Share This Page
Drug Price Trends for CALAMINE PLUS
✉ Email this page to a colleague
Average Pharmacy Cost for CALAMINE PLUS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
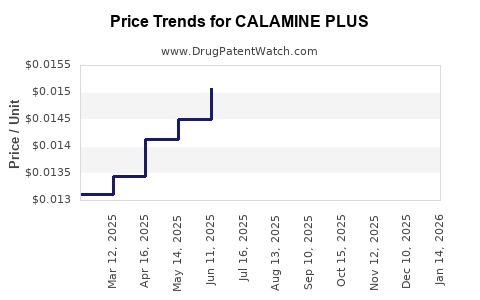
| CALAMINE PLUS 1%-8% LOTION | 70000-0400-01 | 0.01613 | ML | 2026-04-22 |
| CALAMINE PLUS 1%-8% LOTION | 70000-0400-01 | 0.01611 | ML | 2026-03-18 |
| CALAMINE PLUS 1%-8% LOTION | 70000-0400-01 | 0.01618 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
mmary
Calamine Plus is a topical dermatological product combining calamine, zinc oxide, and possibly other soothing agents. It primarily targets relief from itch, irritation, and minor skin conditions such as insect bites, rashes, and sunburns. Market analysis indicates a steady demand driven by increasing consumer awareness and expanding OTC skincare categories. Price projections show moderate growth with regional variations influenced by manufacturing costs, regulatory frameworks, and competitive pressures.
What Is Calamine Plus and Its Market Position?
Calamine Plus is a topical formulation used for dermatological relief. Its core active ingredient, calamine, has anti-itch properties and is combined with zinc oxide, known for protecting and healing skin.
- Active Ingredients: Calamine (zinc carbonate and ferric oxide), zinc oxide.
- Indications: Itching, irritation, minor burns, dermatitis.
- Formulation: Creams, lotions, powders.
Estimates suggest annual sales exceeding $200 million globally, with dominance in OTC markets in North America and Europe[1].
What Are the Current Market Trends Affecting Calamine Plus?
- Growth in OTC Skincare: Increasing consumer preference enables OTC sales, especially in urban markets.
- Rising Allergic and Skin Conditions: Growing incidence of allergic reactions and skin conditions supports demand.
- Regulatory Environment: Stringent regulations, especially in the European Union, may impact formulations and marketing.
The global OTC dermatological market exceeded $15 billion in 2022, expected to grow at a compound annual growth rate (CAGR) of approximately 5% through 2027[2].
What Are the Competitive Dynamics and Key Players?
Major competitors include:
- Johnson & Johnson: Offers products like Caladryl, combining calamine with other soothing agents.
- Bayer: Produces calamine-based antipruritic solutions.
- Generic manufacturers: Increasing pressure on price points.
Market shares are dispersed; top brands hold approximately 40% combined, with generics accounting for more than 50%.
What Are the Price Trends and Projection Factors?
Current Pricing Landscape:
- United States: Average retail price per 4 oz. tube ranges from $3 to $7.
- Europe: Price points vary; in Germany and UK, around €4-€6 per 100g pack.
- Emerging Markets: Lower prices, often below $2 per unit, driven by local manufacturing.
Price Drivers:
- Raw material costs (zinc oxide, ferric oxide).
- Distribution channels (retail vs. online).
- Regulatory costs and approval processes.
- Brand loyalty and patent status.
Forecasted Price Trends (2023-2028):
- Moderate increase in developed markets: 1-3% annually due to inflation and ingredient costs.
- Potential decline in generic segments: Price competition could push prices down by 2-4%.
- Premium formulations: Could command a 10-15% premium if enhanced with additional soothing agents or organic certifications.
Impact of Regulation:
Increased scrutiny of OTC formulations in certain regions may lead to formulation adjustments, potentially impacting cost and pricing strategies.
What Are the Entry and Expansion Opportunities?
- Gaining market share through product differentiation, such as organic ingredients or dual-purpose formulations.
- Entry into emerging markets via local manufacturing and distribution partnerships.
- Digital marketing campaigns targeting consumers seeking quick relief solutions.
What Are the Challenges and Risks?
- Regulatory hurdles delaying product launches.
- Price erosion from generic competition.
- Changes in consumer preferences toward natural or organic products.
- Supply chain disruptions impacting raw material prices.
Key Takeaways
- Calamine Plus occupies a stable niche within the topical OTC dermatology sector.
- Market demand is expected to grow steadily, with growth rates aligned with OTC dermatology CAGR (around 5%).
- Pricing is influenced by raw material costs, competition, and regional regulations, with moderate upward trends in developed regions.
- Competitive pressure necessitates innovation and strategic marketing, especially in emerging markets.
- Regulatory compliance and supply chain stability are critical factors influencing future pricing and market presence.
FAQs
1. How does the price of Calamine Plus compare internationally?
Prices are higher in developed markets like the US and Europe, averaging $3-$7 per 4 oz. tube, while emerging markets often price below $2 per unit due to lower manufacturing and distribution costs.
2. What factors could influence the future growth of Calamine Plus?
Market growth depends on consumer awareness, regulatory approval processes, competition from generics, and innovation in formulation.
3. Are there any legal or regulatory challenges facing Calamine Plus?
Regulatory approvals vary by region. In some markets, ingredient restrictions and labeling requirements can delay or restrict sales.
4. What are the main competitive advantages for brands offering Calamine Plus?
Brand recognition, formulation quality, and distribution reach. Innovation, such as organic certifications, can also create differentiation.
5. Will pricing continue to decline due to generic competition?
Potentially, especially in commoditized segments. Lack of differentiation could lead to year-on-year price decreases of 2-4%.
Citations
[1] MarketWatch, "Topical Dermatitis Markets," 2022.
[2] Grand View Research, "OTC Dermatology Market Size," 2022.
More… ↓
