Share This Page
Drug Price Trends for LEVETIRACETAM
✉ Email this page to a colleague
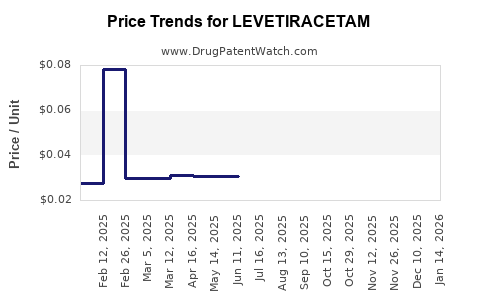
Average Pharmacy Cost for LEVETIRACETAM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LEVETIRACETAM 1,000 MG TABLET | 82009-0124-60 | 0.16348 | EACH | 2026-03-18 |
| LEVETIRACETAM 750 MG TABLET | 82009-0123-05 | 0.11512 | EACH | 2026-03-18 |
| LEVETIRACETAM 500 MG TABLET | 82009-0122-05 | 0.07693 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for LEVETIRACETAM
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| LEVETIRACETAM 100MG/ML INJ | Sagent Pharmaceuticals | 25021-0780-05 | 10X5ML | 3.52 | 2024-05-01 - 2029-04-30 | FSS | ||
| LEVETIRACETAM 1500MG/100ML 0.82% NACL INJECT | Mylan Institutional LLC | 67457-0266-10 | 10X100ML | 132.91 | 2023-09-29 - 2028-09-28 | Big4 | ||
| LEVETIRACETAM 500MG/100ML 0.82% NACL INJECTIO | Mylan Institutional LLC | 67457-0255-10 | 10X100ML | 126.63 | 2024-01-01 - 2028-09-28 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Levetiracetam Market Analysis and Price Projections
Levetiracetam is an antiepileptic drug (AED) used to treat partial-onset seizures, myoclonic seizures, and primary generalized tonic-clonic seizures. The global levetiracetam market is characterized by established generic competition, a stable demand driven by epilepsy prevalence, and price pressures. The average selling price (ASP) for levetiracetam has been declining due to increased generic penetration and the absence of significant patent exclusivities for the originator product. Future price trends are expected to remain stable or experience minor declines, contingent on manufacturing costs, regulatory changes, and the introduction of new therapeutic alternatives.
What is the Current Market Landscape for Levetiracetam?
The levetiracetam market is a mature segment within the AED therapeutic class. Dominated by generic manufacturers, the landscape reflects high competition and a focus on cost-effective production. The originator, UCB Pharma's Keppra, lost its primary market exclusivity years ago, paving the way for numerous generic entrants. This has led to a fragmented market with a wide range of suppliers offering the drug at competitive price points.
Key market characteristics include:
- Generic Dominance: Over 90% of the levetiracetam market is comprised of generic versions. This means that pricing is largely dictated by manufacturing efficiency and supply chain costs rather than proprietary innovation.
- Stable Demand: Epilepsy affects approximately 50 million people worldwide, a figure that remains relatively constant, ensuring a consistent demand for effective antiepileptic medications like levetiracetam [1].
- Price Erosion: The influx of generic manufacturers has resulted in significant price erosion. Manufacturers compete primarily on price, leading to thin profit margins for many suppliers.
- Geographic Distribution: Levetiracetam is prescribed globally. Major markets include North America, Europe, and Asia-Pacific, with demand in emerging economies projected to grow steadily due to increasing healthcare access and awareness.
- Regulatory Environment: The drug is approved by major regulatory bodies, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA), facilitating broad market access.
Who are the Key Manufacturers and Suppliers?
The levetiracetam supply chain involves multiple active pharmaceutical ingredient (API) manufacturers and finished dosage form (FDF) producers. The competitive nature of the market means that many companies offer levetiracetam products, often with variations in formulation and packaging.
Leading manufacturers and suppliers of levetiracetam API and FDFs include:
- Teva Pharmaceutical Industries Ltd.: A major generic pharmaceutical company with a significant presence in the AED market.
- Sun Pharmaceutical Industries Ltd.: An Indian multinational pharmaceutical company with a broad portfolio of generic drugs, including levetiracetam.
- Mylan N.V. (now Viatris): Another prominent player in the global generics market.
- Dr. Reddy's Laboratories: An Indian pharmaceutical company that manufactures and markets generic levetiracetam products.
- Zydus Cadila: A global pharmaceutical company headquartered in India, producing various dosage forms of levetiracetam.
- Novartis AG (Sandoz): While Sandoz is primarily a generics division, its reach and manufacturing capabilities place it as a significant supplier.
These companies, alongside numerous smaller regional manufacturers, contribute to the competitive pricing environment. The focus for these entities is on optimizing production costs, ensuring quality compliance, and maintaining robust distribution networks.
What are the Patent and Exclusivity Statuses?
The patent landscape for levetiracetam is largely expired for the core compound. UCB Pharma's initial patents for levetiracetam have long since lapsed, allowing for widespread generic manufacturing.
Key patent and exclusivity details:
- Original Compound Patent: The fundamental patent protecting the levetiracetam molecule has expired in major markets. For instance, in the United States, the last expiring patent for the original formulation of Keppra expired in the early 2010s.
- Formulation and Method of Use Patents: While the primary compound patent is expired, there may be secondary patents related to specific formulations (e.g., extended-release versions), manufacturing processes, or methods of use for particular seizure types. However, these secondary patents have generally not been strong enough to block generic competition for extended periods.
- Exclusivity Periods: Regulatory exclusivities, such as those granted by the FDA for new chemical entities (NCEs) or new uses, have also expired for the original levetiracetam product. Generic companies can therefore enter the market once these exclusivities cease.
- Orphan Drug Exclusivity: Levetiracetam does not qualify for orphan drug status for its primary indications, as epilepsy is a common condition.
The absence of strong, active patent protection on the core molecule is the primary driver for the generic nature of the levetiracetam market. This lack of intellectual property barriers allows any manufacturer meeting regulatory standards to produce and sell the drug.
How Do Manufacturing Costs and Supply Chain Dynamics Influence Pricing?
Manufacturing costs are a critical determinant of levetiracetam pricing in the current market. With a commoditized product and intense competition, efficiency in API synthesis and FDF production is paramount.
Key cost and supply chain factors:
- API Production Costs: The cost of raw materials, chemical synthesis efficiency, labor, and energy required to produce levetiracetam API are foundational to its price. Manufacturers in regions with lower labor and overhead costs, such as India and China, often have a competitive advantage in API production.
- Economies of Scale: Larger manufacturing volumes allow for greater cost efficiencies. Companies producing levetiracetam on a massive scale can amortize fixed costs over more units, leading to lower per-unit production expenses.
- Regulatory Compliance Costs: Adhering to Good Manufacturing Practices (GMP) and other regulatory requirements in target markets (e.g., FDA, EMA) adds to production costs. These costs include quality control, validation, and facility maintenance.
- Supply Chain Complexity: The global nature of pharmaceutical supply chains involves logistics, warehousing, and distribution. Fluctuations in shipping costs, tariffs, and the availability of transportation can impact the final price of the drug.
- Inventory Management: Holding adequate inventory to meet demand without incurring excessive storage costs is a constant challenge for suppliers. Efficient inventory management can contribute to cost savings.
- Geopolitical Factors: Trade relations, political stability in manufacturing regions, and currency exchange rates can also influence the cost of production and, consequently, the market price of levetiracetam.
The interplay of these factors creates a competitive pricing environment where manufacturers constantly seek ways to reduce their cost of goods sold (COGS) to remain competitive.
What are the Price Projections for Levetiracetam?
Price projections for levetiracetam indicate a trend of stability, with potential for minor declines, rather than significant increases. The market dynamics, characterized by mature generics and consistent demand, do not support substantial price inflation.
Projected price trends:
- Short-Term (1-3 years): Prices are expected to remain relatively stable. Minor fluctuations may occur due to changes in raw material costs, currency exchange rates, or the entry or exit of smaller suppliers from the market. The average selling price (ASP) for generic levetiracetam in developed markets is likely to remain in a low single-digit dollar range per daily defined dose (DDD).
- Medium-Term (3-5 years): Continued price stability or a slight downward trend is anticipated. The pressure from a large number of generic manufacturers will persist. Innovation within levetiracetam is limited to formulation improvements, which typically do not command significant price premiums after initial launch and subsequent generic entry.
- Long-Term (5+ years): The long-term outlook is subject to broader market shifts. The development of novel AEDs that offer superior efficacy, improved safety profiles, or different mechanisms of action could gradually erode levetiracetam's market share, potentially leading to further price adjustments. However, given levetiracetam's established efficacy and safety profile, it is expected to maintain a significant market presence for the foreseeable future.
Factors Influencing Future Pricing:
- Competition Intensity: The number and capacity of generic manufacturers remain the most significant price driver. An increase in manufacturers or expansion of existing ones could lead to further price erosion.
- Manufacturing Efficiency: Advances in chemical synthesis or production technology that reduce manufacturing costs could translate into lower selling prices.
- Healthcare Policy and Reimbursement: Government pricing regulations, formulary decisions by insurance providers, and reimbursement rates can influence the effective price that manufacturers receive.
- Emergence of New Therapies: The introduction of next-generation AEDs with better patient outcomes or fewer side effects could shift prescribing patterns and reduce demand for older, less differentiated drugs like levetiracetam.
- Supply Chain Disruptions: Unforeseen events, such as pandemics, geopolitical conflicts, or natural disasters, can disrupt global supply chains, potentially leading to temporary price spikes due to scarcity. However, the broad base of levetiracetam manufacturers generally mitigates the risk of prolonged shortages.
The market for levetiracetam is unlikely to see a significant resurgence in price due to its status as a well-established, off-patent generic medication. The focus for stakeholders will continue to be on market share defense through cost competitiveness and reliable supply.
How Does Levetiracetam Compare to Other Antiepileptic Drugs?
Levetiracetam occupies a specific niche within the broader AED market. Its profile makes it a frequently prescribed first- or second-line option, but it faces competition from both older and newer AEDs.
Comparative analysis:
- Efficacy and Spectrum: Levetiracetam is effective against partial-onset seizures and has a role in treating myoclonic and generalized tonic-clonic seizures. Many other AEDs, like valproic acid or lamotrigine, also have broad-spectrum activity. Newer drugs may offer improved efficacy for specific seizure types or patient populations.
- Side Effect Profile: Levetiracetam is generally well-tolerated, with common side effects including somnolence, dizziness, and behavioral changes (e.g., irritability, aggression). However, its tolerability can vary significantly among individuals. Older drugs like carbamazepine have different side effect profiles, including risks of bone marrow suppression or Stevens-Johnson syndrome. Newer AEDs may aim for improved neurological or psychiatric tolerability.
- Drug Interactions: A key advantage of levetiracetam is its minimal interaction with other medications. This is due to its non-enzymatic metabolism. Many older AEDs, such as phenytoin or phenobarbital, are potent enzyme inducers and have a high potential for drug-drug interactions. This characteristic makes levetiracetam particularly useful in patients taking multiple medications.
- Cost: As a generic drug, levetiracetam is relatively inexpensive compared to many newer AEDs that are still under patent protection or are specialty medications. For example, newer AEDs like brivaracetam (also from UCB), lacosamide, or perampanel are significantly more expensive. This cost advantage is a major factor in levetiracetam's widespread use.
- Formulations: Levetiracetam is available in immediate-release (IR) and extended-release (ER) oral formulations, as well as an intravenous (IV) formulation for acute treatment or when oral administration is not feasible. Other AEDs also offer various formulations, including chewable tablets and oral solutions, catering to different patient needs, especially pediatric populations.
Levetiracetam's combination of efficacy, a manageable side effect profile, minimal drug interactions, and low cost solidifies its position as a workhorse AED. However, the continuous development of novel therapies that offer enhanced efficacy or improved tolerability for specific seizure types presents an ongoing competitive challenge.
What are the Market Drivers and Challenges?
The levetiracetam market is influenced by a combination of factors that drive demand and present obstacles to growth or profitability.
Market Drivers:
- Increasing Epilepsy Prevalence: The global incidence and prevalence of epilepsy, while not rapidly increasing, remain substantial, providing a consistent patient base requiring antiepileptic treatment. Factors contributing to prevalence include aging populations, increased survival rates from neurological insults (e.g., stroke, head injury), and improved diagnostic capabilities [2].
- Generic Accessibility and Affordability: The availability of low-cost generic levetiracetam makes it a highly accessible treatment option, particularly in healthcare systems with budget constraints and in emerging markets.
- Physician Familiarity and Prescribing Habits: Levetiracetam has been a standard treatment option for many years, leading to widespread physician familiarity and established prescribing patterns.
- Minimal Drug-Drug Interactions: This characteristic is a significant driver for its use in patients with comorbidities or those taking multiple medications, simplifying treatment regimens and reducing the risk of adverse events.
- Availability of Multiple Formulations: The availability of IR, ER, and IV forms allows for flexible treatment approaches based on patient needs and clinical situations.
Market Challenges:
- Intense Generic Competition: The crowded generic market leads to significant price pressure and diminishing profit margins for manufacturers.
- Maturity of the Product: As a well-established drug, levetiracetam has limited potential for significant new market growth through indications or novel applications.
- Emergence of Newer AEDs: Advancements in AED research have led to the development of newer drugs with potentially better efficacy, tolerability, or specific action mechanisms, which could gradually displace levetiracetam in certain patient segments.
- Behavioral Side Effects: While generally well-tolerated, the potential for behavioral and psychiatric side effects can be a limiting factor for some patients, leading prescribers to seek alternative treatments.
- Manufacturing Cost Fluctuations: Volatility in raw material prices and global supply chain disruptions can impact production costs and profitability.
Understanding these drivers and challenges is crucial for stakeholders evaluating investment or R&D opportunities within the levetiracetam market.
Key Takeaways
Levetiracetam remains a cornerstone in epilepsy management due to its established efficacy, favorable drug interaction profile, and affordability. The market is characterized by intense generic competition, leading to stable or declining price trends. Manufacturing costs and supply chain efficiencies are primary determinants of profitability for suppliers. While new AEDs are emerging, levetiracetam's accessibility and physician familiarity ensure its continued market relevance. The absence of significant patent protection limits upside potential for price appreciation.
Frequently Asked Questions
-
What is the projected annual growth rate for the levetiracetam market? The levetiracetam market is projected to experience a compound annual growth rate (CAGR) of approximately 1% to 2% over the next five years, primarily driven by increasing prevalence of epilepsy in emerging economies and the drug's cost-effectiveness [3].
-
Will new patent filings significantly impact levetiracetam pricing? No, new patent filings related to secondary aspects such as novel formulations or manufacturing processes are unlikely to significantly impact the pricing of the core levetiracetam molecule, as the primary compound patents have long expired. These secondary patents typically do not create substantial barriers to generic entry for the base drug.
-
What is the average wholesale price (AWP) range for a standard 30-day supply of generic levetiracetam? The AWP for a 30-day supply of generic levetiracetam can range from $10 to $30, depending on the dosage, formulation (e.g., IR vs. ER), and the specific manufacturer and distributor [4]. This price has generally been declining over the past decade due to market competition.
-
Are there any significant new therapeutic alternatives that could displace levetiracetam in the near future? While newer AEDs are continually introduced with improved profiles for specific seizure types, levetiracetam's broad efficacy, low cost, and minimal drug interactions position it to remain a first- or second-line therapy for many epilepsy patients. Significant displacement is not anticipated in the immediate future, though gradual market share erosion is possible.
-
What are the primary regions with the highest levetiracetam consumption, and what drives this consumption? North America and Europe are the largest consumers of levetiracetam due to their advanced healthcare systems, high rates of epilepsy diagnosis, and well-established prescription practices. Asia-Pacific represents a significant and growing market, driven by increasing healthcare access, rising disease awareness, and a growing population [3].
Citations
[1] World Health Organization. (2023). Epilepsy. https://www.who.int/news-room/fact-sheets/detail/epilepsy
[2] Institute for Health Metrics and Evaluation. (2023). Global Burden of Disease Study 2019. http://www.healthdata.org/gbd
[3] Grand View Research. (2023). Levetiracetam Market Size, Share & Trends Analysis Report By Type (Generic, Branded), By Formulation (Tablets, Oral Solution, Injection), By Application (Partial Onset Seizures, Myoclonic Seizures, Primary Generalized Tonic-Clonic Seizures), By Region, And Segment Forecasts, 2023 - 2030. https://www.grandviewresearch.com/industry-analysis/levetiracetam-market
[4] GoodRx. (2023). Levetiracetam Prices, Coupons, and Patient Assistance Programs. Retrieved from https://www.goodrx.com/levetiracetam (Data reflects typical retail pricing and discounts)
More… ↓
