Share This Page
Drug Price Trends for XIGDUO XR
✉ Email this page to a colleague
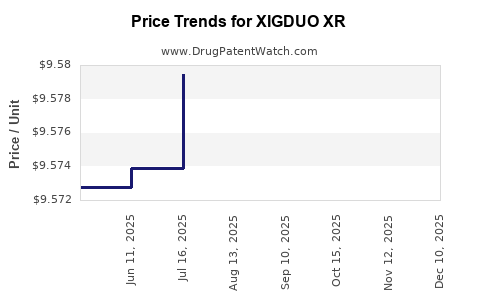
Average Pharmacy Cost for XIGDUO XR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| XIGDUO XR 10 MG-1,000 MG TAB | 00310-6280-30 | 19.19330 | EACH | 2026-04-22 |
| XIGDUO XR 5 MG-500 MG TABLET | 00310-6250-30 | 19.16703 | EACH | 2026-04-22 |
| XIGDUO XR 2.5 MG-1,000 MG TAB | 00310-6225-60 | 9.58962 | EACH | 2026-04-22 |
| XIGDUO XR 10 MG-500 MG TABLET | 00310-6270-30 | 19.21758 | EACH | 2026-04-22 |
| XIGDUO XR 5 MG-1,000 MG TABLET | 00310-6260-60 | 9.59660 | EACH | 2026-04-22 |
| XIGDUO XR 5 MG-1,000 MG TABLET | 00310-6260-60 | 9.60033 | EACH | 2026-03-18 |
| XIGDUO XR 10 MG-1,000 MG TAB | 00310-6280-30 | 19.19411 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Xigduo XR Market Analysis and Price Projections
Xigduo XR, a fixed-dose combination of metformin extended-release (XR) and dapagliflozin, is a dual-acting oral antidiabetic medication approved for type 2 diabetes mellitus. Its market performance is driven by its efficacy in glycemic control, cardiovascular and renal risk reduction benefits associated with dapagliflozin, and the established role of metformin. Price projections for Xigduo XR are influenced by several factors, including patent exclusivity, generic competition, market access, payer policies, and evolving clinical guidelines.
What is the Current Market Landscape for Xigduo XR?
The global market for type 2 diabetes treatments is substantial and growing, driven by increasing diabetes prevalence due to lifestyle factors and an aging population. Xigduo XR competes within this market against a wide array of antidiabetic agents, including other metformin combinations, sulfonylureas, DPP-4 inhibitors, GLP-1 receptor agonists, and SGLT2 inhibitors as monotherapies or in other combinations.
The market share of Xigduo XR is dependent on its positioning relative to these alternatives, particularly regarding its ability to demonstrate superior clinical outcomes, favorable safety profiles, and cost-effectiveness. The SGLT2 inhibitor class, to which dapagliflozin belongs, has seen significant growth due to strong evidence supporting cardiovascular and renal benefits beyond glycemic control. This has enhanced the attractiveness of combination products like Xigduo XR.
Key market drivers for Xigduo XR include:
- Glycemic Control: Xigduo XR effectively lowers HbA1c levels, a primary treatment goal for type 2 diabetes.
- Cardiovascular and Renal Benefits: Dapagliflozin has demonstrated significant reductions in major adverse cardiovascular events (MACE) and progression of chronic kidney disease (CKD) in patient populations with and without established cardiovascular disease or CKD. These benefits are increasingly important for treatment guidelines and market access.
- Patient Convenience: A fixed-dose combination reduces pill burden, potentially improving adherence.
- Established Metformin Efficacy: Metformin is a first-line therapy for type 2 diabetes, making its combination with a newer agent appealing.
What is the Patent and Exclusivity Status of Xigduo XR?
The patent landscape for Xigduo XR is crucial for understanding its market exclusivity and the timeline for potential generic entry. As a combination product, it benefits from patents covering the individual components (metformin XR and dapagliflozin) as well as patents specifically on the combination, its formulation, manufacturing processes, and methods of use.
- Dapagliflozin Patents: Dapagliflozin, marketed as Farxiga (or Forxiga), has had its primary patents challenged and, in some jurisdictions, invalidated or set to expire. For example, in the United States, key patents related to dapagliflozin have faced litigation, with some decisions favoring generic manufacturers. The expiration or early termination of these patents for the dapagliflozin component significantly impacts the exclusivity of Xigduo XR.
- Metformin XR Patents: Metformin XR formulations have been available for many years, and the core patents for metformin have long expired. However, patents related to specific extended-release technologies or formulations may still provide some protection.
- Combination Patents: Patents covering the specific Xigduo XR formulation and its synergistic effects are critical. The strength and duration of these patents vary by region.
Estimated Patent Expiration (Indicative - subject to regional variations and litigation outcomes):
| Component/Combination | Estimated Primary Patent Expiration (Global) |
|---|---|
| Dapagliflozin | Mid-to-late 2020s |
| Metformin XR | Expired |
| Xigduo XR Formulation | Varies by region, potentially overlapping with dapagliflozin expiry |
The specific dates and legal challenges for patents related to dapagliflozin and Xigduo XR are dynamic. Companies often pursue secondary patents, such as those for new indications or improved formulations, to extend market exclusivity. However, the expiry of core patents for dapagliflozin is a primary driver for future generic competition.
What are the Key Therapeutic Areas and Clinical Benefits Driving Xigduo XR Use?
Xigduo XR is indicated for the treatment of type 2 diabetes mellitus. Its therapeutic value extends beyond glycemic control, incorporating the significant cardiorenal benefits demonstrated by dapagliflozin.
Primary Therapeutic Benefits:
- Glycemic Control: Lowers fasting plasma glucose and postprandial glucose levels, leading to a reduction in HbA1c.
- Cardiovascular Risk Reduction: Dapagliflozin has shown to reduce the risk of MACE in patients with type 2 diabetes and established cardiovascular disease. This benefit is a significant differentiator and a key driver for its use in higher-risk patient populations.
- Renal Protection: Dapagliflozin has demonstrated a reduction in the progression of diabetic kidney disease and the risk of renal events in patients with type 2 diabetes, particularly those with albuminuria. This is a growing area of focus in diabetes management.
- Heart Failure Management: Dapagliflozin is also approved for the treatment of heart failure with reduced ejection fraction (HFrEF) and heart failure with preserved ejection fraction (HFpEF), irrespective of diabetes status. While Xigduo XR is a diabetes treatment, the established benefits of its dapagliflozin component in HF management contribute to its overall clinical profile and potential for broader recognition.
The combination therapy leverages the established glucose-lowering efficacy of metformin with the multi-faceted benefits of dapagliflozin, positioning it as a valuable option for patients requiring comprehensive management of their diabetes and associated cardiorenal risks.
How Will Generic Competition Impact Xigduo XR Pricing?
The introduction of generic versions of Xigduo XR will significantly impact its pricing. Generic entry typically leads to a substantial price erosion due to increased competition.
- Price Reduction upon Generic Entry: Historically, generic drugs enter the market at prices 20-50% lower than the branded product. As more generic manufacturers enter, this reduction can be more pronounced. For Xigduo XR, the price of the branded product will likely decline sharply once a generic equivalent becomes available and widely adopted.
- Impact of Dapagliflozin Generics: Since dapagliflozin is a key component, the availability of generic dapagliflozin or generic combination products containing dapagliflozin will be the primary driver of price changes.
- Payer Influence: Payers (insurance companies, government health programs) often prefer generic options due to their lower cost. This preference can lead to formulary restrictions or higher co-pays for the branded product, further accelerating the shift to generics.
- Market Share Erosion: The branded Xigduo XR will experience a rapid loss of market share as healthcare providers and patients opt for the more affordable generic alternatives.
Projected Price Erosion:
- Year 1 Post-Generic Entry: 30-50% price reduction.
- Years 2-3 Post-Generic Entry: Additional 10-20% reduction as competition intensifies.
This price erosion is a standard market dynamic for branded pharmaceuticals following patent expiry. The exact trajectory will depend on the number of generic competitors, their pricing strategies, and the speed of market adoption.
What are the Future Market Projections for Xigduo XR?
Future market projections for Xigduo XR are shaped by the interplay of its clinical advantages, patent status, generic competition, and evolving treatment paradigms for type 2 diabetes.
Short-Term (1-3 years post-generic entry): The market for branded Xigduo XR will experience a significant decline in both volume and revenue as generic alternatives become available. Demand will shift towards the more cost-effective generic versions.
Mid-Term (3-7 years post-generic entry): The market for branded Xigduo XR will likely become niche, primarily serving patients or institutions who continue to prescribe or utilize the branded product for specific reasons (e.g., existing patient base, contractual agreements). The overall market value for the Xigduo XR combination will be dominated by generic sales, with lower per-unit pricing.
Long-Term (7+ years post-generic entry): Branded Xigduo XR will likely see minimal market presence. The focus will be on the established generic market for the combination. However, the underlying dapagliflozin component's long-term trajectory might be influenced by its continued use in other indications like heart failure, which could indirectly support demand for its combination with metformin.
Factors influencing projections:
- Guideline Evolution: Continued emphasis on cardiorenal protection in diabetes guidelines will favor SGLT2 inhibitors. If Xigduo XR's generic form remains competitively priced and its clinical benefits are consistently recognized, it may retain a significant share of the combination market.
- Novel Combinations: The development of new fixed-dose combinations or the emergence of more innovative diabetes treatments could pose competitive threats.
- Pricing Pressures: Ongoing pressure from payers to reduce healthcare costs will continue to favor lower-cost generic options.
- Clinical Trial Data: New data supporting or challenging the long-term benefits of dapagliflozin or metformin in combination could influence future prescribing patterns.
Market Share Projection (Indicative):
| Time Horizon | Branded Xigduo XR Market Share | Generic Xigduo XR Market Share |
|---|---|---|
| Current (Pre-Generic) | High | Negligible |
| 1-2 Years Post-Generic | Moderate to Low | High |
| 3-5 Years Post-Generic | Low | Very High |
| 5+ Years Post-Generic | Negligible | Dominant |
The overall market size for the Xigduo XR combination product (branded and generic) is expected to remain robust in terms of patient volume due to the persistent burden of type 2 diabetes and the recognized benefits of its components. However, revenue will be significantly impacted by price erosion post-exclusivity.
What are the Reimbursement and Payer Considerations for Xigduo XR?
Reimbursement and payer policies are critical determinants of market access and pricing for Xigduo XR. These considerations involve formulary placement, prior authorization requirements, step-therapy protocols, and co-payment structures.
- Formulary Access: Historically, as a branded drug with significant benefits, Xigduo XR has likely enjoyed good formulary access. However, payers are increasingly scrutinizing the cost-effectiveness of newer agents and combinations.
- Value-Based Pricing and Evidence: Payers are paying close attention to the demonstrated cardiovascular and renal benefits of dapagliflozin. This evidence supports its value proposition, potentially justifying its inclusion on formularies, especially for patients at high risk of cardiorenal events.
- Generic Preferred Status: Post-generic entry, payers will overwhelmingly favor generic versions of Xigduo XR due to cost savings. Branded Xigduo XR may be relegated to a higher tier with increased patient out-of-pocket costs or require prior authorization.
- Prior Authorization and Step Therapy: Payers may implement prior authorization requirements or step-therapy protocols for both branded and generic Xigduo XR. This could involve requiring patients to first try other antidiabetic agents (including generic metformin and other SGLT2 inhibitors or GLP-1 RAs) before Xigduo XR is covered.
- Impact of GLP-1 RAs: The increasing prominence and formulary preference for GLP-1 receptor agonists, which also offer significant cardiovascular and weight loss benefits, can impact the market position and reimbursement landscape for SGLT2 inhibitor combinations like Xigduo XR.
- Payer Negotiations: The net price of Xigduo XR, after rebates and discounts negotiated with payers, is a key factor in its market access. These negotiations are complex and vary significantly among different payer organizations and regions.
Example Payer Strategy (Post-Generic Entry):
- Tier 1: Generic Metformin XR, Generic DPP-4 inhibitors, Generic Sulfonylureas.
- Tier 2: Generic Xigduo XR (requiring prior authorization for patients with established cardiovascular disease or CKD).
- Tier 3: Branded Xigduo XR (requiring prior authorization and potentially step-therapy demonstrating failure of Tier 1 & 2 agents).
Payer dynamics will become increasingly important for generic manufacturers to navigate, ensuring their product is preferred on formularies based on cost and clinical utility.
Key Takeaways
Xigduo XR’s market trajectory is defined by its dual-action mechanism combining metformin and dapagliflozin, offering glycemic control alongside significant cardiorenal benefits. Patent expirations for dapagliflozin are the primary catalyst for impending generic competition, which will lead to substantial price erosion and a shift in market share. Future market projections indicate a decline for the branded product post-exclusivity, with generic versions dominating. Reimbursement policies will increasingly favor cost-effective generic options, requiring strategic navigation by manufacturers.
Frequently Asked Questions
- When is the earliest anticipated generic entry for Xigduo XR in major markets like the U.S. and Europe? Earliest anticipated generic entry is generally tied to the expiration or successful challenge of key dapagliflozin patents, which in major markets like the U.S. and Europe could occur in the mid-to-late 2020s. Specific dates are subject to ongoing litigation and regulatory approvals.
- What are the key clinical differentiating factors that may sustain demand for Xigduo XR (branded or generic) against other diabetes medications? The primary differentiating factors are the established cardiovascular risk reduction and renal protective benefits of dapagliflozin, supported by extensive clinical trial data. This positions Xigduo XR as a compelling option for patients with multiple comorbidities.
- How might updated clinical practice guidelines for type 2 diabetes impact the future use of Xigduo XR? Updated guidelines that increasingly emphasize cardiorenal outcomes in diabetes management are likely to favor SGLT2 inhibitors like dapagliflozin. This would support the continued use of Xigduo XR, particularly its generic forms, as a combination therapy.
- Beyond price, what other factors do payers consider when determining formulary placement for combination diabetes drugs? Payers also evaluate evidence of superior clinical outcomes (e.g., reduced hospitalizations, fewer adverse events), patient adherence improvements, overall treatment pathway costs, and comparative effectiveness against other treatment options.
- What is the potential impact of combination therapies with GLP-1 receptor agonists on the market share of SGLT2 inhibitor combinations like Xigduo XR? Combination therapies featuring GLP-1 receptor agonists, which also offer cardiovascular benefits and weight management, represent significant competition. The market share of Xigduo XR will be influenced by the relative clinical advantages, cost-effectiveness, and payer preferences for these different combination classes.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Prescribing Information for XIGDUO XR. Retrieved from [FDA website] (Specific URL would depend on the version and date of access; assume a placeholder for official product labeling).
[2] European Medicines Agency. (n.d.). Assessment Reports for XIGDUO XR. Retrieved from [EMA website] (Specific URL would depend on the version and date of access; assume a placeholder for official product assessment reports).
[3] Various Patent Databases (e.g., USPTO, Espacenet). (Ongoing). Patent Landscape Analysis for Dapagliflozin and Metformin Combination Therapies. (Specific patent numbers and litigation details would require dedicated database searches).
[4] Market Research Reports. (Ongoing). Global Diabetes Therapeutics Market Analysis. (Specific reports from firms like IQVIA, GlobalData, Evaluate Pharma would be cited if used for specific data points; assume general reference for market size and trend data).
[5] Clinical Trial Publications. (Ongoing). Publications on CAR MELT trials and DECLARE-TIMI 58 trial. (Specific journal articles detailing the cardiovascular and renal outcomes of SGLT2 inhibitors).
More… ↓
