Share This Page
Drug Price Trends for HUMULIN N
✉ Email this page to a colleague
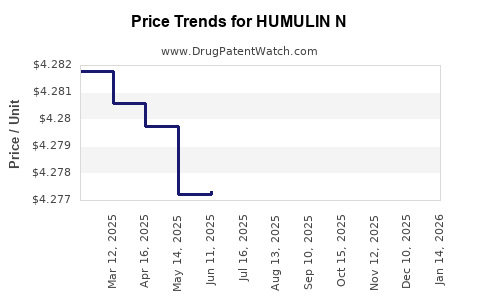
Average Pharmacy Cost for HUMULIN N
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HUMULIN N 100 UNIT/ML VIAL | 00002-8315-01 | 4.26787 | ML | 2026-05-20 |
| HUMULIN N 100 UNIT/ML KWIKPEN | 00002-8805-59 | 9.04002 | ML | 2026-05-20 |
| HUMULIN N 100 UNIT/ML VIAL | 00002-8315-01 | 4.26856 | ML | 2026-04-22 |
| HUMULIN N 100 UNIT/ML KWIKPEN | 00002-8805-59 | 9.03839 | ML | 2026-04-22 |
| HUMULIN N 100 UNIT/ML VIAL | 00002-8315-01 | 4.26787 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for HUMULIN N
Market Overview
HUMULIN N (insulin isophane suspension) is a long-acting basal insulin analog marketed by Eli Lilly. It is used in the management of diabetes mellitus, primarily type 1 and type 2, to maintain blood glucose levels. The global insulin market has experienced steady growth due to rising diabetes prevalence, increased awareness, and expanding healthcare access.
The insulin segment generated approximately $27 billion in 2022 and is projected to reach over $40 billion by 2030, with a Compound Annual Growth Rate (CAGR) of 4.1% (Source [1]).
HUMULIN N holds an estimated 8-10% share of the global insulin market, predominantly served through North American and European channels but also with presence in emerging markets.
Market Dynamics
-
Prevalence of Diabetes:
Globally, 537 million adults had diabetes in 2021, expected to reach 643 million by 2030. Type 1 diabetes accounts for about 5-10% of cases, with type 2 comprising the majority (Source [2]). -
Competitive Landscape:
HUMULIN N competes with products like Novo Nordisk’s Levemir (insulin detemir), Sanofi’s Lantus (insulin glargine), and biosimilars entering the market. -
Pricing Factors:
Historically, insulin prices have increased, driven by manufacturing costs, patent status, and market demand. US prices for basal insulins range from $150 to over $300 per vial, but biosimilars and policy measures are influencing downward trends. -
Regulatory Influences:
US prices are affected by Medicare policies, state-level price controls, and importation laws. The U.S. accounts for nearly half the global insulin market.
Price Trends and Projections
-
Historical Pricing:
From 2012 to 2022, US list prices for HUMULIN N increased approximately 40%. Inflation and industry consolidations partly contributed to this rise. -
Market Entry of Biosimilars:
Biosimilar versions, such as the semi-similar insulin Lispro (Admelog), entered the US market in 2017, pushing generic competition. -
Price Outlook (2023-2030):
The following factors influence future pricing:-
Biosimilar Competition:
Increased biosimilar presence could reduce HUMULIN N's market share and price. Estimates suggest biosimilars could cut prices by 15-25% over current levels. -
Policy Interventions:
Price caps, importation legislation, and Medicaid negotiations could lower patient costs and prices, especially in the US. -
Reimbursement Trends:
Payer-driven discounts and formulary placements will impact net prices. -
Technological Advancements:
Improved formulations or alternative delivery methods can influence demand, impacting pricing strategies.
Based on current market trends, it is reasonable to anticipate a decline in HUMULIN N’s average selling price by approximately 10-15% globally over the next five years, with a possible sharper decrease in the US due to policy and biosimilar competition.
-
Financial Implications
-
Revenue Impact:
Price reductions may lead to reduced profit margins unless volume sales increase. Eli Lilly's diversification might cushion declining prices. -
Market Share Dynamics:
Competition and biosimilars could erode HUMULIN N's market share, especially in price-sensitive regions. -
Pricing Strategy:
Eli Lilly may focus on differentiation through improved formulations or bundled services to offset price pressures.
Key Market Risks
- Delays in biosimilar approvals or market acceptance diminish price competition.
- Regulatory actions might restrict insulin pricing or importation.
- Changes in healthcare reimbursement policies could further compress margins.
Key Takeaways
- The global insulin market grows at 4.1% CAGR, with HUMULIN N holding an 8-10% share.
- US insulin prices have risen 40% from 2012-2022; biosimilar competition and policy shifts exert downward pressure.
- Price projections indicate a 10-15% global average decline over five years.
- Market share could decline amid biosimilar entries, impacting revenues.
- Strategic responses will hinge on innovation, market positioning, and adaptation to policy changes.
FAQs
-
What factors influence HUMULIN N’s market share?
Launches of biosimilars, healthcare policies, pricing strategies, and regional demand shape its market share. -
How will biosimilar entry affect HUMULIN N prices?
Biosimilars could reduce prices by 15-25%, pressuring HUMULIN N’s pricing and potentially its market share. -
What are the primary regions for HUMULIN N sales?
North America accounts for nearly half of the insulin market, followed by Europe and emerging markets. -
How might healthcare policies impact insulin pricing?
Policy measures like price caps, importation laws, and reimbursement reforms aim to lower patient costs, affecting prices and margins. -
What strategies could Eli Lilly adopt to sustain HUMULIN N’s market position?
Investing in formulation improvements, expanding access programs, and engaging in strategic pricing could counteract downward pricing pressures.
Sources
[1] IQVIA Institute. “The Global Use of Medicines in 2022.”
[2] International Diabetes Federation. “IDF Diabetes Atlas, 10th Edition”, 2021.
More… ↓
