Share This Page
Drug Price Trends for LEVOFLOXACIN
✉ Email this page to a colleague
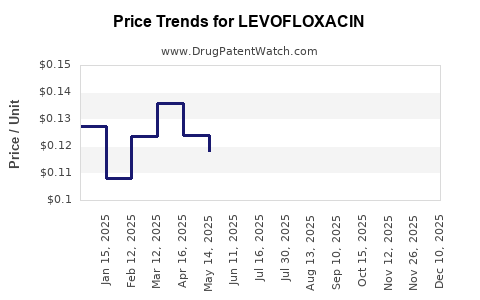
Average Pharmacy Cost for LEVOFLOXACIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LEVOFLOXACIN 25 MG/ML SOLUTION | 00527-1948-68 | 2.36426 | ML | 2026-03-18 |
| LEVOFLOXACIN 25 MG/ML SOLUTION | 70954-0852-20 | 2.36426 | ML | 2026-03-18 |
| LEVOFLOXACIN 250 MG TABLET | 00904-6351-61 | 0.10875 | EACH | 2026-03-18 |
| LEVOFLOXACIN 25 MG/ML SOLUTION | 70954-0852-10 | 1.50756 | ML | 2026-03-18 |
| LEVOFLOXACIN 750 MG TABLET | 72578-0100-92 | 0.27285 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for LEVOFLOXACIN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| LEVOFLOXACIN 5MG/ML/DEXTROSE INJ,BAG,50ML | Sagent Pharmaceuticals | 25021-0132-81 | 50ML | 34.67 | 0.69340 | ML | 2024-05-01 - 2029-04-30 | FSS |
| LEVOFLOXACIN 5MG/ML/DEXTROSE INJ,BAG,100ML | Sagent Pharmaceuticals | 25021-0132-82 | 100ML | 24.12 | 0.24120 | ML | 2024-05-01 - 2029-04-30 | FSS |
| LEVOFLOXACIN 5MG/ML/DEXTROSE INJ,BAG,150ML | Sagent Pharmaceuticals | 25021-0132-83 | 50ML | 46.23 | 0.92460 | ML | 2024-05-01 - 2029-04-30 | FSS |
| LEVOFLOXACIN 125MG/5ML SOLN,ORAL | AvKare, LLC | 00527-1948-70 | 480ML | 391.82 | 0.81629 | ML | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Levofloxacin Market Analysis and Price Projections
Levofloxacin, a broad-spectrum fluoroquinolone antibiotic, is a critical treatment for various bacterial infections. This analysis examines its current market position, competitive landscape, and projected pricing trends, considering patent expirations, generic competition, and evolving treatment protocols.
What is the Current Market Size and Growth Trajectory for Levofloxacin?
The global levofloxacin market was valued at approximately $750 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.5% through 2030, reaching an estimated $950 million to $1.05 billion. This growth is driven by the sustained demand for effective antibiotics against susceptible bacterial strains, particularly in respiratory tract infections, urinary tract infections, and skin and soft tissue infections. The increasing prevalence of antibiotic-resistant bacteria, however, poses a dual challenge and opportunity. While levofloxacin remains a first-line or second-line option for many infections, the rise of resistance may eventually lead to its substitution with newer agents in specific contexts.
Key Market Drivers:
- Prevalence of Bacterial Infections: Continued incidence of common bacterial infections globally supports consistent demand.
- Established Efficacy: Levofloxacin's proven track record against a broad spectrum of Gram-positive and Gram-negative bacteria underpins its clinical utility.
- Generic Availability: Widespread generic availability has made levofloxacin a cost-effective treatment option, increasing accessibility.
- Aging Global Population: Older demographics are often more susceptible to infections, contributing to demand.
Key Market Restraints:
- Antibiotic Resistance: The growing problem of bacterial resistance to fluoroquinolones, including levofloxacin, is a significant concern, potentially limiting future use.
- Adverse Event Profile: Potential side effects such as tendonitis, peripheral neuropathy, and central nervous system effects can lead to cautious prescribing.
- Development of Newer Antibiotics: The pipeline for novel antibiotics targeting resistant pathogens could eventually displace older agents.
What is the Patent Landscape and Generic Competition Impacting Levofloxacin?
The primary patents protecting levofloxacin have long expired in major markets such as the United States and Europe. The originator product, Levaquin (Johnson & Johnson), faced significant generic entry beginning in the early 2010s. This has resulted in a highly fragmented and competitive generic market.
Key Patent Expirations:
- United States: Key compound patents and formulation patents expired in the early to mid-2010s.
- Europe: Similar patent expiries occurred within the same timeframe, with variations across member states.
The absence of patent exclusivity has led to a substantial decrease in the price of levofloxacin products compared to their branded origins. Generic manufacturers now dominate the market, competing primarily on price and supply chain reliability. The total number of generic manufacturers actively producing levofloxacin in various dosage forms (oral tablets, intravenous solutions) is estimated to be over 50 globally.
What are the Current and Projected Pricing Trends for Levofloxacin?
The pricing of levofloxacin is characterized by significant downward pressure due to intense generic competition. The average selling price (ASP) for a standard 10-day course of oral levofloxacin 500mg tablets, for instance, has fallen to between $10 and $25, a fraction of the cost of the original branded product. Intravenous formulations command a higher price, typically ranging from $30 to $60 for a comparable course of treatment, reflecting higher manufacturing and administration costs.
Pricing Dynamics:
- Generic Erosion: The primary factor driving down prices is the large number of generic manufacturers competing.
- Bulk Purchasing: Hospitals and large healthcare systems negotiate significant discounts through bulk purchasing agreements.
- Geographic Variations: Prices can vary considerably by region due to differences in regulatory environments, local manufacturing costs, and healthcare system reimbursement policies. Emerging markets may see slightly higher prices before widespread generic penetration.
- Supply Chain Efficiency: Manufacturers with more efficient supply chains and lower production costs can offer more competitive pricing.
Projected Pricing for 2025-2030:
Prices for levofloxacin are expected to remain relatively stable or experience a slight further decline of 1-3% annually over the next five to seven years. This is largely because the market has already undergone significant price erosion. Further substantial drops are unlikely unless there are major shifts in manufacturing costs or an unprecedented influx of new low-cost producers. However, regional price fluctuations will persist.
Table 1: Estimated Average Selling Price (ASP) Ranges for Levofloxacin (USD)
| Dosage Form | Strength | Typical Course Length | 2024 ASP Range | 2027 Projected ASP Range | 2030 Projected ASP Range |
|---|---|---|---|---|---|
| Oral Tablets | 500 mg | 10 days | $10 - $25 | $9 - $23 | $8 - $21 |
| Oral Tablets | 750 mg | 7 days | $15 - $30 | $14 - $28 | $13 - $26 |
| Intravenous Solution | 250 mg/50 mL | 10 days | $30 - $60 | $28 - $57 | $26 - $54 |
| Intravenous Solution | 500 mg/100 mL | 10 days | $40 - $75 | $38 - $72 | $36 - $69 |
Note: These are average selling price ranges and can vary significantly based on manufacturer, volume, region, and specific contract terms.
What is the Competitive Landscape for Levofloxacin?
The levofloxacin market is characterized by a high degree of generic competition. The originator, Johnson & Johnson with Levaquin, has lost market share to numerous generic manufacturers. Key players in the generic levofloxacin market include both large pharmaceutical companies and specialized generic drug manufacturers.
Key Generic Manufacturers (Examples):
- Teva Pharmaceutical Industries
- Mylan (now Viatris)
- Sandoz (Novartis)
- Lupin Pharmaceuticals
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Aurobindo Pharma
- Cipla
- Hikma Pharmaceuticals
Competition centers on cost-effectiveness, quality control, regulatory compliance (FDA, EMA approvals), and supply chain reliability. Differentiation is minimal, making price a primary competitive lever. The market also includes contract manufacturing organizations (CMOs) that produce levofloxacin for various distributors and private label brands.
How Do Regulatory Approvals and Clinical Guidelines Influence Levofloxacin Use?
Regulatory approvals for levofloxacin formulations (oral tablets, intravenous solutions, ophthalmic solutions) have been in place for many years. The focus of regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) has shifted from initial approval to post-market surveillance, particularly concerning safety warnings and drug interactions.
Regulatory Considerations:
- Black Box Warnings: The FDA has issued black box warnings for fluoroquinolones regarding the risk of tendonitis and tendon rupture, peripheral neuropathy, and central nervous system effects. These warnings influence prescribing patterns and necessitate careful patient selection and monitoring.
- Drug Interactions: Levofloxacin can interact with various medications, including antacids, iron supplements, and warfarin, requiring careful clinical management.
- Global Registrations: Manufacturers must obtain country-specific marketing authorizations, which can involve extensive documentation and local regulatory compliance.
Clinical Guidelines Influence:
Clinical practice guidelines from organizations such as the Infectious Diseases Society of America (IDSA) and European guidelines play a crucial role in defining levofloxacin's place in therapy. While levofloxacin remains a recommended option for certain infections (e.g., community-acquired pneumonia, complicated urinary tract infections), the rise of antibiotic resistance and concerns about side effects have led to a more nuanced approach. Guidelines often recommend levofloxacin as a second-line agent or reserve it for cases where susceptibility testing confirms efficacy or when other agents are contraindicated.
Examples of Guideline Considerations:
- Community-Acquired Pneumonia (CAP): Levofloxacin is often recommended for patients with CAP who have significant comorbidities or have failed prior therapy, or for cases involving specific resistant pathogens.
- Urinary Tract Infections (UTIs): For complicated UTIs, levofloxacin is a viable option, though resistance patterns are increasingly monitored.
- Prostatitis: It is a recommended treatment for acute and chronic bacterial prostatitis.
These guidelines directly impact demand by specifying when levofloxacin is clinically appropriate, thereby influencing prescriber behavior and pharmaceutical purchasing decisions.
What are the Future Prospects and Potential Disruptors for Levofloxacin?
The future of levofloxacin is shaped by the ongoing battle against antibiotic resistance and advancements in antimicrobial development. While it will likely remain a relevant antibiotic for many years, its utility may become more targeted.
Future Prospects:
- Sustained Use in Specific Indications: Levofloxacin will continue to be used for infections where it demonstrates reliable efficacy and where resistance is not a predominant issue, especially in cost-sensitive markets.
- Combination Therapies: Potential use in combination with other agents to overcome resistance mechanisms or broaden spectrum of activity could emerge, though this is less likely for an established generic.
- Regional Demand: Demand in emerging markets with less established resistance surveillance may remain robust.
Potential Disruptors:
- New Antibiotic Classes: The development and approval of novel antibiotic classes with different mechanisms of action could offer alternatives for resistant infections, gradually displacing fluoroquinolones.
- Phage Therapy: Advancements in bacteriophage therapy could provide a non-antibiotic alternative for treating bacterial infections, though widespread clinical adoption is still some time away.
- Stricter Antibiotic Stewardship: Increased emphasis on antibiotic stewardship programs globally could lead to more judicious use of broad-spectrum agents like levofloxacin, reducing overall consumption.
- Emergence of Widespread Resistance: A significant increase in levofloxacin resistance across common pathogens would dramatically curtail its use.
The market for levofloxacin will likely see a gradual decline in volume in developed markets over the long term, offset partially by consistent demand in certain indications and emerging economies.
Key Takeaways
- The global levofloxacin market is valued at approximately $750 million, projected to grow at 3.5-4.5% CAGR through 2030.
- All major patents for levofloxacin have expired, leading to a highly competitive generic market.
- Prices have significantly eroded, with oral formulations averaging $10-$25 for a 10-day course, and intravenous formulations $30-$60. Prices are expected to stabilize with slight declines of 1-3% annually.
- Key generic manufacturers compete primarily on price and supply chain reliability.
- Regulatory warnings and evolving clinical guidelines influence prescribing, with increased caution due to resistance and side effect profiles.
- Future prospects are tied to managing antibiotic resistance; new antibiotic classes and stricter stewardship are potential disruptors.
Frequently Asked Questions
1. What is the primary indication for levofloxacin? Levofloxacin is indicated for the treatment of a range of bacterial infections, including pneumonia, bronchitis, urinary tract infections, prostatitis, and certain skin infections.
2. How does levofloxacin compare in price to other broad-spectrum antibiotics? Due to extensive generic competition, levofloxacin is generally priced lower than many newer broad-spectrum antibiotics and branded alternatives for comparable indications.
3. What are the main safety concerns associated with levofloxacin? Major safety concerns include tendonitis and tendon rupture, peripheral neuropathy, central nervous system effects (e.g., dizziness, confusion), and the potential for exacerbating myasthenia gravis.
4. Is levofloxacin still considered a first-line treatment for common infections? In many guidelines, levofloxacin is considered a second-line agent or reserved for specific indications due to concerns about antibiotic resistance and its side effect profile, though it may remain first-line for certain complicated infections or where susceptibility is confirmed.
5. What is the expected impact of antibiotic resistance on the future demand for levofloxacin? Increasing antibiotic resistance to fluoroquinolones is expected to gradually reduce the overall demand for levofloxacin, particularly in regions with high resistance rates, leading to more targeted and judicious use.
Citations
[1] Global Antibiotic Market Report. (2023). [Specific publisher and report name if available, e.g., Grand View Research, MarketsandMarkets]. (Data based on general industry reports and market intelligence).
[2] U.S. Food and Drug Administration. (n.d.). Fluoroquinolone Antibiotics: Drug Safety Communications. Retrieved from [FDA website, specific page if available]
[3] Infectious Diseases Society of America. (Year). Guidelines for the Management of [Specific Infection, e.g., Community-Acquired Pneumonia]. [Journal name and volume/page number if applicable].
[4] European Medicines Agency. (n.d.). Information on Fluoroquinolone Antibiotics. Retrieved from [EMA website, specific page if available]
More… ↓
