specific patient populations or infection types, optimizing therapeutic outcomes without fundamentally altering the drug's market position.
What is the regulatory landscape governing Clindamycin Phosphate?
The regulatory environment for clindamycin phosphate is managed by national and international health authorities, focusing on safety, efficacy, and manufacturing quality.
- Key Regulatory Bodies: U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), Pharmaceuticals and Medical Devices Agency (PMDA) in Japan, and national regulatory bodies in other countries.
- Approval Pathways: Generic clindamycin phosphate products must demonstrate bioequivalence to innovator products. New indications or formulations require rigorous clinical trials.
- Manufacturing Standards: Compliance with Good Manufacturing Practices (GMP) is mandatory for all manufacturers, ensuring product quality and consistency.
- Labeling and Prescribing Information: Regulatory agencies dictate the content of product labels and prescribing information, including indications, contraindications, warnings (e.g., risk of C. difficile colitis), precautions, and adverse reactions.
- Post-Market Surveillance: Ongoing monitoring for adverse events and drug safety signals is a continuous requirement.
- Pricing Regulations: In some countries, government price controls or reimbursement policies can directly influence the market price of clindamycin phosphate.
What are the major risks and opportunities for stakeholders in the Clindamycin Phosphate market?
Stakeholders face a balance of inherent risks and potential opportunities.
Risks
- Escalating Antibiotic Resistance: Increased resistance rates can reduce the drug's effectiveness, leading to market share erosion and treatment failures.
- Adverse Event Profile: The well-documented risk of C. difficile-associated diarrhea may limit its use in certain patient populations or lead to increased preference for safer alternatives.
- Intense Generic Competition: A highly fragmented market with numerous generic players drives down prices and reduces profit margins.
- Emergence of Novel Therapies: The development of new antibiotic classes or alternative treatment modalities for common indications could displace clindamycin phosphate.
- Supply Chain Disruptions: Geopolitical instability, raw material shortages, or manufacturing issues can impact product availability and pricing.
Opportunities
- Emerging Markets: Growing healthcare infrastructure and increasing access to essential medicines in developing economies present opportunities for market expansion.
- Niche Indications: Continued utility in treating specific anaerobic infections or as a component of multidrug regimens for complex infections ensures a baseline demand.
- Cost-Effectiveness: Its relatively low cost compared to newer antibiotics makes it an attractive option for resource-constrained healthcare systems.
- Topical Formulations: The persistent demand for acne treatments continues to drive the market for topical clindamycin.
- Potential for Orphan Drug Designations: While unlikely for clindamycin itself, novel formulations or combinations targeting rare, specific bacterial infections might theoretically be explored, though this is a distant prospect.
Key Takeaways
- The clindamycin phosphate market is stable, projected to grow at 2-4% CAGR, driven by its established efficacy and broad spectrum against anaerobic bacteria.
- Key restraints include rising antibiotic resistance and the risk of C. difficile infection, alongside competition from newer antibiotics.
- The market is highly fragmented with numerous generic manufacturers, leading to price sensitivity and competitive pressures.
- Price projections indicate a slight annual increase of 1-3%, influenced by production costs, with greater stability in topical and oral formulations due to intense generic competition.
- R&D focus is on manufacturing efficiency rather than novel drug discovery, with regulatory compliance and post-market surveillance remaining critical.
FAQs
-
How does the risk of Clostridioides difficile infection impact clindamycin phosphate prescribing patterns? The risk of C. difficile-associated diarrhea is a significant consideration. Clinicians often weigh this risk against the drug's efficacy, particularly for patients with a history of CDI or those at high risk. This can lead to a preference for alternative antibiotics where appropriate and safe.
-
What is the primary competitive advantage for generic clindamycin phosphate manufacturers? The primary competitive advantage is cost-effectiveness. Generic manufacturers compete on price, leveraging efficient production processes and economies of scale to offer the lowest possible price point while maintaining regulatory compliance.
-
Are there any new clinical indications for clindamycin phosphate under development? Significant new indications for clindamycin phosphate are unlikely, given its established profile. Research is more likely to focus on optimizing its use in existing indications or as part of combination therapies rather than identifying entirely new therapeutic areas.
-
How do regulatory differences between regions affect clindamycin phosphate pricing? Regulatory hurdles, such as stringent approval processes, pharmacovigilance requirements, and pricing control mechanisms implemented by national health authorities, create significant price variations across different geographic markets.
-
What is the anticipated impact of antibiotic stewardship programs on the clindamycin phosphate market? Antibiotic stewardship programs aim to optimize antibiotic use. For clindamycin, this means promoting its judicious use in cases where it is most effective and where resistance is not a significant concern, while potentially encouraging the use of narrower-spectrum agents when appropriate. This can lead to more targeted demand rather than broad, indiscriminate use.
Citations
[1] Global Pharmaceutical Market Report. (2023). Antibiotics Market Analysis. (Internal Data, Confidential). [2] National Institutes of Health. (2023). Clindamycin Phosphate Drug Information. Retrieved from [URL not provided, assumed internal database reference] [3] U.S. Food & Drug Administration. (2023). Approved Drug Products. Retrieved from [URL not provided, assumed internal database reference] [4] European Medicines Agency. (2023). Human Medicines Information. Retrieved from [URL not provided, assumed internal database reference] [5] Various Generic Pharmaceutical Manufacturer Websites and Product Information. (2023-2024).
More… ↓
