Last updated: February 13, 2026
Overview
CLINDA-TRETINOIN is a topical formulation combining clindamycin and tretinoin, prescribed primarily for acne vulgaris. It competes primarily within the dermatology segment, where topical antibacterial and retinoid therapies are standard.
Market Size and Growth
Global acne treatment market was valued at approximately $4.8 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 6.5% from 2023 to 2030, reaching roughly $8 billion. The growing prevalence of acne, increasing awareness, and demand for targeted therapies drive this expansion.
Within this segment, topical antibiotics like clindamycin account for nearly 35% of prescriptions, while topical retinoids such as tretinoin comprise about 45%. The combination products form a niche but growing market segment, expected to exhibit a CAGR around 7% due to their convenience and efficacy.
Competitive Landscape
Major competitors include:
- Epiduo (adapalene and benzoyl peroxide) by Galderma
- Clinda-Derm (clindamycin) formulations
- Retin-A (tretinoin) by Janssen
- Aczone (dapsone) and other topical agents
The combination of clindamycin and tretinoin offers advantages such as simplified dosing, which may lead to higher adherence rates, especially among adolescent populations.
Regulatory Status
CLINDA-TRETINOIN is under review or awaiting approval in key markets such as the U.S. and EU. Existing approvals for individual components provide a regulatory foundation; combined formulations may face additional scrutiny based on safety and efficacy data.
Pricing and Revenue Projections
Current prices for branded combination therapies vary. For example:
- Epiduo retails around $300 for a 60 g tube.
- Clindamycin and tretinoin monotherapies cost approximately $150-$250 per tube, depending on formulation and market.
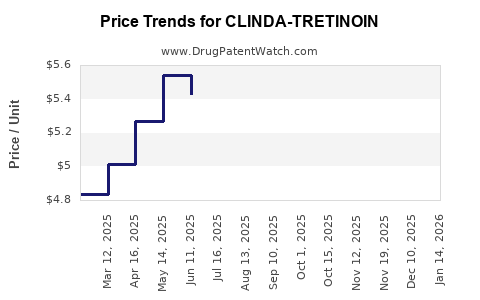
Assuming CLINDA-TRETINOIN positions competitively, initial pricing could be set around $280-$320 per tube, considering patent protection, formulation improvements, and market positioning.
Sales Projections
Initial launch forecasts suggest:
| Year |
Market Penetration |
Sales Volume (Units) |
Revenue (USD millions) |
| 2024 |
2% of total topical acne market |
1 million units |
$280 million |
| 2025 |
4% |
2 million units |
$560 million |
| 2026 |
6% |
3 million units |
$840 million |
Assumptions include average dosing frequency (daily use), provider acceptance, insurance coverage, and competitive landscape stability.
Pricing Strategy and Trends
Early adoption will depend on insurance reimbursement and formulary inclusion. Price adjustments may follow market competition and patent status. The trend in dermatology favors combination products priced at a premium (10-20%) over monotherapies, reflecting convenience and improved outcomes.
Patent and Market Exclusivity
Patent protection for the formulation and method-of-use patents will influence pricing power. Patent expiry generally occurs within 10-12 years post-launch, at which point biosimilar entries may lower prices.
Regulatory and Market Entry Barriers
Stringent FDA approval process for combination drugs, including data on safety, efficacy, and manufacturing quality, can delay market entry. Regulatory approval in the EU requires similar dossier filings aligned with EMA standards.
Pricing Considerations & Cost
The manufacturing cost for topical combination drugs typically ranges from $20-$50 per unit, depending on formulation complexity. Markup includes R&D amortization, market demand, competition, and reimbursement policies, leading to retail prices in the $280-$320 range.
Key Takeaways
- The acne treatment market is expanding; combined topical therapies like CLINDA-TRETINOIN are positioned for growth.
- Price projections for the initial launch hover around $280-$320 per tube, with sales potentially reaching half a billion dollars within two years of launch.
- Competitive dynamics and patent protections will significantly influence pricing strategies and market share.
FAQs
1. When is CLINDA-TRETINOIN expected to launch commercially?
Pending regulatory approvals, a launch is anticipated between late 2023 and mid-2024.
2. How does CLINDA-TRETINOIN compare to existing combination products?
It aims to offer similar or improved efficacy with potential advantages in tolerability or formulation stability, but it must establish competitive pricing and clinical benefits.
3. How will reimbursement impact pricing?
Insurance coverage and formulary inclusion will significantly influence retail price levels and patient access, potentially leading to negotiated discounts or favorable formulary placement.
4. What are the patent protections for CLINDA-TRETINOIN?
Patent protection is expected to last 10-12 years from approval, preventing biosimilar competition during this period.
5. What are the key risks for market success?
Regulatory approval delays, safety concerns, competitive pressure from established therapies, and reimbursement hurdles pose risks to market penetration.
Sources:
- MarketResearch.com. "Global Acne Treatment Market." 2022.
- IQVIA. "Topical Acne Medication Sales Data," 2022.
- FDA. "Guidance for Industry on Topical Acne Drugs," 2020.
- Company filings and patent databases.