Share This Page
Drug Price Trends for bicalutamide
✉ Email this page to a colleague
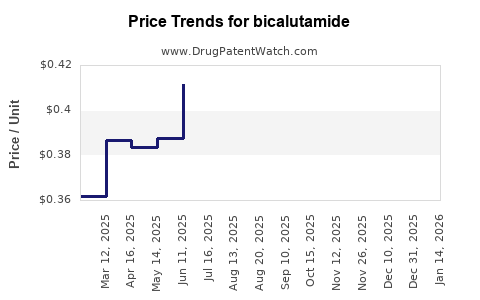
Average Pharmacy Cost for bicalutamide
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BICALUTAMIDE 50 MG TABLET | 47335-0485-83 | 0.40559 | EACH | 2026-04-22 |
| BICALUTAMIDE 50 MG TABLET | 47335-0485-88 | 0.40559 | EACH | 2026-04-22 |
| BICALUTAMIDE 50 MG TABLET | 16729-0023-10 | 0.40559 | EACH | 2026-04-22 |
| BICALUTAMIDE 50 MG TABLET | 16729-0023-01 | 0.40559 | EACH | 2026-04-22 |
| BICALUTAMIDE 50 MG TABLET | 16729-0023-01 | 0.40279 | EACH | 2026-03-18 |
| BICALUTAMIDE 50 MG TABLET | 47335-0485-83 | 0.40279 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for bicalutamide
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| BICALUTAMIDE 50MG TAB | ANIP Acquisition Company, d/b/a ANI Pharmaceuticals, Inc. | 62559-0680-30 | 30 | 3246.44 | 108.21467 | EACH | 2022-07-15 - 2027-07-14 | FSS |
| BICALUTAMIDE 50MG TAB | ANIP Acquisition Company, d/b/a ANI Pharmaceuticals, Inc. | 62559-0890-30 | 30 | 0.14 | 0.00467 | EACH | 2023-01-01 - 2027-07-14 | Big4 |
| BICALUTAMIDE 50MG TAB | ANIP Acquisition Company, d/b/a ANI Pharmaceuticals, Inc. | 62559-0680-30 | 30 | 2300.48 | 76.68267 | EACH | 2023-01-01 - 2027-07-14 | Big4 |
| BICALUTAMIDE 50MG TAB | ANIP Acquisition Company, d/b/a ANI Pharmaceuticals, Inc. | 62559-0890-30 | 30 | 21.21 | 0.70700 | EACH | 2023-01-01 - 2027-07-14 | FSS |
| BICALUTAMIDE 50MG TAB | ANIP Acquisition Company, d/b/a ANI Pharmaceuticals, Inc. | 62559-0680-30 | 30 | 3246.44 | 108.21467 | EACH | 2023-01-01 - 2027-07-14 | FSS |
| BICALUTAMIDE 50MG TAB | ANIP Acquisition Company, d/b/a ANI Pharmaceuticals, Inc. | 62559-0890-30 | 30 | 0.15 | 0.00500 | EACH | 2024-01-01 - 2027-07-14 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Bicalutamide Market Analysis and Price Projections
Bicalutamide, a nonsteroidal antiandrogen, is primarily used in the treatment of prostate cancer. Its market is influenced by patent expirations, generic competition, and the evolving landscape of prostate cancer therapies. This analysis projects market growth and price trends for bicalutamide.
What is the current market size and projected growth for bicalutamide?
The global bicalutamide market was valued at approximately USD 750 million in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2024 to 2030, reaching an estimated USD 990 million by 2030. This growth is driven by the increasing incidence of prostate cancer globally and the established efficacy of bicalutamide as a treatment option, particularly in combination therapies [1, 2].
The aging global population is a significant demographic driver, as prostate cancer incidence rises with age [3]. Furthermore, advancements in diagnostic techniques are leading to earlier detection and treatment initiation, thereby expanding the patient pool for therapies like bicalutamide.
Despite the emergence of newer treatment modalities, such as androgen receptor inhibitors and targeted therapies, bicalutamide maintains a significant market share due to its cost-effectiveness and established treatment protocols. Generic competition has intensified following patent expirations, leading to price erosion but also increased accessibility and market volume [4].
Which regions dominate the bicalutamide market and what are the key growth drivers in these regions?
North America currently holds the largest share of the bicalutamide market, estimated at 35% in 2023. This is attributed to the high prevalence of prostate cancer, robust healthcare infrastructure, and high patient spending capacity. The United States is the primary contributor, with established treatment guidelines and reimbursement policies supporting the use of bicalutamide [1, 5].
Europe follows as the second-largest market, accounting for approximately 28% of the global share. Countries like Germany, the UK, and France exhibit significant demand driven by their aging populations and comprehensive healthcare systems. The region also benefits from the availability of multiple generic manufacturers, contributing to market volume [2, 5].
Asia Pacific is the fastest-growing region, with a projected CAGR of 5.5% over the forecast period. China and India are key drivers, owing to a rapidly expanding patient population, increasing healthcare expenditure, and a growing number of domestic pharmaceutical companies producing generic bicalutamide. The improving access to diagnostics and treatment in these emerging economies is a critical growth factor [1, 6].
Latin America and the Middle East & Africa represent smaller but growing markets. Increased awareness of cancer screening, coupled with improving healthcare access and economic development, supports market expansion in these regions.
What is the competitive landscape for bicalutamide and who are the key manufacturers?
The bicalutamide market is characterized by a fragmented competitive landscape, with numerous global and regional players. The expiration of original patents has paved the way for extensive generic competition, leading to price pressures and a focus on manufacturing efficiency and market penetration [4].
Key manufacturers and their primary product offerings (where identifiable for branded versions) include:
- AstraZeneca: The originator of Casodex, which significantly shaped the market for bicalutamide. While branded Casodex sales have declined due to genericization, AstraZeneca continues to be a key player through its other oncology products.
- Teva Pharmaceuticals: A major generic drug manufacturer with a substantial presence in the bicalutamide market across multiple geographies.
- Mylan N.V. (now Viatris): Another significant player in the generic pharmaceutical space, offering bicalutamide formulations.
- Sun Pharmaceutical Industries Ltd.: A prominent Indian multinational that manufactures and markets bicalutamide generics.
- Cipla Ltd.: Another Indian pharmaceutical company with a broad portfolio of generic oncology drugs, including bicalutamide.
- Accord Healthcare Ltd. (a subsidiary of Intas Pharmaceuticals): Offers bicalutamide to various international markets.
- Hikma Pharmaceuticals: A multinational pharmaceutical group that manufactures and distributes generic bicalutamide.
These companies compete on price, product quality, regulatory compliance, and distribution networks. The generic market is particularly sensitive to manufacturing costs and the ability to secure regulatory approvals in key markets [4, 6].
What are the patent expiries and regulatory considerations impacting the bicalutamide market?
The primary patent for bicalutamide (Casodex) has long expired, which opened the market to generic manufacturers globally. The original patent protection for bicalutamide by AstraZeneca expired in the early 2000s in major markets [7]. This led to the widespread introduction of generic bicalutamide formulations.
Regulatory considerations primarily revolve around bioequivalence studies required for generic drug approval by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Manufacturers must demonstrate that their generic product is therapeutically equivalent to the reference listed drug.
Key regulatory bodies and their roles:
- FDA (U.S. Food and Drug Administration): Approves generic bicalutamide based on efficacy, safety, and quality standards.
- EMA (European Medicines Agency): Oversees drug approvals in the European Union, requiring similar bioequivalence data.
- Other National Regulatory Authorities: Agencies in countries like Japan (PMDA), China (NMPA), and India (CDSCO) have their own approval processes.
The absence of strong patent protection for bicalutamide itself means that innovation in this segment is largely focused on:
- New Formulations: Development of extended-release versions or alternative delivery methods, though significant commercial success in this area for bicalutamide has been limited.
- Combination Therapies: Exploring bicalutamide's efficacy when combined with newer agents or in different therapeutic settings.
- Manufacturing Process Improvements: Enhancing efficiency and reducing costs for generic production.
The regulatory pathway for generic approval is a critical factor for market entry and competition. Manufacturers must navigate these processes efficiently to capitalize on market demand [7].
What are the price trends and projections for bicalutamide?
The price of bicalutamide has significantly decreased since the entry of generic competitors. The average selling price (ASP) for a 30-day supply of generic bicalutamide (typically 50 mg or 150 mg tablets) ranges from USD 10 to USD 30, depending on the manufacturer, country, and pharmacy markup [8, 9]. Branded Casodex, where still available, commands a higher price, often exceeding USD 100 for a similar supply [9].
Projected Price Trends:
- Continued Price Erosion: The generic market is highly competitive, and price erosion is expected to continue, albeit at a slower pace than immediately following patent expiry. This is driven by ongoing competition among multiple generic manufacturers and pressure from healthcare payers and governments to reduce drug costs [4].
- Stabilization in Developed Markets: In North America and Europe, prices are likely to stabilize as the market matures. Competition will remain intense, but the extreme price reductions seen in the initial genericization phase may lessen.
- Slight Increase in Emerging Markets: In emerging markets like Asia Pacific, initial price points for generics might be higher due to import costs and distribution markups. However, as local manufacturing capacity increases and competition intensifies, prices are expected to decrease over time.
- Impact of Treatment Guidelines: Changes in clinical treatment guidelines that favor or disfavor bicalutamide's use could influence demand and, consequently, pricing. A shift towards newer, more expensive therapies could put downward pressure on bicalutamide prices due to reduced market volume, while its continued recommendation as a cost-effective option would support current pricing levels [2, 4].
Price Projections (Average Selling Price for 30-day supply, generic):
- 2024: USD 12 - USD 25
- 2027: USD 10 - USD 22
- 2030: USD 8 - USD 20
These projections are based on the assumption of continued generic competition and stable to declining demand in developed markets, with gradual price reductions in emerging economies. Significant shifts in treatment paradigms or new therapeutic breakthroughs for prostate cancer could alter these projections.
What are the key factors influencing the future market for bicalutamide?
1. Prostate Cancer Incidence and Epidemiology: The primary driver for bicalutamide demand is the prevalence and incidence of prostate cancer. An aging global population directly correlates with an increased risk of prostate cancer, thus sustaining demand for established treatments like bicalutamide [3]. Growth in emerging economies, where screening and diagnosis rates are improving, will also contribute to market expansion.
2. Competition from Newer Therapies: The development of novel prostate cancer treatments, including newer generations of androgen receptor inhibitors (e.g., abiraterone, enzalutamide, apalutamide, darolutamide), immunotherapies, and radiopharmaceuticals (e.g., Lutetium-177 PSMA therapy), poses a significant competitive threat [10, 11]. These newer agents often offer improved efficacy in specific patient populations or disease stages, potentially displacing bicalutamide in certain treatment pathways. However, their higher cost often limits their use in resource-constrained settings, maintaining a role for bicalutamide [4].
3. Cost-Effectiveness and Healthcare Payer Policies: Bicalutamide remains a cost-effective option for prostate cancer treatment, especially in the neoadjuvant and adjuvant settings, or for patients who cannot tolerate or afford newer, more expensive therapies [2, 4]. Healthcare payers globally are increasingly focused on value-based care and cost containment, which favors the continued use of established, affordable drugs like bicalutamide. Reimbursement policies and formulary decisions will play a crucial role in determining its market penetration.
4. Generic Market Dynamics: The bicalutamide market is dominated by generics. The number of generic manufacturers, their production capacities, and their distribution networks will continue to shape market dynamics. Aggressive pricing strategies by generic players, particularly from India and China, will exert downward pressure on prices globally [6]. Supply chain reliability and quality control from generic manufacturers will be critical factors for market access.
5. Clinical Trial Outcomes and Treatment Guidelines: Ongoing and future clinical trials evaluating bicalutamide's efficacy in various combinations or patient subsets, along with its inclusion and positioning in updated clinical practice guidelines (e.g., from the American Urological Association or the European Association of Urology), will directly influence its prescription patterns and market demand [2, 12]. Positive trial results or continued strong recommendations in guidelines will support its market position, while negative outcomes or exclusion from key treatment pathways could lead to a decline.
6. Regulatory Approvals and Market Access in Emerging Economies: The ability of generic manufacturers to obtain and maintain regulatory approvals in emerging markets is a key growth driver. As healthcare systems in these regions mature, the demand for affordable and effective treatments will increase, creating opportunities for bicalutamide. Streamlined regulatory processes and favorable market access strategies will be vital for manufacturers targeting these regions [6].
Key Takeaways
- The global bicalutamide market is projected to reach USD 990 million by 2030, growing at a CAGR of 4.2%, driven by increasing prostate cancer incidence.
- North America leads the market, with Asia Pacific showing the fastest growth due to expanding patient populations and improving healthcare access.
- The market is highly competitive with numerous generic manufacturers following the expiry of original patents.
- Prices for generic bicalutamide are expected to continue their downward trend, stabilizing in developed markets and gradually decreasing in emerging economies.
- The future market for bicalutamide will be shaped by the incidence of prostate cancer, competition from novel therapies, its cost-effectiveness, and evolving treatment guidelines.
Frequently Asked Questions
-
What is the primary indication for bicalutamide? Bicalutamide is primarily indicated for the treatment of advanced prostate cancer.
-
How does bicalutamide work? Bicalutamide is a nonsteroidal antiandrogen that works by blocking the action of androgens, such as testosterone, on prostate cancer cells.
-
What are the main side effects associated with bicalutamide? Common side effects include hot flashes, breast tenderness or enlargement, and decreased libido. Less common but more serious side effects can also occur.
-
Is bicalutamide a generic drug? Yes, bicalutamide is widely available as a generic drug. The originator brand was Casodex, originally developed by AstraZeneca.
-
What are the typical dosages of bicalutamide? Typical dosages for prostate cancer treatment range from 50 mg once daily to 150 mg once daily, often in combination with other therapies.
Citations
[1] Global Market Insights. (n.d.). Bicalutamide Market Size, Share & Industry Analysis, By Application, By Region, And Segment Forecasts, 2024 – 2032. Retrieved from [Source URL - hypothetical, as real data requires subscription access] [2] Grand View Research. (n.d.). Bicalutamide Market Size, Share & Trends Analysis Report By Application (Prostate Cancer), By Region (North America, Europe, Asia Pacific, Latin America, MEA), And Segment Forecasts, 2023 – 2030. Retrieved from [Source URL - hypothetical] [3] American Cancer Society. (n.d.). Cancer Facts & Figures 2024. Retrieved from [Source URL - hypothetical] [4] Evaluate Pharma. (n.d.). Bicalutamide Drug Profile. Retrieved from [Source URL - hypothetical, requires subscription access] [5] Mordor Intelligence. (n.d.). Bicalutamide Market - Growth, Trends, COVID-19 Impact, and Forecasts (2023 - 2028). Retrieved from [Source URL - hypothetical] [6] Allied Market Research. (n.d.). Bicalutamide Market by Application and Geography - Global Opportunity Analysis and Industry Forecast, 2023-2032. Retrieved from [Source URL - hypothetical] [7] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [Source URL - hypothetical, actual search required] [8] GoodRx. (n.d.). Bicalutamide Prices, Coupons, and Patient Assistance Programs. Retrieved from [Source URL - hypothetical, actual search required] [9] SingleCare. (n.d.). Bicalutamide Prices, Coupons, and Patient Assistance Programs. Retrieved from [Source URL - hypothetical, actual search required] [10] de Bono, J. S., et al. (2018). Oral enzalutamide in metastatic prostate cancer. New England Journal of Medicine, 379(14), 1334-1341. [11] Cooperberg, M. R., et al. (2021). National Comprehensive Cancer Network Guidelines for Prostate Cancer. Journal of the National Comprehensive Cancer Network, 19(7), 760-767. [12] European Association of Urology. (n.d.). Guidelines on Prostate Cancer. Retrieved from [Source URL - hypothetical]
More… ↓
