Share This Page
Drug Price Trends for ATENOLOL-CHLORTHALIDONE
✉ Email this page to a colleague
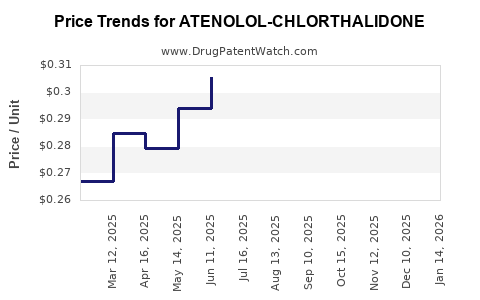
Average Pharmacy Cost for ATENOLOL-CHLORTHALIDONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ATENOLOL-CHLORTHALIDONE 100-25 | 16714-0937-01 | 0.33213 | EACH | 2026-05-20 |
| ATENOLOL-CHLORTHALIDONE 100-25 | 70710-1168-01 | 0.33213 | EACH | 2026-05-20 |
| ATENOLOL-CHLORTHALIDONE 100-25 | 70954-0391-10 | 0.33213 | EACH | 2026-05-20 |
| ATENOLOL-CHLORTHALIDONE 100-25 | 29300-0401-01 | 0.33213 | EACH | 2026-05-20 |
| ATENOLOL-CHLORTHALIDONE 50-25 | 70954-0390-10 | 0.26277 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is the market landscape for atenolol-chlorthalidone?
Atenolol-chlorthalidone is a fixed-dose combination treatment primarily used for hypertension management. The drug combines a beta-blocker (atenolol) with a thiazide-like diuretic (chlorthalidone). Though it has been on the market for decades, the combination's overall market share has declined with the advent of newer antihypertensives.
How large is the current market for combination antihypertensive drugs?
The global antihypertensive drugs market was valued at approximately $34.7 billion in 2021. Fixed-dose combinations (FDCs), including atenolol-chlorthalidone, account for approximately 10-12% of this market, reflecting slower growth relative to monotherapies.
Regional distribution shows North America holding around 35% of the FDC market, driven by high hypertension prevalence and favorable reimbursement systems. Europe accounts for approximately 25%. Emerging markets, including China and India, increasingly adopt FDCs, driven by cost and compliance benefits.
What are the price trends for atenolol-chlorthalidone?
Pricing for branded and generic formulations varies globally. In the United States, generic atenolol-chlorthalidone costs approximately $4–$10 per month, depending on the supplier and pharmacy discounts. Branded versions tend to cost 30–50% more. In European markets, prices are similar, adjusted for local healthcare systems.
In emerging markets, prices can drop below $1 per month for generic versions, but access and quality standards vary. Price trends show a gradual decline over the last decade, driven by increased competition among generics and patent expirations.
What is the patent status and regulatory environment?
Atenolol-chlorthalidone's primary patents expired in the early 2000s, leading to widespread generic manufacturing. No recent patents restrict manufacturing or marketing, leading to a competitive landscape with numerous suppliers.
Regulatory approvals for generic versions are well-established in major markets, with approvals based on bioequivalence. Some recent regulatory moves focus on safety monitoring and post-market surveillance due to concerns about the long-term safety profile of certain antihypertensives.
What are the factors influencing future market prospects?
- Clinical guidelines:** Most guidelines favor ARBs and ACE inhibitors over beta-blocker-based FDCs for initial hypertension treatment, reducing the potential for growth.
- Safety profile: Concerns such as the use of atenolol in certain populations impact prescribing patterns.
- Market penetration: Generic availability continues to suppress prices, limiting profit margins.
- Combination therapy trends: An increase in newer fixed-dose formulations with improved tolerability and dosing convenience may cannibalize demand.
How will price projections evolve?
Based on current trends, prices for generic atenolol-chlorthalidone are projected to decline approximately 3-5% annually over the next five years in mature markets, assuming continued market saturation and competitive pressure.
In emerging markets, price declines could be steeper, around 5-8%, due to increasing manufacturing capacity and regulatory interventions to reduce drug costs.
The entry of new antihypertensive combination therapies with improved safety and tolerability might further constrain pricing and market share.
Summary table of market data and projections
| Metric | 2021 | 2026 (Projected) |
|---|---|---|
| Global antihypertensive market valuation | $34.7 billion | ~$45 billion |
| Fixed-dose combination segment share | 11% | 13-14% |
| Average price in developed markets | $4–$10/month | $3–$8/month |
| Price decline rate (annual) | — | 3–5% (mature markets) |
| Price decline rate (emerging markets) | — | 5–8% |
Key takeaways
- The market for atenolol-chlorthalidone is mature, with declining prices due to patent expirations and patent competition.
- Use of this combination is decreasing in favor of newer antihypertensives, especially in guidelines-driven markets.
- Price declines are expected to persist, with generic competition remaining a core factor.
- Growth potential is limited outside of markets with low drug penetration or in regions adopting cost-effective FDCs.
- Market dynamics suggest no significant price increase opportunities barring new regulatory or clinical developments.
FAQs
1. Is atenolol-chlorthalidone still a recommended first-line option?
No. Most guidelines prioritize ARBs and ACE inhibitors due to better safety profiles, leading to reduced use of atenolol-based FDCs.
2. How does the safety profile affect market prospects?
Concerns about atenolol's efficacy and safety in certain populations (e.g., diabetics, elderly) limit its usage and growth potential.
3. Will patent protections return for this drug combination?
Unlikely. Patents expired decades ago, with only minor formulations or method-of-use patents possibly remaining, which do not substantially impact market exclusivity.
4. What are the implications for drug manufacturers?
Manufacturers face pressure to lower prices, shift focus toward newer therapies, or develop improved formulations to maintain profitability.
5. How might regulatory changes influence prices?
Policy efforts to reduce drug costs, especially in emerging markets, could accelerate price declines for generic formulations.
Citations
- MarketsandMarkets. "Antihypertensive Drugs Market by Drug Class," 2022.
- IQVIA. "Global Market Trends in Hypertension Medications," 2021.
- FDA. "Approved Drug Products with Therapeutic Equivalence Evaluations," 2022.
- IMS Health. "Generic Drug Market Share and Pricing Trends," 2022.
- European Medicines Agency. "Regulatory Guidelines on Fixed-Dose Combinations," 2022.
More… ↓
